Services on Demand
Journal
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Dental Journal
On-line version ISSN 0375-1562Print version ISSN 0011-8516
S. Afr. dent. j. vol.80 n.2 Johannesburg Mar. 2025
https://doi.org/10.17159/sadj.v80i02.20399
RESEARCH
Prevalence and risk factors of oral lesions among adults attending a dental clinic in Pietermaritzburg, KwaZulu-Natal
S ShongweI; TG GinindzaII
IDiscipline of Public Health Medicine, School of Nursing and Public Health, University KwaZulu-Natal, Durban, South Africa. ORCID: https://orcid.org/0000-0002-2423-7200
IIDiscipline of Public Health Medicine, School of Nursing and Public Health, University KwaZulu-Natal, Durban, South Africa. ORCID: https://orcid.org/0000-0002-0993-8308
ABSTRACT
INTRODUCTION: The Global Burden of Disease 2017 reported that 2.3 billion people suffered from tooth decay and untreated dental caries, the most common condition globally in permanent teeth. The prevalence and associated risk factors of oral lesions among adults remain limited in KwaZulu-Natal and South Africa.
AIMS AND OBJECTIVES: To determine the prevalence and associated risk factors of oral lesions among adults (18 years and older) attending the dental clinic for dental health care in Pietermaritzburg, KwaZulu-Natal.
DESIGN: Analytical cross-sectional study design.
METHODS: A structured questionnaire was used to obtain participants' characteristics, and an intra-oral examination was performed. Multiple regression was performed to assess the risk factors associated with oral lesions.
RESULTS: The overall prevalence of oral lesions among adults was 94.4% (95% CI: 93.8-97.8). Dental caries was more prevalent than any other oral lesion (92.9%, 95% CI: 90.3-94.8). In the multivariate analysis, age and poor oral hygiene status were associated with oral lesions.
CONCLUSION: Oral lesions are highly prevalent among adults in Pietermaritzburg; living in urban areas, poor oral hygiene and increasing age are associated with developing oral lesions. Preventative, cost-effective strategies should be implemented and promoted at all levels from early childhood to adulthood.
Keywords: Oral lesions, dental caries, oral health, oral disease, prevalence, risk factors, adults, dental clinic
BACKGROUND
Oral lesions are a significant global public health problem and negatively impact economic and health burdens.1 The presence of oral lesions in the oral cavity may result in the patient suffering from pain, discomfort and embarrassment2 A healthy oral cavity with no lesions enables the individual to socialise, masticate and communicate without hindrances.1 The World Health Organization (WHO) estimates that 3.5 billion people worldwide are affected by oral diseases, with dental caries being the most prevalent oral lesion globally3 In low- and middle-income countries (LIMICs), oral lesions persist and are commonplace despite being highly preventable. This is a reflection of the economic and social inequalities.3 Oral lesions remain untreated in many LMICs due to inadequate availability and accessibility of oral health care.1,3 The consequences of untreated oral lesions are severe, such as excruciating pain, which results in decreased productivity at work or school, reduced quality of life and may disrupt family life, and sepsis causing swellings affecting the facial tissues.3 The prevalence of oral lesions globally indicates how oral health is neglected in global health policy. 1 A national survey was conducted in Western Europe, Australia and the US, showing that oral lesions contribute to lower life satisfaction.3 The prevalence of oral lesions differs globally, ranging from 15% in Saudi Arabia4 to 51.1% in Spain.5
A wide range of diseases and disorders affect the soft and hard tissues of the mouth, including craniofacial disorders, congenital anomalies, injuries and various infections.1,3 Critical clinical conditions considered global health priorities include dental caries, periodontal diseases and oral cancers.1,3 The most common oral health burdens are global periodontal diseases and dental carious lesions.3 In most high to middle-income countries, dental carious lesions are a significant health problem, affecting 60%-90% of adults.3 In Sub-Saharan Africa, data from 39 countries shows that 90% of adults have untreated dental carious lesions.6
The high prevalence of dental carious lesions is due to increased urbanisation, increased consumption of sugars and inadequate exposure to fluorides1,3 Research done in Brazil has shown that socioeconomic status from childhood is negatively associated with untreated dental carious lesions in adulthood.7 Dental carious lesions in adults have been reported to affect 2.3 billion people globally, making it the most prevalent nonfatal, noncommunicable disease in the world.3 The prevalence of dental carious lesions in Austria has been reported to be 84% in adults between the ages of 18-19;8 in Norway, the prevalence is 90%9 and in Indonesia 85.1%.9 A study done in Greece with participants ranging from 33 to 44 years old reported a prevalence of 100%,11 an Islamic Republic of Iran study reported a prevalence of 99.2%12 and a study in Germany 94.6%.10 In Cape Town, South Africa, a total of 1885 participants underwent clinical examination from 2014-2016 and the results revealed that 98.7% of the participants had dental caries.11
Periodontal diseases may result in tooth loss and have been found to affect 5%-20% of the adult population worldwide.1,3 The International Agency for Cancer stated that oral and lip cancers are among the most common cancers in the world.1,12 The prevalence of oral and pharyngeal cancer in high-income countries is estimated to be 25 cases per 100,000.3 The increased use of tobacco and alcohol consumption has been linked to the rapid increase in the prevalence of oral cancer.1,3,12
Oral mucosal lesions appear on the mucosal surfaces of the oral cavity.13 These lesions can be caused by several aetiologies, including systematic diseases, poor oral hygiene status, injuries due to trauma and cancer, to name a few.5,14 Oral mucosal lesions affect 25%-50% of the population, depending on the population being studied.1,3 A study done in Spain reported that 51.1% of the participants enrolled in the study were diagnosed with oral mucosal lesions.5 A study done in Lebanon showed that oral mucosal lesion prevalence was 61.8%.15 In a birth cohort done in Southern Brazil on young adults, the prevalence was 23.3%.7
Studies conducted in other countries globally stated that Turkey had a prevalence of 15.5%,16 China 9.5%,17 Malaysia 9.2%18 and Iran 49.3%.19 In 2016, a study was conducted in Cape Town where 1976 participants were enrolled and 262 lesions were detected in 252 patients. The overall prevalence was 13%.20
Several risk factors have been associated with the prevalence of oral lesions. In a cohort study done in Brazil, the results showed that low socioeconomic status at birth, bad oral hygiene status and smoking habits were all associated with the presence of oral lesions.7 Other risk factors that have been investigated to have caused the presence of oral lesions include gender, employment status, residential area, educational level, oral hygiene practices, underlying medical conditions and the use of alcohol.5,7 Some research studies have shown that hormonal factors affect females more than males.16,21 Hormonal changes during pregnancy, menstruation and puberty decrease saliva volume, making the oral cavity dry and prone to oral lesions.21 A study done in Turkey revealed that the prevalence of oral lesions decreased with age. In Italy, the level of education was negatively associated with the presence of oral lesions.16 In Cape Town, South Africa, the prevalence of oral squamous cell carcinoma was 2.5 times higher in males than females.20 Underlying medical conditions such as diabetes and HIV/AIDS are risk factors for being susceptible to oral lesions.22,23 Studies have shown that oral manifestations of HIV/AIDS are prevalent and most commonly include fungal infections such as those caused by candida, necrotising gingivitis or oral hairy leukoplakia.24 A study done in Maseru, Lesotho, found that 74% of the study participants who were HIV-positive presented with one or more oral mucosal lesions.25
METHODS AND MATERIALS
Study design and population
The study participants were adults (18 years and above) who attended the dental clinic for dental services from mid-May 2021 to August 2021. All adult patients who came to the dental clinic for treatment met the inclusion criteria and provided written informed consent were included in the study. The study design is an analytical cross-sectional study that was conducted within a clinic-based sample of adults (age 18 years and above).
Sample size
To estimate the prevalence of oral lesions assuming 95% confidence and an acceptable margin of error of 5% maximum variability, ie 50% (given unknown prevalence), a sample size of the calculated sample size is 385. The sample was further increased by a margin of 10% to account for contingencies such as non-response or recording errors, and it was also increased by design effect to correct any difference in design. The sample size was multiplied by the design effect (D) of 1.3. Therefore, the final sample size needed for the study was 550 patients. Increasing the sample size reduced the type I and type II errors. The study's power (1 -β) (the % chance of detecting difference) was set at 80%.
Sampling strategy
The study participants were recruited based on the non-random consecutive sampling strategy because of the convenience and ease of access. All adult patients seeking dental treatment during the study period were recruited for the study. Informed consent was obtained from the participants, and only those patients who agreed to be part of the study and signed the consent forms were included. The investigator explained the study aims and procedures to the patients willing to participate. Before the participants signed the consent forms, the potential risks or possible discomforts associated with participating in the study were explained and discussed. After obtaining written informed consent, the recruitment of the participants began, and all were adults aged 18 years and older seeking dental services from the study sites. Recruitment continued until the desired sample size was reached. The participants who agreed to participate were assigned an anonymous participant identification number (eg P001 for patient number 1). There is no linkage between the patient's name and other identifiable information with the study findings and results, only through the participant identification number given anonymously to the patient.
Data collection procedure
A questionnaire and oral examination were done for each participant; the questionnaire was used to collect detailed data concerning sociodemographic characteristics (age, sex, race, education level and employment status), oral hygiene status (including oral infections), sexual history, medical history and other lifestyle characteristics (alcohol use and tobacco use). The oral lesions were identified based on the lesion's area in the oral cavity (location), distribution and definition, size, shape, texture, consistency, colour and history of the lesion. After that, a differential diagnosis was formed. The primary investigator did the dental examination and interview in a private dental surgery room. Each questionnaire was assigned a unique identifier number for confidentiality. The data was collected by the researcher/primary investigator and processed and analysed in Stata 16 (StataCorp College Station, Texas, US).
Quality control
Before the data collection, a pilot study was conducted at Howick Clinic to test the questionnaires. The researcher was calibrated by an expert clinician in the department before data collection. To ensure intra-examiner reliability, every fifth examination was repeated and compared to ensure consistency in the recording process. The relevant guidelines and regulations were observed and adhered to during the data collection process to the analysis of the data collected. The primary investigator undertook periodic reviews to ensure gaps during data collection.
Statistical analysis
Data was transferred from MS Excel, cleaned and analysed using Stata 16.0 SE (StataCorp College Station, Texas, US). Data were further checked for possible errors and missing values. Regular data analysis was carried out to describe the prevalence of oral lesions and distribution by age. Descriptive and inferential statistics were used for analysis. Descriptive statistics were used to present the differences in the prevalence of oral lesions by categorical variables such as sex were assessed using the Chi-square test. The logistic regression model estimated odds ratios (unadjusted and adjusted) with corresponding 95% confidence intervals for all the risk factors associated with having an oral lesion. We further estimated the marginal probabilities of experiencing oral lesions from the primary model using Stata's "margins" command. A 5% significance level was used for all the analyses.
Ethical consideration
The study was approved by the Provincial Health Research and Ethics Committee (PHREC) (KZ_202102_004) and the UKZN Biomedical Research Ethics Committee (BREC/00002280/2021), and the Ethics committee at the healthcare facility granted gatekeeper permission. The consent form used to obtain written informed consent was approved by the Ethics committees mentioned. Before the patients signed the consent forms, the study's objective, purpose and procedures were explained. Adult patients (18 years and above) attending the dental clinic were eligible to participate in the study.
RESULTS
Characteristics of the study population
The mean (± standard deviation [SD]) age for the enrolled patients was 33.10 (±13.61) years (Table I). 166 (30%) of the participants were between 20-29, and the majority (60%) were female. Of the 550 participants, 323 (58.9%) lived in the urban areas, 448 (81%) reported to have never been married, 130 (24%) were employed and 87% studied up to high/secondary school. In terms of lifestyle, 133 (24.9%) of the participants reported that they were current smokers and about 39% reported taking alcohol. Nearly 40% of the participants reported that they were on some form of treatment - the majority 133 (30.3%) were on HIV treatment. Finally, the majority of the sample, 456 (69.2%), had poor oral hygiene status and only 94 (30.8%) of the 550 patients examined had a good oral health status. Tooth brushing frequency: 288 (55.1%) brushed their teeth once a day and 260 (44.9%) brushed twice daily.
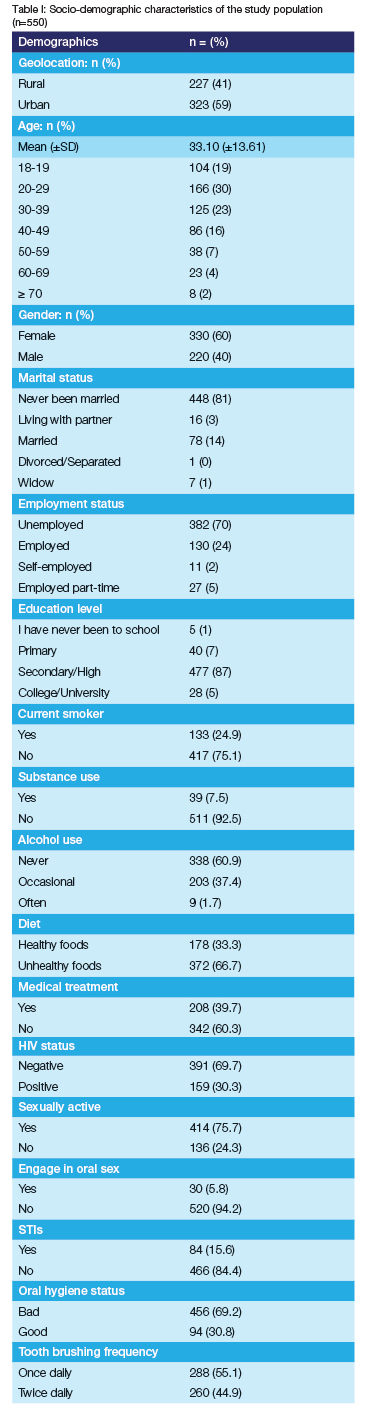
Oral lesion prevalence
The overall prevalence of oral lesions was 94.4% (95% CI: 93.8-97.8). The prevalence of oral lesions was highest among patients who live in urban areas at 59% (95% CI: 54.7-63.1) as compared to patients who live in rural areas at 41% (95% CI: 36.9-45.3) (Table II). The prevalence is significantly higher among the young age group and substantially decreases with age (p<0.001). The highest prevalence was among the 20-29 age group (29%, 95% CI: 25.7-33.6). The prevalence of oral lesions was higher among females (59%, 95% CI: 55.1-63.5) respectively. Unemployed participants had the highest prevalence, 69% (95% CI: 64.5-72.5), compared to full-time employed participants, 24% (95% CI: 21.0-28.4). Participants with a high school education had the highest prevalence at 86% (95% CI: 83.1-89.2), and those who went to college/university had the lowest at 5% (95% CI: 3.4-7.3). The prevalence rate for participants on a healthy diet was 33% (95% CI: 29.4-37.5), and those on unhealthy diets had the highest prevalence rate of 67% (95% CI: 62.5-70.6). Participants with a bad oral hygiene status had the highest prevalence rate of 69% (95% CI: 65.1-73.0), and those with good oral hygiene status had a rate of 31% (27.0-34.9). Those who brushed once daily had the highest prevalence rate of 55% (95% CI: 50.8-59.3) and those who brushed twice daily, 45% (95% CI: 40.7-49.2).
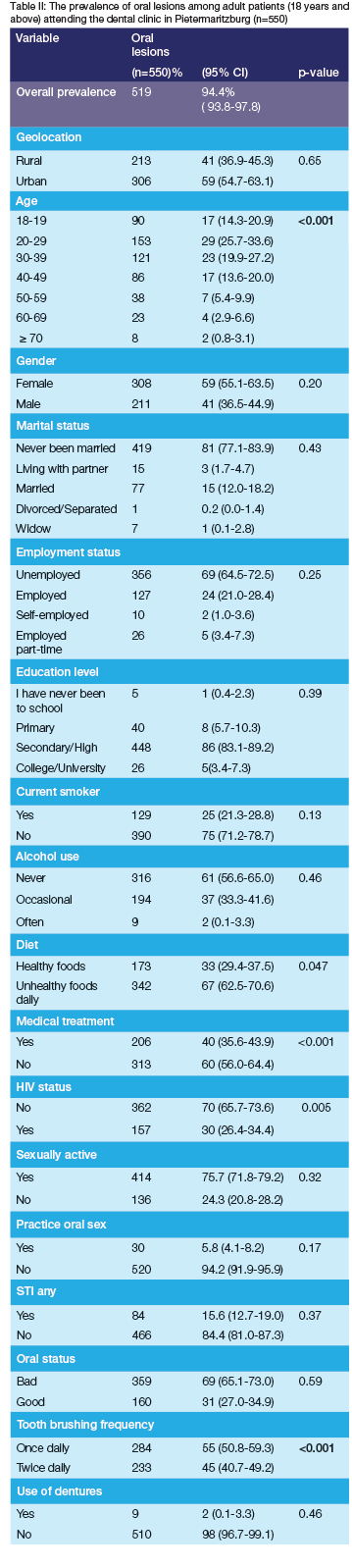
The most common oral lesions were diagnosed in the study participants.
Table III below shows the most common oral lesions diagnosed in the study participants. From the results, we see that the common superficial oral lesions in this sample include dental carious lesions 92.9% (95% CI=90.3-94.8), gingivitis 30.4% (95% CI=26.6-34.5), periodontics 5.4% (95% CI=3.7-7.7) and dental abscess 3.7% (95% CI=2.3-5.7).
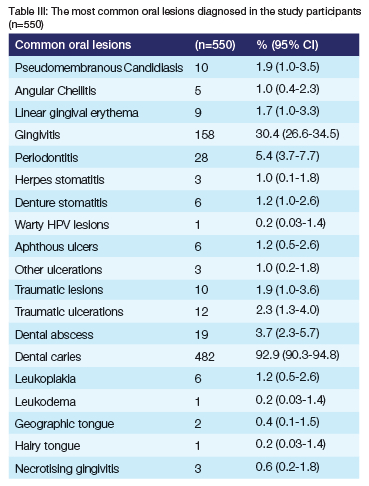
Risk factors associated with the presence of oral lesions among adults
Based on the univariate analysis: increasing age (18-19yrs: OR=0.09, 95% CI: 0.03-0.29, p=<0.001; 20-29yrs OR=0.17 95% CI: 0.05-0.53 p=0.002), practising good oral hygiene (OR=0.02, 95% CI 0.04-0.1, p=<0.001) and brushing teeth twice daily (OR=0.12, 95% CI 0.04-0.35, p=<0.001) were inversely associated with oral lesion (Table IV). Living in urban areas was associated with an increased risk of having an oral lesion (OR=3.35 95% CI 1.08-10.4, p=0.037). Even after multivariable adjustment, the risk of oral lesion increased with increasing age (18-19yrs: OR=0.07 95% CI: 0.01-0.45 p=0.005; 20-29yrs: OR=0.18 95% CI: 0.04-0.85 p=0.030). Those practising good oral hygiene (OR=0.013 95% CI 0.01-0.06, p<0.001) and brushing their teeth twice daily (OR=0.18 95% CI 0.05-0.67, p=0.011) had a decreased risk of having an oral lesion.
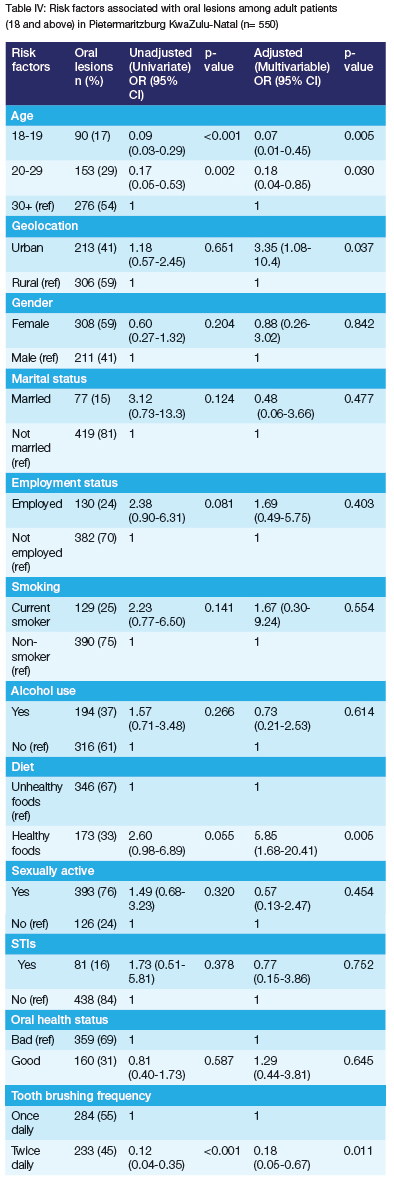
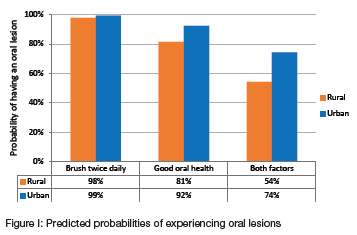
Predicted probabilities of experiencing oral lesions
Figure 1 shows the results of the adjusted predicted probabilities of having an oral lesion at each value of the main predictor variables statistically significant in the primary model in Table 3. Brushing teeth twice daily reduces the probability of having an oral lesion by a small amount, with over 98% probability of having an oral lesion regardless of location. Practising good oral hygiene only and living in rural areas reduces the chances of having an oral lesion by 19%, with an 81% probability, compared to those residing in urban areas, with a 92% probability. Brushing teeth twice daily and practising good oral hygiene for those living in rural areas reduces the chances of having an oral lesion by nearly half, with about 53% probability of having an oral lesion compared to 74% for those living in urban areas.
DISCUSSION
This is the first study assessing the prevalence and risk factors for oral lesions in Pietermaritzburg, KwaZulu-Natal. The present study shows a high prevalence (94.4%) of oral lesions in adults aged 18 years and above. The prevalence of oral lesions was high among participants aged 20-29 years (29%) and 30-39 (23%). The prevalence of oral lesions was higher among females (59%) than males (41%). Participants who reside in the urban areas were shown to be more prone to having oral lesions (59%) than those patients who reside in the rural areas (41%). Study participants were examined intra-orally to check the health of the oral cavity; 69% of the participants had poor/bad oral hygiene status, and only 31% had good oral hygiene status. The participants were asked about the frequency of brushing their teeth, and 55% stated that they brush once daily, and 45% of the participants said they brush twice daily. The univariate analysis showed that increasing age and living in an urban area were all associated with an increased risk of having an oral lesion. Practising good oral hygiene and brushing twice daily was associated with a reduced oral lesion risk. Even after multivariable adjustment, the risk of oral lesion increased with age 18-19 years OR=0.07 and 20-29 years OR=0.013. The most common oral lesions that were diagnosed in this study were dental carious lesions (92.9%), gingivitis (30.4%), periodontitis (5.4%) and dental abscesses (3.7%). Practising good oral hygiene only and living in rural areas reduces the chances of having an oral lesion by 19%, with an 81% probability, compared to those residing in urban areas, with a 92% probability.
There have been many studies done around the world on the prevalence of oral lesions and, compared to those studies, the prevalence of oral lesions in this study shows that this is a public health concern. Other researchers have reported the prevalence of oral lesions ranging from 23.5% to 86.6%. This study's prevalence was very high at 94.4%, indicating that preventative dental strategies in the public sector must be utilised due to multifactorial challenges. The overall prevalence of oral lesions in Vietnam was 14%,26 Saudi Arabia 15.0%,4 Iran 19.4%,19 Mexico 23.2%,27 Brazil 23.3%,7 US 27.9%28 and Hong Kong the prevalence was 36%.29 A few countries reported a prevalence of more than 50% - Chile reported a prevalence of 53%,30 Slovenia 61.6%,31 Lebanon 61.8%15 and in Cape Town, South Africa, the overall prevalence was 60.4%.32 The variations in prevalence may be due to the difference in populations being studied, sample size, geographic location, age, the classification system used to classify the oral lesions, methods and the diagnostic criteria used.1,3
In this study, oral lesion prevalence was highest among participants aged 20-29 years (29%). In Iran, the reported prevalence among the 20-29-year-old age group was 34.5%;19 in Saudi Arabia, the 21-30-year-old group had a 17.8% prevalence;4 in Cape Town, the prevalence was 18% in the >24 years age group and 21% among the 25-34 age group.20 In Iran, the high prevalence of oral lesions was in the 30-40 age group, which was 53.9%.19 A study on the adult Chinese population reported that in the 35-44-year-old category, the overall prevalence was 38%.33 This study reported the prevalence of oral lesions at 23% in the 31-40 age group. The differences in the age groups may be due to lack of access and affordability of dental treatment, bad oral hygiene, not practising good oral hygiene habits such as tooth brushing, eating unhealthy foods high in sugars and carbohydrates and underlying medical conditions such as HIV.1,3,34 This study reported the prevalence of oral lesions among participants living in urban areas (59%) than in rural areas (41%). This is contrary to a study done in Australia, where oral lesions were prevalent among participants who resided in rural areas.35 In China, the prevalence was equal among urban and rural areas.29 The difference in prevalence may be due to the accessibility of oral healthcare facilities and the affordability of the services.35 Based on this study, the prevalence of oral lesions was higher among female participants (59%) than male participants (41%). A study done in Saudi Arabia also reported the prevalence of oral lesions was higher among females (57.7%) than males (42.3%)4 and in Brazil, the prevalence was higher among male participants, 25.1%, than females 21.2%.7 The differences are due to the different variables studied in the study populations and the overall number of participants based on gender.15 Hormonal levels in females during pregnancy or menstruation have been linked to decreased salivary production, making the oral cavity susceptible to oral lesions.11 In this study, 30% of the HIV-positive participants presented with at least one oral lesion. The oral manifestations of HIV were also present in some of the participants who were HIV positive; all the HIV-positive participants stated to be on treatment. The use of antiretroviral treatment decreases the chances of being exposed to opportunistic infections in HIV-positive patients as it decreases the viral load and increases the CD4 count.32 The most common oral lesion that is linked to HIV in this study was pseudomembranous candidiasis (1.9%). In a southeastern clinic in the US, 35.8% of the study participants who were HIV positive were diagnosed with at least one oral lesion (31). In Southeast Iran, 57.1% of the study participants were diagnosed with pseudomembranous candidiasis and 34.1% with linear gingival erythema.36 The highest prevalence of candida was reported in Mali, where 95% of the population had candidiasis.37 In Johannesburg, South Africa, the prevalence of oral mucosal lesions in HIV-positive adults was 21.2%, with linear gingival erythema being the most common one (13.8%), necrotising gingivitis 2.2% and pseudomembranous candidiasis 1.7%.34 In Cape Town, 37.8% of the 600 participants were diagnosed with candida lesions.32 Candida is not only found in HIV-positive patients but can also be found in patients who have been on antibiotics for long periods, asthmatic patients on steroid inhalers, diabetics and mouth breathers.38 This study's highest prevalence of oral lesions was dental carious lesions. The prevalence was 92.9%. These unacceptable levels of caries, according to WHO, are higher than in other African countries and are comparable to other LICs and MICs in South America, such as Brazil, Colombia and Peru.1,3 The prevalence of caries varies depending on the population being studied and the country's socioeconomic status.1,3 In 2015, the age-standardised prevalence of dental caries globally was 48.0%, the largest peak at age 25 years.39 In a study done in China, the prevalence was 41.15%,40 Brazil 72.9%,41 Kosovo 72.80%42 and Eastern Mediterranean 70%.43 In Africa, in countries such as Ethiopia the prevalence of dental caries was 40.98%,44 50.3% in Kenya45 and in East Africa, the prevalence was 45.7%.6 The pooled prevalence was high in Eritrea, 65.2%, followed by Sudan, 57.8% and a low prevalence was found in Tanzania 30.7%.6 In Johannesburg, South Africa, the prevalence of dental caries was 56.76%. The presence of dental caries was directly related to the prevalence of oral mucosal lesions.34. In 2019, as part of the Vascular and Metabolic Health Study, 1885 participants in Bellville, Cape Town were examined for decayed, missing and filled teeth scores.11 The study results showed that dental caries' prevalence was 93.7%.11
The prevalence of gingivitis in this study was 30.4%. The prevalence of gingivitis varies from 50%-100%.1,3 In North Jordan, the prevalence of gingivitis was 76% (50); Saudi Arabia had the highest prevalence, with 100% of the study population between the ages of 18-40 years presenting with gingivitis.46 Countries such as Nepal and Australia had nearly similar estimates of the prevalence of gingivitis as in this current study: Nepal had a prevalence of 40%47 and Australia 28.8%.47 In Cape Town, South Africa, 8.5% of the participants presented with gingival/periodontal lesions.20 In this study, the prevalence of a dental abscess was 3.7%, which globally has been reported to be between 5% and 46%.48 In a study in Pakistan in 2014, it was 1.14% for dental abscesses.48 Dental abscesses form when dental carious lesions are left untreated; they can also be caused by trauma or periodontitis.48 This study's prevalence of periodontal lesions was 5.4%, comparable with Nigeria's 6.9%.49.The prevalence of periodontal lesions ranges from 20%- 50%.1,3 The prevalence of periodontal lesions in other countries is very high, especially in HICs such as Germany, where 73% of the 34 to 44-year-olds were found to have periodontallesions,50 in Nepal 64%,51 Poland 63%,52 Malaysia 60%,53 Iran54 and Taiwan 53%.55 The prevalence of periodontitis is high among adults aged between 34 and 90 years.56 Periodontitis causes tooth mobility, leading to the eventual extraction of the loose tooth/teeth. This causes problems when chewing and speaking, which causes a low quality of life and self-confidence.57 The main causes of periodontitis are poor oral hygiene, underlying systematic diseases such as diabetes and limited access to preventative dental services.58
This study showed that increasing age was a risk factor for developing oral lesions. Similarly, a study done in Saudi Arabia noted an association between oral lesions and increasing age.4 The prevalence of oral lesions tends to increase with age. This is caused by unhealthy dietary habits, knowledge and attitude toward the oral health prevention programmes in place and oral hygiene practices.6,40 Also, in this study living in the urban areas was associated with oral lesions, where participants residing there had a 3.4 chance of developing an oral lesion. In India, the prevalence among those living in urban areas was low compared to those in rural areas.14 The reasons for these differences are the methodology used in each study, the sample size and the population being studied.5 In this current study, 69% of the participants had a poor oral hygiene status; 31% had an excellent oral hygiene status and tooth brushing frequency. In this study, 55% brushed once daily and 45% brushed twice daily. In the US, the effects of age and oral hygiene showed an increase in the prevalence of oral lesions; throughout all age groups, the prevalence is much higher in participants with poor oral hygiene than those with good oral hygiene status; more than 95% of the participants with good oral hygiene status did not present with any oral lesions 59. In India, 53% of the participants claimed to brush only once a day, while 38% claimed to brush twice.14 In Nigeria, 12.5% of participants with at least one oral lesion had a good oral hygiene status, while 19.1% had a foul one.49 The systematic review in EA found poor tooth brushing habit OR=1.967 was an independent risk factor of dental lesions.6 In Johannesburg, the study discovered that lack of access and availability of dental services, poor oral hygiene and social and demographic challenges were all contributing factors, as those participants with good oral hygiene behavioural habits were associated with fewer dental caries occurrences.34 The study also reported that the frequency of tooth brushing determines the occurrence of dental lesions, especially carious lesions. The odds of developing dental lesions were 1.5 times higher among those who reported brushing their teeth less than those who brush twice daily (OR: 1.542), and the odds were 1.5 higher for those who consumed sweetened diets on a daily basis.34
Strengths and limitations of the study
The use of primary data was one of the strengths of this study, as it made the data collected authentic and up to date. The information presented in this study is necessary for the promotion of preventative oral health care, and it also provides a platform for further research on oral lesions. The associated risk factors mentioned in this study can be used to educate the community and strengthen community capacity in promoting oral hygiene. This is the first study done on the prevalence of oral lesions in Pietermaritzburg, KwaZulu-Natal. It is the first; therefore, it gives perspective on the burden of oral lesions in PMB, KZN and South Africa as a whole. This study also serves as a benchmark for further research on the risk factors linked to causing oral lesions and how they play a role in causing oral lesions.
This study also has a couple of limitations. The study's results can only be generalised to the patients attending the dental clinic in PMB as this study was hospital-based; the patients were all seeking dental-related healthcare services. The majority wanted their teeth extracted because of pain due to untreated dental caries. It was impossible to identify and establish cause-and-effect relationships as it was a cross-sectional study. This cross-sectional study did not distinguish between acute and chronic oral lesions presented by the participants. One of the limitations of this study is that while intra-examiner reliability was assessed, the results were not reported; therefore, there was no analysis of the intra-examiner reliability. The final diagnosis of some oral mucosal lesions was based only on clinical assessment and not laboratory analysis. The study occurred on weekdays, which may have resulted in selection bias regarding patients employed and working during the week.
CONCLUSION
The studied population presented a high burden of oral lesions, specifically dental carious lesions. The results highlight the limited preventative oral healthcare and the high rate of tooth extractions due to untreated carious teeth. Most dental public health care facilities offer curative oral health care due to the lack of equipment and materials to perform preventative procedures such as fillings and fissure sealants. The findings of this study show that poor oral hygiene status and the lack of basic oral hygiene practices such as tooth brushing have a detrimental impact on oral cavity health. Prevention-orientated programmes should be planned and implemented and made readily available at all primary healthcare facilities, making them accessible to everyone, including patients in rural areas. Preventative programmes such as oral health promotions and education should be implemented at all levels, such as schools and workplaces, as good oral hygiene practices must start at a young age.
Abbreviations
AIDS Acquired Immunodeficiency Syndrome
AOR Adjusted odds ratio
ARVs Antiretroviral treatment
BREC Biomedical Research Ethics Committee
CI Confidence Interval
COR Crude odds ratio
HAART Highly active antiretroviral therapy
HICs High-income countries
HIV Human Immunodeficiency Virus
LICs Low-income countries
MICs Middle-income countries
MS Microsoft
OR Odds Ratio
PHREC Provincial Health Research Ethics Committee
SD Standard deviation
SSA Sub-Saharan Africa
STI Sexually transmitted infections
UKZN University of KwaZulu-Natal
US United States of America
WHO World Health Organization
ACKNOWLEDGEMENTS
The authors would like to thank the University of KwaZulu-Natal, Discipline of Public Health Medicine, for the provision of resources towards the study. The authors would also like to thank the Northdale Hospital administration and the KwaZulu-Natal Provincial Department of Health for allowing us to implement the research and the patients who agreed to participate.
Funding
This research received no funding from public or private agencies; the study was self-funded.
Availability of data and materials
Data from this study is the property of the KwaZulu-Natal Provincial Department of Health and the University of KwaZulu-Natal and cannot be publicly available. All interested readers can access the data set from the KZN Provincial Health Research and Ethics Committee (PHREC) and the University of KwaZulu-Natal Biomedical Research Ethics Committee (BREC) from the following contacts: Health Research and Knowledge Management Unit, Private Bag X9050, 330 Langalibalele Str, Pietermaritzburg 3201 Tel: 033 395 3189/3123/2805, Fax: 033 394 3782, Email hrkm@kznhealth.gov.za. The chairperson, Biomedical Research Ethics Administration, Research Office, Westville Campus, Govan Mbeki Building, University of KwaZulu-Natal P/Bag X54001, Durban 4000, KwaZulu-Natal, South Africa, Tel: 031 260 4769 Fax: 031 260 4609 Email: BREC@ ukzn.ac.za.
Conflict of interest
The authors declare there are no conflicts of interests
Consent for publication
Not applicable
REFERENCES
1. Petersen PE, Bourgeois D, Ogawa H, Estupinan-Day S, Ndiaye C. The global burden of oral diseases and risks to oral health. Bulletin of the World Health Organization. 2005;83:661-9 [ Links ]
2. Saraswathi T, Ranganathan K, Shanmugam S, Sowmya R, Narasimhan PD, Gunaseelan R. Prevalence of oral lesions about habits: Cross-sectional study in South India. Indian journal of dental research. 2006;17(3):121 [ Links ]
3. Peres MA, Macpherson LM, Weyant RJ, Daly B, Venturelli R, Mathur MR, et al. Oral diseases: a global public health challenge. The Lancet. 2019;394(10194):249-60 [ Links ]
4. Al-Mobeeriek A, AlDosari AM. Prevalence of oral lesions among Saudi dental patients. Annals of Saudi medicine. 2009;29(5):365-8 [ Links ]
5. Garcia-Pola Vallejo M, Martinez Diaz-Canel A, Garcia Martin J, Gonzalez Garcia M. Risk factors for oral soft tissue lesions in an adult Spanish population. Community dentistry and oral epidemiology. 2002;30(4):277-85 [ Links ]
6. Teshome A, Muche A, Girma B. Prevalence of Dental Caries and Associated Factors in East Africa, 2000-2020: Systematic Review and Meta-Analysis. Frontiers in Public Health. 2021;9 [ Links ]
7. Tarquinio SBC, Oliveira LJCd, Correa MB, Peres MA, Peres KG, Gigante DP, et al. Factors associated with prevalence of oral lesions and oral self-examination in young adults from a birth cohort in Southern Brazil. Cadernos de saude publica. 2013;29:155-64 [ Links ]
8. Bernabé E, Sheiham A, Sabbah W. Income, income inequality, dental caries and dental care levels: an ecological study in rich countries. Caries research. 2009;43(4):294-301 [ Links ]
9. Suratri MAL, Tjahja I, Setiawaty V. Correlation between dental health maintenance behavior with Dental Caries Status (DMF-T). Bali Medical Journal. 2018;7(1):56-60 [ Links ]
10. Carvalho JC, Schiffner U. Dental caries in European adults and senior citizens 1996-2016: ORCA Saturday Afternoon Symposium in Greifswald, Germany - part II. Caries research. 2019;53(3):242-52 [ Links ]
11. Chikte U, Pontes CC, Karangwa I, Kimmie-Dhansay F, Erasmus R, Kengne AP, et al . Dental caries in a South African adult population: findings from the Cape Town Vascular and Metabolic Health Study. International Dental Journal. 2020;70(3):176-82 [ Links ]
12. Higginson J. The International Agency for Research on Cancer: a brief review of its history, mission, and program. Toxicological Sciences. 1998;43(2):79-85 [ Links ]
13. Axéll T, Zain RB, Siwamogstham P, Tantiniran D, Thampipit J. Prevalence of oral soft tissue lesions in out-patients at two Malaysian and Thai dental schools. Community Dentistry and Oral Epidemiology. 1990;18(2):95-9 [ Links ]
14. Goyal R, Jadia S, Jain L, Agarawal C. A clinical study of oral mucosal lesions in patients visiting a tertiary care centre in central India. Indian Journal of Otolaryngology and Head & Neck Surgery. 2016;68(4):413-6 [ Links ]
15. El Toum S, Cassia A, Bouchi N, Kassab I. Prevalence and distribution of oral mucosal lesions by sex and age categories: A retrospective study of patients attending lebanese school of dentistry. International Journal of Dentistry. 2018;2018 [ Links ]
16. Mumcu G, Cimilli H, Sur H, Hayran O, Atalay T. Prevalence and distribution of oral lesions: a cross-sectional study in Turkey. Oral Diseases. 2005;11(2):81-7 [ Links ]
17. Feng J, Zhou Z, Shen X, Wang Y Shi L, Wang Y, et al. Prevalence and distribution of oral mucosal lesions: a cross-sectional study in Shanghai, China. Journal of Oral Pathology & Medicine. 2015;44(7):490-4 [ Links ]
18. Zain RB, Ikeda N, Razak IA, Axéll T, Majid ZA, Gupta PC, etal. A national epidemiological survey of oral mucosal lesions in Malaysia. Community dentistry and oral epidemiology. 1997;25(5):377-83 [ Links ]
19. Ghanaei FM, Joukar F, Rabiei M, Dadashzadeh A, Valeshabad AK. Prevalence of oral mucosal lesions in an adult Iranian population. Iranian Red Crescent Medical Journal. 2013;15(7):600 [ Links ]
20. Pontes CC, Chikte U, Kimmie-Dhansay F, Erasmus RT, Kengne AP, Matsha TE. Prevalence of Oral Mucosal Lesions and Relation to Serum Cotinine Levels -Findings from a Cross-Sectional Study in South Africa. International Journal of Environmental Research and Public Health. 2020;17(3):1065 [ Links ]
21. Lukacs JR, Largaespada LL. Explaining sex differences in dental caries prevalence: Saliva, hormones, and "life-history" etiologies. American Journal of Human Biology: The Official Journal of the Human Biology Association. 2006;18(4):540-55 [ Links ]
22. Chi AC, Neville BW, Krayer JW, Gonsalves WC. Oral manifestations of systemic disease. American family physician. 2010;82(11):1381-8 [ Links ]
23. Askinytè D, Matulionytè R, Rimkevidus A. Oral manifestations of HIV disease: A review. Baltic Dental and Maxillofacial Journal. 2015;17:21-8 [ Links ]
24. Butt F, Chindia M, Vaghela V, Mandalia K. Oral manifestation of HIV/AIDS in a Kenyan provincial hospital. East African Medical Journal. 2001;78(8):398-401 [ Links ]
25. Kamiru H, Naidoo S. Oral HIV lesions and oral health behaviour of HIV-positive patients attending the Queen Elizabeth II Hospital, Maseru, Lesotho. SADJ: journal of the South African Dental Association= tydskrif van die Suid-Afrikaanse Tandheelkundige Vereniging. 2002;57(11):479-82 [ Links ]
26. Nair R, Samaranayake L, Philipsen H, Graham R, Itthagarun A. Prevalence of oral lesions in a selected Vietnamese population. International Dental Journal. 1996;46(1):48-51 [ Links ]
27. Castellanos JL, Díaz-Guzmán L. Lesions of the oral mucosa: an epidemiological study of 23785 Mexican patients. Oral Surgery, Oral Medicine, Oral Pathology, Oral Radiology, and Endodontology. 2008;105(1):79-85 [ Links ]
28. Patton L, McKaig R, Strauss R, Jr JE. Oral manifestations of HIV in a southeast USA population. Oral diseases. 1998;4(3):164-9 [ Links ]
29. Corbet EF, Holmgren CJ, Philipsen HP. Oral mucosal lesions in 65-74-year-old Hong Kong Chinese. Community dentistry and oral epidemiology. 1994;22(5):392-5 [ Links ]
30. Espinoza I, Rojas R, Aranda W, Gamonal J. Prevalence of oral mucosal lesions in elderly people in Santiago, Chile. Journal of Oral Pathology & Medicine. 2003;32(10):571-5 [ Links ]
31. Kovač-Kavčič M, Skalerič U. The prevalence of oral mucosal lesions in a population in Ljubljana, Slovenia. Journal of Oral Pathology & Medicine. 2000;29(7):331-5 [ Links ]
32. Arendorf TM, Bredekamp By, Cloete CAC, Sauer G. Oral manifestations of HIV infection in 600 South African patients. Journal of Oral Pathology & Medicine. 1998;27(4):176-9 [ Links ]
33. Lin H, Corbet E, Lo E. Oral mucosal lesions in adult Chinese. Journal of Dental Research. 2001;80(5):1486-90 [ Links ]
34. Malele Kolisa Y, Yengopal V, Shumba K, Igumbor J. The burden of oral conditions among adolescents living with HIV at a clinic in Johannesburg, South Africa. Plos One. 2019;14(10):e0222568-e [ Links ]
35. Do L, Spencer A, Dost F, Farah C. Oral mucosal lesions: findings from the Australian National Survey of Adult Oral Health. Australian Dental Journal. 2014;59(1):114-20 [ Links ]
36. Saravani S, Nosratzehi T, Mir S. Oral manifestations and related factors of HIV positive patients in south-east of Iran. Journal of Dental Materials and Techniques. 2017;6(1):11-8 [ Links ]
37. Tamí-Maury I, Coulibaly YI, Cissoko SS, Dao S, Kristensen S. First report of HIV-related oral manifestations in Mali. Pan African Medical Journal. 2012;11(1) [ Links ]
38. Hu L, He C, Zhao C, Chen X, Hua H, Yan Z. Characterization of oral candidiasis and the Candida species profile in patients with oral mucosal diseases. Microbial pathogenesis. 2019;134:103575 [ Links ]
39. Costa SM, Martins CC, Bonfim MdLC, Zina LG, Paiva SM, Pordeus IA, et al . A systematic review of socioeconomic indicators and dental caries in adults. International Journal of Environmental Research and Public Health. 2012;9(10):3540-74 [ Links ]
40. Xuedong Z. Dental caries: principles and management: Springer; 2015 [ Links ]
41. Narvai PC, Frazão P, Roncalli AG, Antunes J. Dental caries in Brazil: decline, polarization, inequality and social exclusion. Revista Panamericana de Salud Publica. 2006;19(6):385-93 [ Links ]
42. Kamberi B, Koçani F, Begzati A, Kelmendi J, Ilijazi D, Berisha N, et al . Prevalence of dental caries in Kosovar adult population. International Journal of Dentistry. 2016;2016 [ Links ]
43. da Silveira Moreira R. Epidemiology of dental caries in the world. Oral Health Care -Pediatric, Research, Epidemiology and Clinical Practices. 2012;8:149-68 [ Links ]
44. Wondwossen F, Ästrom AN, Bjorvatn K, Bárdsen A. The relationship between dental caries and dental fluorosis in areas with moderate- and high-fluoride drinking water in Ethiopia. Community dentistry and oral epidemiology. 2004;32(5):337-44 [ Links ]
45. Manji F, Fejerskov O, Baelum V Pattern of dental caries in an adult rural population. Caries research. 1989;23(1):55-62 [ Links ]
46. Idrees MM, Azzeghaiby SN, Hammad MM, Kujan OB. Prevalence and severity of plaque-induced gingivitis in a Saudi adult population. Saudi Medical Journal. 2014;35(11):1373 [ Links ]
47. Stamm JW. Epidemiology of gingivitis. Journal of Clinical Periodontology 1986;13(5):360-6 [ Links ]
48. Sanders JL, Houck RC. Dental Abscess. StatPearls [Internet]. 2020 [ Links ]
49. Agbelusi G, Wright A. Oral lesions as indicators of HIV infection among routine dental patients in Lagos, Nigeria. Oral diseases. 2005;11(6):370-3 [ Links ]
50. Deinzer R, Micheelis W, Granrath N, Hoffmann T. More to learn about: periodontitisrelated knowledge and its relationship with periodontal health behaviour. Journal of Clinical Periodontology. 2009;36(9):756-64 [ Links ]
51. Pradhan S, Goel K. Interrelationship between diabetes and periodontitis: a review. JNMA; Journal of the Nepal Medical Association. 2011;51(183):144-53 [ Links ]
52. Tomczyk J, Myszka A, Borowska-Strugińska B, Zalewska M, Turska-Szybka A, Olczak-Kowalczyk D. Periodontitis in the historical population of Radom (Poland) from the 11th to 19th centuries. International Journal of Osteoarchaeology. 2018;28(4):397-406 [ Links ]
53. Mohd Dom TN, Ayob R, Abd Muttalib K, Aljunid SM. National economic burden associated with management of periodontitis in Malaysia. International Journal of Dentistry. 2016;2016 [ Links ]
54. Rahimi A, Hadavi MM. Relationship between periodontitis and cardiovascular disease. ARYA atherosclerosis. 2020;16(6):306 [ Links ]
55. Mu H-C, Su N-Y Huang J-Y Lee S-S, Chang Y-C. Trends in the prevalence or periodontitis in Taiwan from 1997 to 2013: A nationwide population-based retrospective study. Medicine. 2017;96(45) [ Links ]
56. Persson GR. Dental geriatrics and periodontitis. Periodontology 2000. 2017;74(1):102-15 [ Links ]
57. Flemmig TF. Periodontitis. Annals of periodontology. 1999;4(1):32-7 [ Links ]
58. Taylor GW, Borgnakke WS. Periodontal disease: associations with diabetes, glycemic control and complications. Oral diseases. 2008;14(3):191-203 [ Links ]
59. Dye BA, Albino J, D'Souza RN. Oral health problems are global and need to be addressed in the USA. The Lancet. 2022;399(10320):127-8 [ Links ]
 Correspondence:
Correspondence:
Name: Sinegugu Shongwe
Tel: +27 31 2604214
Email: shongweS2@ukzn.ac.za
Author's contribution
1. Sinegugu Shongwe - principal researcher (40%), study design and conception (20%), data analysis and interpretation (20%), writing of article (15%), drafting of manuscript (5%)
2. Themba G Ginindza - simplifying data acquisition (20%), critical review (20%), final approval of manuscript (10%), drafting of manuscript (5%)













