Services on Demand
Journal
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Dental Journal
On-line version ISSN 0375-1562Print version ISSN 0011-8516
S. Afr. dent. j. vol.80 n.4 Johannesburg May. 2025
https://doi.org/10.17159/sadj.v80i04.22714
REVIEW
U Ripamonti
MD, DDS, MDent, MFS, PhD (Med), FBSE The Department of Internal Medicine. School of Clinical Medicine. Faculty of Health Sciences University of the Witwatersrand, Johannesburg 2193 Parktown, South Africa ORCID Number: 0000-0002-65673594
ABSTRACT
This contribution to the induction of tissue formation starts with seemingly simple questions, "Why Bone?" and "Why Cartilage?", the essential ingredients to compose the skeleton ad thus the speciation of the vertebrates, the induction of long bone via endochondral ossification, the induction of the growth plate, body erection and the speciation thus of the Homo clade, walking upright toward the spectacular creativity of extant Homo sapiens. The title wishes to pay tribute to grand pioneer scientists such as Pollettini, Levander, Moss, Urist and Reddi who persevered to study the induction of bone formation as initiated by devitalized demineralized bone matrices. "Tissue Induction" is the title of a seminal paper by Gustav Levander in Nature, 1945. Levander hypothesized that unknown substances from heterotopically implanted bone matrices would activate recipient resident cells to initiate the induction of bone formation, where there is no bone. Levander went further by using the term "Tissue Induction" linking the induction of bone formation to embryonal development as described by Hans Spemann and Hilde Mangold, the 1935 Nobel Prize for Medicine and Physiology. Phylogenetically, bones were an ancestral character, and cartilage developed later, providing the growth plate, to growth vertebrate' long bones establishing body erection in selected hominid' clades. The TGF-β supergene family includes several osteogenic proteins endowed with the remarkable capacity to initiate the heterotopic induction of bone. Besides the sub-family of the bone morphogenetic proteins (BMPs), in primates and in primates only, the three mammalian TGF-β isoforms also initiate the induction of bone formation. Heterotopic implantation of recombinant hTGF-ß3 initiates the induction of bone formation by priming resident intramuscular cells, pericytes, myoendothelial cells and myoblastic cells to express and secrete BMPs genes and gene products; the expression and synthesis of BMPs initiate the induction of bone formation regulated by Noggin expression. Combined morphological and molecular analyses have indicated that doses of hTGF-ß3 in Matrigel®Matrix set into motion the in vivo development of multiple tissues and multicellular organoids within the implanted furcation bioreactors. Organoids form by gene expression pathways from available different cellular populations within the exposed furcation bioreactor. Our molecular and morphological data using undecalcified whole mounted sections cut by the Exakt diamond saw technique have indicated that hTGF-ß3 in Matrigel®Matrix induces distinct supracellular phases that together with morphological transformation and organogenesis result in the generation of intramuscular mineralized bone organoids.
The generation of transformed periodontal bioreactors into organogenesis of alveolar bone is connected to a highly vascularized periodontal ligament system patterned by newly generated collagenic fibers. These attach into substantial cementogenesis with capillary sprouting and angioblastic activity that result in cementogenesis in angiogenesis with de novo cementoid formation.
INTRODUCTION: "WHY BONE?"
With a seemingly simple question, Romer1 asks: "Why bone?". This contribution to "Tissue Induction" would like to ask another seemingly simple question, that is "Why cartilage?".
These altogether interesting and certainly difficult question proposes the first digital images of the manuscript, the extraordinary induction of chondrogenesis by a coral-derived macroporous hydroxyapatite-based bioreactor when implanted solo in the dorsal musculature of the Selachian' fish, the dusky shark Carcharhinus obscurus (Fig. 1).2,3
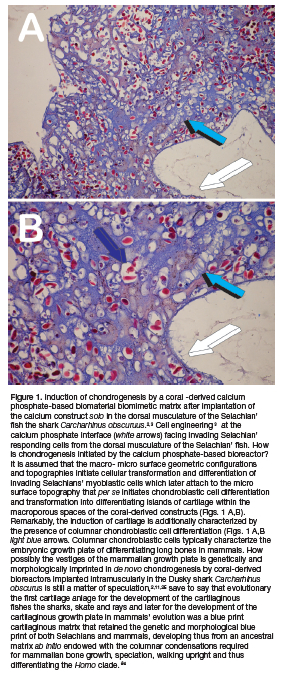
The images presented in Figure 1 morphologically show the emerging era of cell engineering. 4 The work of Lim in Science proposes the new era of cell engineering whereby cells are used as building blocks to initiate cell differentiation and thus induction of tissue morphogenesis (Lim 2022). Cell engineering controllably "push a cell's button" to initiate desired morphological responses.4
In context, the induction of chondrogenesis by a coral-derived biomimetic biomaterial bioreactor when implanted solo in the dorsal musculature of the Selachian' fish the shark Carcharhinus obscurus figuratively shows how the macro- and micro-porous surface' characteristics of the intramuscularly implanted bioreactor ultimately "push a cell's button" that results in the induction of chondrogenesis (Fig. 1) by molecularly triggering invading myoblastic cells of into the macroporous spaces of heterotopically implanted bioreactors.2,3
In his lucid and clear contribution to the evolutionary development of the vertebrate skeletal tissues, Romer presents a concise assay on the "Ancient history of bone".1 Tissue induction and the developmental biology of both cartilage and bone are controlled by a vast array of genes and gene products molecularly controlling cellular and extracellular matrices synthesis, deposition and gene expression pathways.5-9
Romer in his quest to address the question "Why bone?"1 touch upon the hypothesis that bone formed as a storage of ions, particularly Ca++ and several pleiotropic proteins. These must include the structural collagenous proteins, i.e. collagens type I, IV and II, osteonectins, fibronectins together with an array of altogether different morphogenetic proteins, i.e. proteins initiators that de novo set into motion the extraordinary induction of bone formation, or, as per G Levander' classic paper in Nature: "Tissue Induction".10
The work or Romer has indicated that the induction of bone formation and thus "Why bone?" is for the development of the dermal skeletal armor.1 "It thus seems highly probable that the bony skeleton without which the evolution of the vertebrates could never have taken place, owes its origin close to half a billion years ago, to the threat of invertebrate predation on our feeble primitive ancestors" (Romer 1963)1.
Perhaps however, the grand contribution of Romer to both knowledge of the cartilaginous fishes and the emergence of the bony armor was that his classic assay presented evidence that bone has an ancestral character, and that cartilaginous fishes like sharks, skates and rays were not primitive when compared to phylogenetically ancestral sharks. 1 Indeed, Romer argues that the cartilaginous skeleton developed following a "degenerative slump from bone bearing ancestors".1 Romer further states that ancient sharks were bone-bearing fishes, later "degenerating" the ancestral bone into newly developed cartilaginous endoskeletons.1
The work of Romer grandly shows that the origins and development of the vertebrates is "the reverse of the truth". 1 The cartilage as seen in vertebrates is only an embryonic adaptation to properly growth and expand the long bones of the axial skeleton. The development of the growth plate was only possible via the development of the cartilaginous ancestral matrix that not only retained but possessed the fundamental morphological and molecular mechanisms of the cartilaginous growth plate of mammals. Of note, these were ancestrally present within the induced cartilage by the macroporous spaces of the coral-derived bioreactor intramuscularly implanted in the shark Carcharhinus obscurus (Fig. 1).2,3,11
Intriguingly, high power images of chondrogenesis as induced by the coral-derived bioreactor reveal the columnar assembly of chondroblastic cells within the chondrogenic extracellular matrix as initiated within the coral-derived macroporous spaces implanted in the dorsal musculature of Carcharinus obscurus (Fig. 1).2.11 The columnar chondroblastic assembly of the cartilaginous growth plate is the very mechanism of the longitudinal growth of the endochondral long bones in mammals, phylogenetically present within the ancestral matrix that diverged into cartilaginous Elasmobranchs' skeletons.
The uniqueness of the mammalian cartilaginous growth plate is a fundamental compartmentalized biological bioreactor that masterminds the three-dimensional growth of the mammalian axial skeleton. Osteogenesis and the induction of bone, form via endochondral ossification, i.e. via the development of the cartilage anlage. Intriguingly, high power images of chondrogenesis by coral-derived bioreactors implanted intramuscularly in Carcharhinus obscurus, reveal the columnar assembly of chondroblastic cells within the extracellular matrix as initiated by coral derived macroporous bioreactors when implanted in the dorsal musculature of the Selachian's' fishes (Fig. 1).
Ancestrally thus, the induction of chondrogenesis by a macroporous coral-derived bioreactor implanted in the dorsal musculature of the Selachian's fish Carcharhinus obscurus is a cartilaginous matrix that retains the molecular blueprints for the induction of endochondral bone formation in mammals. This developmental pathway was only possible by the development of the cartilaginous growth plate, phylogenetically however not predating the induction and development of the cartilaginous fishes or Chondrichthyes.2,11
The development of a cartilaginous skeleton, not primitive but formed from ancestral bony skeletons inherited by sharks, skates and rays has been masterminded by genetic mutations that resulted in the ablation of angiogenic mechanisms controlling the evolution and development of the cartilaginous anlage.2,3,11, Such evolutionary pathways resulted in the expression and synthesis of powerful inhibitors of angiogenesis13-15 that blocked "osteogenesis in angiogenesis".16-19
The lack of cartilage vascular invasion or chondrolysis effectively blocked the induction of bone formation, as angiogenesis is a prerequisite for osteogenesis. 19 The lack of the induction of bone formation evolutionary speciated the Chondrichthyes or Elasmobranchs as genera with highly resilient cartilaginous skeletons to swim and feed in deep waters for proper swimming and hunting, altogether optimally surviving in the depths of the oceans.2
Why bone then? In a previous Chapter of a CRC Press Volume focused on the induction of bone formation by the transforming growth factor-ß3 morphogen (TGF-ß3),20 we proposed that the evolutionary development of the growth plate was the morphological and molecular master key responsible for the longitudinal growth of the mammalian axial skeleton and for the induction of bone formation. Thus, the growth plate was the molecular and morphological bioreactor for the emergence of vertebrates and later of the Homo clade.20
The evolution of the skeleton, or rather the induction of bone formation and skeletogenesis, provided the biological tissues for the emergence of the vertebrates. As such, the skeleton acts as a "giant molecular machine" (AH Reddi personal communication 2011). Several key mutations and evolutionary adaptions resulted in the development of the pelvis for both ambulation, body erection and for fetal adaptations during hominins' speciation and the birth of man.
In a previous communication, we proposed that Nature's developmental biological and evolutionary plan was simply to provide "Bone: Formation by autoinduction", 21 skeletogenesis and body erection, pelvis adaptation to body erection and ambulation. This significantly contributed to enforcing industrious Homo-like activities by freeing the upper limbs for superior foraging, for the development of tools not limited to hunting and gathering but above all however for maternal care, physically and continuously guiding the newborn, contributing thus to the speciation of the Homo clade.3
The development of the skeleton, the induction of bone formation via the cartilaginous growth plate was Nature's master plan for the emergence of the vertebrates. The supramolecular assembly of the extracellular matrix of bone developed tissue forming substances or morphogens, first defined by Turing as "forms generating substances",22 that initiate tissue morphogenesis, the genesis of form and function.
A variety of gene and gene products were thus required to set into motion the induction of bone formation and the initiation of skeletogenesis. It is noteworthy that Nature' parsimony in controlling multiple specialized functions or pleiotropy developed several osteogenic molecular signals with minor variation in amino acid sequence' motifs within highly conserved carboxy-terminal regions. 16,23
Remarkably, gene products with ancestral sequences and amino acid motifs expressed in Drosophila melanogaster evolved for a billion years before the emergence of the vertebrates and the induction of skeletogenesis. Recombinantly generated DNA gene products of decapentaplegic and 60A genes of Drosophila melanogaster, the boneless fruit fly, initiate the induction of endochondral bone formation when reconstituted with allogeneic insoluble and inactive collagenous bone matrix and implanted in extraskeletal heterotopic sites of rodents. 24
Nature thus usurped phylogenetically ancient amino acid sequence' motifs controlling dorso-ventral patterning in Drosophila melanogaster to set the unique traits of the vertebrates, i.e. "Tissue induction"10 and "Bone: Formation by autoinduction" 21 using minor modifications of amino acid sequence' motifs ancestrally deployed in Drosophila melanogaster for unrelated functions (Ripamonti 2006; Ripamonti 2019). 25
Perhaps at the end of this sub-heading "Why bone?", reviewing the extraordinary developmental and molecular evolutionary plan that mechanistically frame the fundamental biological mechanisms of unique human biology,26 it is perhaps worth to state again that Nature's plan for "the induction of bone and osteogenesis was only to finalize the evolution of Homo sapiens on the planet earth".3
SOLUBLE MOLECULAR SIGNALS AND THE INDUCTION OF BONE FORMATION
Last Century research has shown that intact demineralized bone matrices induce endochondral bone formation in heterotopic sites of animal model (for reviews: 16,27). The critical experiments of Levander,10,28 Urist, 21 Reddi and Huggins 29 and other showed that the extracellular matrix of mineralized tissues is the repository of differentiating morphogens tightly bound to the mineralized matrix.
In his classic work, "A study of bone regeneration",28 Levander states that "In the healing process of bone the new bone may be pictured as emanating from two different sources; partly from the end of the bone fragments and partly from the connective tissue surrounding the site of fracture. In the latter case, the connective tissue is considered transformed into bony tissue by virtue of a special process - the metaplastic theory of bone formation". 28
Levander' experiments show that after heterotopic implantation of autogenous bone grafts "new bone is formed directly out of the mesenchymal tissue which surround the graft". 28 Astutely, Levander understands that differentiation of bone from the mesenchymal tissue surrounding the graft "must necessarily show that the process is influenced in some way or another by some specific agent". He further states that such specific agents emanate from the grafted tissue. 28
Levander thus hypothesizes that a "specific bone forming substance is liberated from the implanted bone tissue and it is carried by the tissue lymph to the surrounding areas where it is able to activate the mesenchymal tissue in such a way that this becomes differentiated into bone tissue - either directly or by means of the embryonic pre-existing stage of bone and cartilaginous tissue". 28
It is our opinion that the above extraordinary statement summarizes with lucid and clear morphological and molecular insights "The Bone Induction Principle" (Urist et al. 1967), proposing that the extracellular matrix of bone is a reservoir of soluble and insoluble signals that initiate the induction of bone formation. 28
As a matter of semantic perhaps Levander' statements and insights were not perceived then worthy as claims to fame possibly because Levander' studies and publications did not propose a more precise or enticing definition of this unidentified "bone forming substance". This in spite of the major insights into the induction of bone formation, particularly by alcoholic extracts, and the vision of the "bone forming substance" as a soluble signal.28 The above statements were paralleled by the statement that the morphological evaluation of the newly induced bone showed that "fully formed mesenchymal cells ultimately emanate from the endothelial cells of the capillaries".28
The above is a further challenging statement of Levander, who had the extraordinary morphological and somehow the molecular vision to understand "The Role of the Vessels in Osteogenesis" long before the classic paper of Trueta in The Journal of Bone and Joint Surgery [B].19 Trueta defined the induction of osteogenetic vessels as essential morphological and molecular components for the induction of bone formation.19 Several authors did already postulate the role of the vessels in osteogenesis and Aristotle even proposed that vessels and invading capillaries were organogenetic, constructing the frame of the body plan.25,27
Following Levander' studies (for details see 27 Urist recognized the importance of demineralized bone matrix (DBM) to induce reproducible heterotopic endochondral bone induction, 21,30 and later proposed the present terminology hypothesizing the presence of a bone morphogenetic protein complex (BMP) within the bone matrix .31
A quantum leap towards the mechanistic understanding of the phenomenon of "Tissue Induction", 10 has been the dissociative extraction and reconstitution of the bone matrix components which, when combined, trigger the bone induction cascade.31,32 The experiments of AH Reddi, then at the NIH Bone Cell Biology Section31,32 dissociatively extracted demineralized bone matrix in chaotropic agents such as 4M guanidinium hydrochloride or 6M urea resolving an insoluble and inactive collagenous matrix signal and solubilized extracted proteins, or soluble signals.33
Purified extracted proteins by gel filtration chromatography reconstituted with the allogeneic insoluble and inactive collagenous bone matrix restored the biological activity of the extracted proteins, initiating the induction of bone formation in the rodent subcutaneous assay.31 Soluble signals, i.e. osteogenic proteins, need to be reconstituted with allogeneic inactive collagenous bone matrix, 32 since xenogeneic collagenous matrices as carriers block the bone induction cascade.32
The realization that the chaotropically extracted extracellular matrix of bone was a reservoir of structural and morphogenetic proteins set the scientific and biotech industry' race for the isolation and purification to homogeneity of the elusive yet to be isolated and characterized BMP complex postulated by Urist and Strates in 1971 as bone morphogenetic protein.34
A further incisive step ahead was again the work of Urist and co-workers published in PNAS describing the purification of bovine BMP by hydroxyapatite chromatography.35 This experiment reported the adsorption or "absorption" of the BMP complex onto hydroxyapatite chromatography gels. The research experiment reported that a broad band of osteogenic fractions with BMP-like activity would adsorb onto hydroxyapatite chromatography gels. 35 Eluted fractions of 18.5 kDa induced large deposits of bone and newly formed ossicles in heterotopic sites of rodents. 35
Using chaotropically extracted bovine bone matrices, Reddi' team at the NIH Bone Cell Biology Section purified osteogenin, an osteogenic protein with biological activity in the rodent subcutaneous assay. 36 Purification was by sequential hydroxyapatite adsorption, heparin-Sepharose affinity and S-200 Sephacryl gel filtration chromatography, reporting a molecular weight of 22 kDa with osteoinductive activity in heterotopic subcutaneous sites of rodents. 36
Incisive work aided by continuous collaboration and contacts with leading scientists in the field allowed Genetic Institute, Boston US, to purify to homogeneity naturally derived bovine morphogenetic proteins (Wang et al. 1988). Purification steps included hydroxyapatite adsorption chromatography, affinity chromatography on heparine-Sepahrose gels, and Superose 6 and 12 columns connected in series to optimize gel filtration. Biologically active proteins were of approximately 30 kDa on SDS-PAGE.37
Genetic Institute' scientists decided to re-use the original term bone morphogenetic protein proposed by Urist and Strates in 197134 to define the newly purified and cloned proteins thus to ride all the biological in vitro and in vivo scientific background as formidably established by the Bone Research Laboratory at the University of California Los Angeles. 37,38
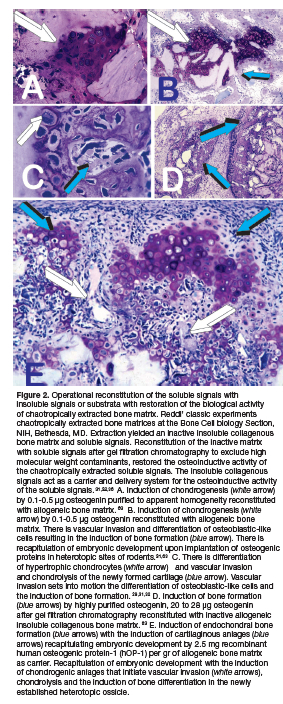
Protein sequences were defined, obtaining amino acid motifs which were used to clone several human recombinant bone morphogenetic proteins (BMPs). Science reported the experiments as "Novel Regulators of Bone Formation: Molecular clones and Activities".38 The contribution to Science primarily identified not one (Fig. 2) but several proteins with osteoinductive activity in the rodent bioassay, and that the newly isolated and cloned proteins were new members of the TGF-ßs supergene family. 38,39
MORPHOGENS, OR SOLUBLE MOLECULAR SIGNALS, INITIATE PERIODONTAL TISSUE INDUCTION
Purification to homogeneity of naturally derived BMPs, molecular cloning and expression of the recombinant human proteins 16,27 (for reviews) did appear, then, to resolve the "Reality of a nebulous enigmatic myth". 40 Tissue regeneration in postnatal life recapitulates events that occur in the normal course of embryonic development. 10,28,29 A highly conserved family of proteins, the transforming growth factor-ß (TGF-β) supergene family, equally regulates both embryonic development and postnatal tissue induction. 16,17,18,23,29,41,42,43,44,45
The pleiotropism of the TGF-ß supergene family underlines the findings that the three mammalian TGF-β isoforms initiate endochondral bone induction in the non-human primate Papio ursinus. 46-49
Pre-clinical studies in the Chacma baboon Papio ursinus showed the induction of bone by the bone morphogenetic proteins (BMPs), pleiotropic members of the TGF-ß supergene family.16,45,48,50 Mammalian naturally derived BMPs and recombinant human BMPs (hBMPs) induce de novo bone formation (Fig. 2). Proteins act as soluble signals for tissue morphogenesis, sculpting the multi-cellular mineralized structures of the periodontal tissues with functionally oriented periodontal ligament fibers inserting into newly formed cementum (Fig. 3).16,25
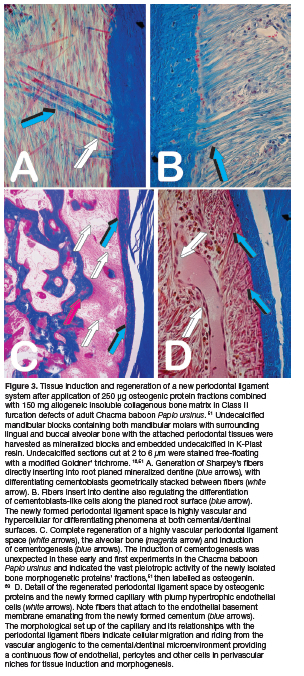
BMPs induce the complex tissue morphologies of the periodontal tissues in the non-human primate Papio ursinus in Class II mandibular furcation defects treated with naturally - derived and recombinantly produced BMPs. 51,52,53,54 The presence of multiple forms of BMPs has a therapeutic significance and the choice of a suitable factor is a formidable challenge to the practicing periodontologist and skeletal reconstructionist-. 55,56
Tissue morphogenesis induced by hOP-1 and hBMP-2 is qualitatively different when the morphogens are applied singly. This indicates that the structure/activity profile amongst BMPs is controlling pleiotropic tissue induction and morphogenesis (Fig. 4). 25,56,57,59, Furcation defects of Papio ursinus with root surfaces long-term exposed to periodontal disease implanted with doses of gamma-irradiated hOP-1 resulted in complete regeneration of the furcation defects with prominent induction of cementogenesis with Sharpey's fibers embedded within the newly formed cementum.58,59
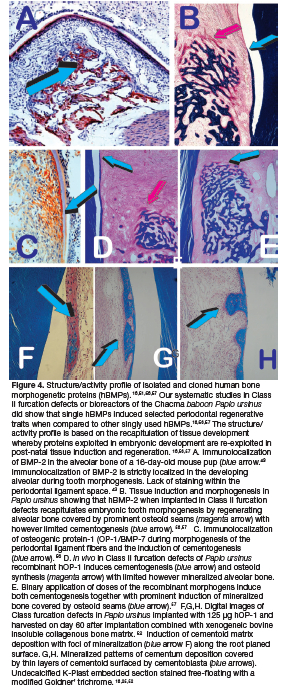
Short-term studies delivering 125 hOP-1 combined with xenogeneic gamma-irradiate bovine bone matrix reported the induction of cementogenesis by day 60 after implantation along the exposed root surfaces of Class II furcation bioreactors (Fig. 4f). 52 Induction of cementogenesis was evident 6 months post-implementation in Class II furcation defects of Papio ursinus (Figs. 2.4g,h).58
A novel regenerative approach but in primates only is the induction of periodontal tissue regeneration with substantial cementogenesis by doses of the recombinant human transforming growth factor-ß3 (hTGF-ß3). 25,56,60 In the nonhuman primate Papio ursinus periodontal tissue induction and regeneration develops as a mosaic structure in which the osteogenic proteins of the TGF-β superfamily singly, synergistically and synchronously initiate and maintain tissue induction and morphogenesis.43,56,59
An alternative bone induction strategy and regenerative approach is to induce in heterotopic sites newly formed ossicles by recombinant hTGF-ß3 later transplanted as morcellated autogenous bone grafts into Class II furcation defects of Papio ursinus 59,61 and with hTGF-ß3 in Matrigel®Matrix with rectus abdominis responding cells.62
Research experiments analyzed both the morphological and gene expression studies of periodontal tissue induction and morphogenesis of selected osteogenic proteins of the TGF-β supergene family. 56,60 Results showed that hOP-1 and hBMP-2 singly or in binary application show pronounced morphological regenerative differences (Fig. 2.4). Recombinant proteins, singly or in binary applications where implanted reconstituted with insoluble collagenous matrices into Class II furcation defects of Papio ursinus. 25,57,59 The results highlighted the site tissue specificity and the structure activity profile of each recombinant hBMP when applied singly to root planed surfaces recapitulating embryonic development of the expressed and secreted proteins (Figs. 4a,c; Figs. 4b,d,e). 25,43,52,56
We defined the capacity of mammalian BMPs to initiate a programmed cellular cascade resulting in the induction of bone 63 as well as cementogenesis 25 as "a functionally conserved process utilized in embryonic development, recapitulated in postfetal osteogenesis, and can be exploited for the therapeutic initiation of bone formation" 63 as well as cementogenesis.25
Previously identified challenges in periodontal tissue regeneration 55 are still unsolved; this despite several research work on cellular populations allegedly initiating periodontal tissue induction. 64,65
The biological significance of redundancy is a still unresolved challenge. 55 Experimentation in non-human primates has shown that the presence of multiple forms of BMPs has a therapeutic significance. 56,59 Limited research addressed this challenge since "the choice of a suitable morphogen is still a formidable challenge to the practicing periodontologist" .55 We have proposed that critical research experimentation would have been to study optimal combinations and developing a structure-activity profile amongst the members of the BMPs family. 51,55,59
Our Unit has been amongst the first to propose heterotopic and orthotopic regenerative studies combining homologous but molecularly different morphogenetic proteins. Binary applications were also applied in periodontal regenerative studies. 57,59,66 Our first heterotopic combination study yielded unprecedented results showing a synergistic interaction between recombinant human osteogenic protein-1 (hOP-1, also known as hBMP-7) and relatively low doses of hTGF-ß1 (Fig. 5). 46,67
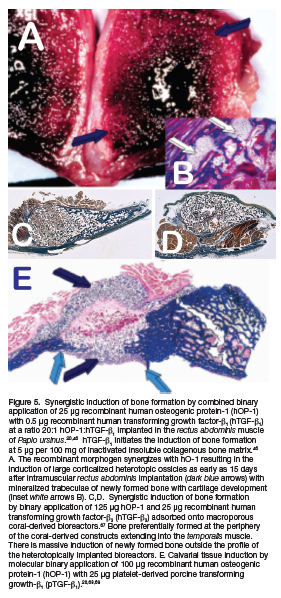
We later provided mechanistic molecular data supporting the profound synergistic interactions between platelets-derived porcine transforming growth factor-ß1 (pTGF-ß1) and recombinant hOP-1. 68 Type IV collagen mRNA was highly expressed in synergistic tissues providing extracellular basement membrane' components for vascular invasion and capillary sprouting within the newly formed synergistic ossicles in the rectus abdominis muscle of Papio ursinus. 68,69
Reconstitution of allogeneic insoluble collagenous bone matrix with 100 hOP-1 combined with 15 pTGF-ß1 and implanted in calvarial defects of Papio ursinus resulted in a substantial synergistic interaction of bone formation on day 30 displacing the temporalis muscle (Fig. 5e). 67,68,69
Reconstituted coral-derived macroporous bioreactors with 125 hOP-1, 125 hTGF-ß3 and binary applications of 125 hOP-1 and 25 hTGF-ß3 in the ratio of 5:1 hOP-1:hTGF-ß3 were implanted in heterotopic intramuscular sites of the rectus abdominis muscle of Papio ursinus. Results showed prominent and substantial induction of bone formation extending far beyond the profile of the implanted super activated bioreactors (Figs. 5c,d). 67 Of interest, qRT-PCR showed prominent induction of TGF-ß3 mRNA with relatively low expression values of OP-1 mRNA.67,69
The morphological hallmark of the synergistic induction of bone formation is the rapid differentiation of large osteoid seams enveloping haematopoietic bone marrow that forms by day 15 in newly generated ossicles in the rectus abdominis muscle of Papio ursinus (Figs. 5a,b).46,67
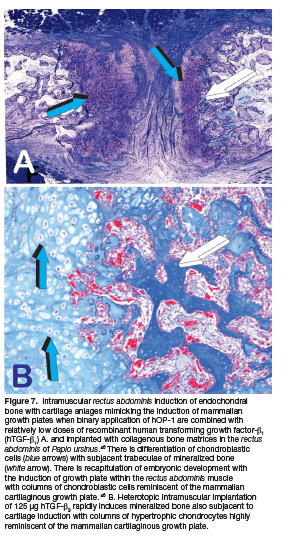
We also reported that synergistic binary application of hOP-1- and hTGF-ß1 in the ratio 20:1 respectively, initiate the heterotopic induction of rudimentary embryonic growth plates (Fig. 7a). This has indicated that the "memory" of developmental events in embryo is re-deployed postnatally by morphogen combinations (Fig. 7a). 46,69 Of interest, tissue induction and morphogenesis by TGF-ß3 applied singly in heterotopic rectus abdominis intramuscular sites also morphogenizes columnar chondrocytes as seen in the mammalian growth plate (Fig. 7b).
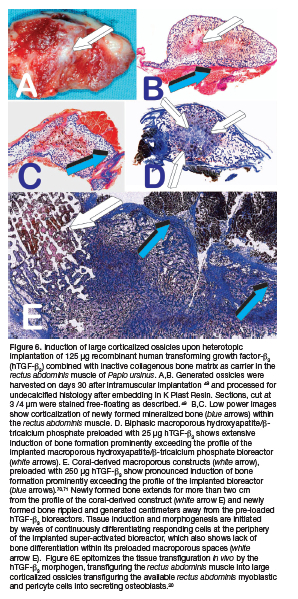
It is noteworthy that single applications of 125 or 250 hTGF-ß3 result in the rapid induction of heterotopic bone formation (Fig. 6). The induction of bone formation is superior to binary applications of recombinant hTGF-ß1 or hTGF-ß3 with hOP-1. 49,67,69 Mechanistically, the initiation of bone by the recombinant hTGF-ß3 invocates the rapid induction and expansion of the transformed mesenchymal tissue into large corticalized heterotopic ossicles with pronounced osteoblast-like cell differentiation at the periphery of the implanted reconstituted specimens with "tissue transfiguration in vivo" (Fig. 6e). 20,49
The induction of bone forms beyond the geometric space of reconstituted carrier matrix, prominently expanding outside the profile of the macroporous delivery system, being either macroporous biphasic hydroxyapatite/ß-tricalcium phosphate (HA/ß-TCP) (Fig. 6d) or coral-derived macroporous bioreactors preloaded with 250 (hTGF-ß3 (Fig. 6g).
The image shown in Figure 6e shows significant and prominent osteogenesis predominantly surrounding the coral-derived macroporous bioreactor super activated by 250 hTGF-ß3 (Fig. 6e). 20,48,70,71 Ofinterest, a tenfold less dose of hTGF-ß3, i.e. 25 pg, initiates prominent induction of bone formation extending outside the profile of the heterotopically implanted super activated HA/ß-TCP bioreactor (Figs. 6d white arrows. 70,71
Molecularly, the rapid induction of bone formation by binary applications of hOP-1 and hTGF-ß3 or by hTGF-ß3 applied singly, resides in the up-regulation of selected genes involved in tissue induction and morphogenesis, i.e. Osteocalcin, RUNX-2, OP-1, TGF-ß1 and TGF-ß3 with however notably lack of TGF-ß2 up-regulation. 69 Of note, the induction of bone formation by the hTGF-ß3 isoform implanted singly is greater than ossicles generated by binary synergistic applications of hOP-1 with relatively low doses of either hTGF-ß1 or hTGF-ß3 (Fig. 2.6a). 46,69 Relatively high doses of the hTGF-ß3 morphogen (125 hTGF-ß3) initiate a developmental cascade of molecular and cellular events primarily characterized by the expression of multiple profiled bone morphogenetic proteins. 69 Together with significant chemotaxis, chemokinesis and cell migration of responding cells at the periphery of the hTGF-ß3-pre-treated bioreactors, the expressed and secreted BMPs induce rapid and extensive bone formation greater than the synergistic induction of bone formation. 69
The reported data on the significant and pleiotropic biological activities of the hTGF-ß3 morphogen indicate that the TGF-ß3 gene masterminds' critical developmental events beyond bone and cartilage morphogenesis, ancestrally regulating skeletogenesis and the emergence of the craniofacial dentate masticatory apparatus, including the differentiation and initiation of cementogenesis. 25,60
Synergistic molecular combinations were thus tested in Class II furcation defects of the Chacma baboon Papio ursinus (Fig. 2.4). 25,57,66 Our study that first attempted to address the structure-activity profile amongst BMPs family members did show that tissue morphogenesis induced by hOP-1 and hBMP-2 is qualitatively different when the morphogens are applied singly, hOP-1 inducing substantial cementogenesis (Figs. 4d,f,g,h). hBMP-2 treated defects showed limited induction of cementogenesis but a temporal enhancement of alveolar bone regeneration and remodeling (Fig. 4b). Although statistically not significant, the extent of cementogenesis by binary application showed pronounced induction of cementogenesis when compared to hBMP-2 treated specimens (Fig. 4e).
The demonstration of therapeutic mosaicism in periodontal tissue regeneration, as previously highlighted by immunolocalization studies during murine craniofacial and periodontal embryonic development, 43 will require extensive testing of ratios and doses of recombinant morphogen' combinations for optimal tissue engineering in clinical contexts.57
Synergistic molecular combinations of hOP-1 and hTGF-ß1 showed pronounced angiogenesis in the chick chorio-allantoic membrane (CAM) 69 when morphogens were applied at 20:1 ratio of hOP-1 and hTGF-ß1, respectively. Remarkably, the study showed that hOP-1 is per se angiogenic at doses of 100 and 1000 pg, comparable to the angiogenic activity of recombinant human basic fibroblast growth factor (hbFGF). 72
Further studies showed that binary applications of hOP-1 and hTGF-ß3 in Matrigel®Matrix implanted in Class II furcation defects of Papio ursinus induced substantial periodontal tissue induction and regeneration. 66 The anatomy of the furcation defects however tempered the full morphogenetic drive of the synergistic binary applications that morphogenized large ossicles expanding toward the muco-periosteal flaps with the remarkable induction of cementogenesis along the planed root surfaces.66
A review of the literature shows the lack of biological studies aimed to define the structure-activity profiles of recombinant hBMPs; studies reporting synergistic interactions amongst members of the morphogenetic protein family are also lacking. 25 We have stated that the "biological acceptance of the inductive activity of a single recombinant human morphogen about the natural milieu and equilibrium of a pleiotropic bone matrix endowed with several naturally derived proteins clustered within the extracellular matrix of bone has been the fundamental biological error of biotech companies developing recombinant BMPs'. 25,56
Clinician scientists were also far too eager to accept unconditionally the reported powerful biological activity of either hBMP-2 or hOP-1 and to test in various clinical settings single recombinant hBMPs, and later recalcitrant to even admit the failure of hBMPs' translation in clinical contexts. 70,71
Biotechnology companies at the forefront of recombinant human inductive proteins marketed selected recombined BMPs as single proteins. Recombinant human proteins were packaged singly recombined with patented delivery systems, hBMP-2 by Genetic Institute, USA, and hOP-1 (also known as hBMP-7) by Stryker Biotech, USA.
Finally, and again despite substantial research experimentation on the formulation of delivery systems for recombinant human morphogens including hBMPs and hTGF-ßs, the ideal carrier matrix for periodontal tissue induction is still not available. The use of hBMPs pre-combined with doses of allogeneic and/or xenogeneic insoluble collagenous bone matrix (ICBM) was used to deliver recombinant hOP-1 in Class II furcation defects of the Chacma baboon Papio ursinus (Fig. 4). 52,57,58 Binary synergistic combinations were also tested (Fig. 4e). 57 The use of allogeneic ICBM proved to deliver the biological activity of doses of naturally derived osteogenic BMPs fractions purified greater than 50.000-fold from crude bovine bone matrix extracts (Fig. 3).51 Xenogeneic bovine ICBM was used to deliver the biological activity of gamma-irradiated hOP-1 in short (Figs. 4f,g,h) 52 and long-term experiments in the Chacma baboon Papio ursinus. 58
The use of Matrigel®Matrix as delivery system for hTGF-ß3 doses has proven to be optimal for periodontal tissue induction when lyophilized doses of hTGF-ß3 in Matrigel®Matrix were implanted in Class II furcation defects of Papio ursinus (Fig, 8).60 Research on osteogenic carriers needs to design therapeutic strategies based on cell biology of matrix-cell interactions for optimal outcome in the periodontal patient.55
The use of Matrigel®Matrix originally developed as a matrix substratum for in vitro research experiments 73 was later used to deliver recombinant hOP-1 in the subcutaneous bioassay in rodents.74 Matrigel is a soluble extract of the Engelbreth-Holm-Swarm tumor which gels at room temperature to form a reconstituted basement membrane gel. 73 Matrigel promotes differentiation of a variety of cells. 73,75 Our studies evaluated the induction of organoids of transitional epithelial cells harvested from baboon fetal bladders and grown on Matrigel®Matrix (Fig. 9).74
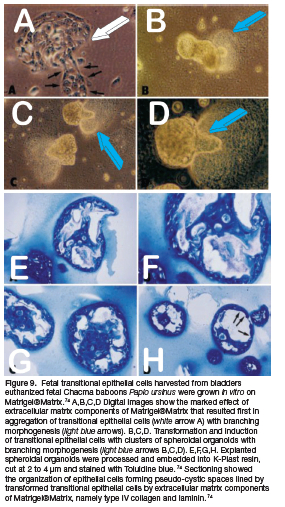
Matrigel®Matrix contains, amongst other extracellular matrix components, laminin and type IV collagen, the essential constituents of capillary basement membranes. Both bind to BMPs 76,77 and TGF-ß. 78 Importantly, not only osteogenic but also angiogenic morphogens are stored within basement membranes deposited into the subendothelial extracellular matrix.79 The multiple binding and storage of morphogenetic and angiogenetic morphogens within the subendothelial basement membrane make the reconstituted basement membrane gel Matrigel®Matrix an ideal carrier based on cell biology of matrix cell interaction (Figs. 8; 9). 60,74
2.4. BIOREACTORS FOR DE NOVO INITIATION OF CEMENTUM AND ALVEOLAR BONE ORGANOIDS
The binding of osteogenin, a bone morphogenetic protein purified to homogeneity from bovine and baboon bone matrices,64 to type IV collagen, laminin, and transforming growth factor-ß1 76,77,78 suggested to combine highly purified osteogenic fractions extracted from baboon bone matrices and recombinant human osteogenic protein-1 (hOP-1, also known as hBMP-7) with Matrigel®Matrix kept fluid on ice, to test the biological activity of purified osteogenic proteins when combined with Matrigel®Matrix implanted in heterotopic extraskeletal sites of the rodent bioassay. 74
Recent time-study experiments combined morphological analyses to a time point molecular study of Class II furcation defects in Papio ursinus super activated by TGF-ß3 in Matrigel®Matrix. 60 The combined morphological and molecular analyses have indicated that relatively low doses of hTGF-ß3 (75 hTGF-ß3 in 600 Matrigel®Matrix) set into motion the in vivo development of multiple tissues and multicellular organoids within the implanted furcation bioreactors (Fig. 2.8).60
Ultimately, the surgical preparation of periodontal furcation defects in animal models including man is a complex surgical wound or bioreactor that after the implantation of morphogens either singly or in combinations initiates regenerative phenomena by gene expression pathways.25,60
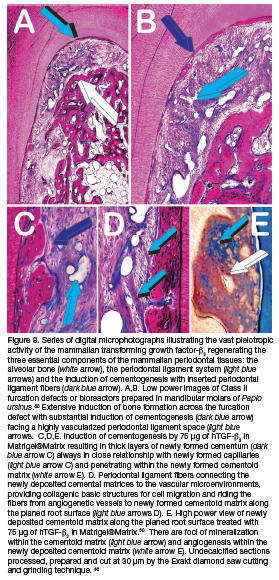
Various tissues and cells with different embryological origins as well as the induction of angiogenesis from the severed periodontal ligament and alveolar bone spaces control the regenerative pathways of the newly established bioreactors. The most critical part of the bioreactor is a completely avascular rigid and mineralized dentin matrix layered with or without the avascular mineralized root cementum (Figs. 8a,b). The bioreactor of the furcation defect is thus a surgical micro-environment that may or may not promote cementogenesis along its avascular and mineralized root planed dentinal surfaces.
We did recently discuss whether differentiation of cementoblasts along the root planed dentinal surfaces occurs either at a considerable coronal distance from the apically positioned severed cementum after root planing, or from the apically positioned notch in the root surface, a niche of migrating responding cells we have defined as "the only true regenerative microenvironment of the complex morphologies of the periodontal tissues".60
Our series of histological analyses of undecalcified sections prepared by Reichert Jung sledge microtomes with tungsten carbide blades or by the Exakt diamond saw cutting and grinding technology shows that there is attachment, spreading and differentiation of cementoblasts coronally along the root surface (Figs. 8a,b light blue arrows). The differentiation of cementoblastic cells together with insertion of Sharpey's' fibers into the root surface is uniformly distributed along the length of the regenerating periodontal ligament space, extending to the furca of the defect (Figs. 2.8a,b light blue arrows).25,56,60
Critical contributions described the mechanical regulation of cell function by geometrically modulated substrata. The available data are critical to mechanistically understand the attachment, differentiation and spreading of cementoblastic cells on rigid substrata. 25
Incisive research by Discher' laboratories has shown in vitro the role of micro pillars to affect subcellular nuclear geometry that further regulates stem cell differentiation and the induction of tissue patterning.80 "Stem cells feel the difference" 80 when cultured on different substrata' consistencies, i.e. between soft and hard substrata. 81,82 Molecular studies in Cell mechanistically reveals how "Matrix elasticity directs stem cell lineage specification". 83 Stem cells commit to specific phenotypes to tissue level elasticity. 80-83
The above work on "stem cells feeling the difference" 80 is summarized by far reaching molecular and differentiating mechanistic insights, i.e. "soft matrices that mimic brain are neurogenic"; in contrast, "comparatively rigid matrices that mimic collagenous bone prove osteogenic". 83 The above statements are "perhaps the most molecularly and intellectual fascinating aspect of biomimetism, biomimetic matrices and the induction of bone formation".84
In context of periodontal regeneration, the above data propose that hard mineralized and avascular matrix of root planed dentine proves to be cementogenic when in contact with mesenchymal stem cells either migrating from cellular niches within the dentinal notch or directly differentiating along the root surfaces, as recently proposed. 60 The dentine/cementum unit retains thus characteristics for the differentiation of selected phenotypes, also initiated by the exogenous application of osteogenic soluble molecular signals. 25,59
Within the implanted furcation defects, de novo generated organoids form by multiple tissue induction of different tissues organized in intra-furcal organoids.25,60
Toward the root planed surfaces, there is the induction of substantial cementogenesis. Cementum is deposited firstly as cementoid matrix yet to be mineralized. 52,56,60 Cementoid tissue forms and extends into the periodontal ligament space with trabeculations underscoring the powerful role of the TGF-ß3 gene controlling cementogenesis in primates (Fig. 8).26,56,60
Matrigel®Matrix is an ideal combined soluble and insoluble signal that control the morphogenesis of organoids in vitro of transitional bladder cells when grown to confluence on Matrigel®Matrix substrata (Figs. 9a,b,c,d). 74 Newly generated organoids embedded in in K-Plast resin were cut at 2 to 3 using carbide tungsten knives mounted on Reichert Polycut sledge microtomes and stained with toluidine blue in 30% ethanol (Figs. 9e,f,g,h). 74
Histological analyses show that organoids are formed by transitional fetal epithelial cells lining pseudo-cystic spaces organized by trabeculation of transitional epithelial cells generating the spheroidal organoids (Figs. 9e,f,g,h).
Analyses of undecalcified histological sections cut at 30 on the Exakt diamond saw, grinding and polishing system 60 suggested that trabeculations of newly formed cementoid matrices surrounding foci of mineralized cementum are recapitulating in extant primate species the induction of substantial cementogenesis as seen on undecalcified sections of dentate specimens of extinct mosasaur Hallisaurus sternbergi 168-165 Ma (Fig. 10). 85 Undecalcified sections of periodontal tissues of extinct mosasaur Hallisaurus sternbergi 84 show trabeculations of cementum with the possible presence of vascular canals and capillaries (Fig. 10g). 25,85

Of interest, in extant primates Papio ursinus, newly formed cementoid and later mineralized cemental matrices are in a very intimate relationship with sprouting capillaries within the newly formed periodontal ligament space (Figs. 8; 10). Importantly, the newly formed cementum is vascularized, showing the presence of sprouting capillaries within the cemental matrix (Figs. 8; 10). 25,56,60
The exquisite relationship between sprouting capillaries morphologically and thus molecularly touching the newly synthesized cemental matrix covered by cementoblasts indicates that the newly deposited cementoid synthesizes cemental extracellular matrix proteins that control angiogenesis within the periodontal ligament space (Fig. 10). 25,60 The role of cementum in the homeostasis of the periodontal ligament space is supported by the isolation of cemental proteins from cemental extracellular matrix and by the cloning of a new cemental protein, cementogenin. Cementogenin is secreted by cementoblasts and has a molecular weight of 18.5 kDa.86
The induction of a three-dimensional in vivo culture by combining the morphogenetic soluble signal of the recombinant hTGF-ß3 with the insoluble signals of the Matrigel®Matrix, collagen type IV and laminin with binding affinity to the TGF-β type 1, 78 morphogenizes the induction of newly formed cementum with capillary invasion within the yet to be mineralized cementoid matrix (Fig. 8).
Newly formed intra-furcal organoids induced by doses of hTGF-ß3 in Matrigel®Matrix show a periodontal ligament space supported by significant angiogenesis often in close contact with the newly formed cementoid matrix, together with the induction of a very vascularized alveolar bone (Figs. 8; 10). 25,56,60
Of interest, the time study of the induction of periodontal tissue regeneration by the recombinant hTGF-ß3 in Matrigel®Matrix was morphologically and molecularly compared to heterotopic organoids generated by combining 250 doses of hTGF-ß3 to coral-derived bioreactors (Fig. 11). 60
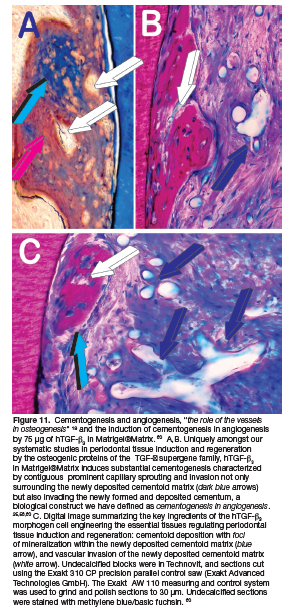
Super activated bioreactors with 250 hTGF-ß3 were implanted in heterotopic sites of the rectus abdominis muscle of the same animals implanted in Class II furcation defects with 75 doses of hTGF-ß3 in Matrigel®Matrix. 60 Heterotopic implantations of coral-derived macroporous bioreactors with or without 250 f hTGF-ß3 were implanted in the rectus abdominis muscle at the time of the periodontal surgical implantation, providing thus periodontal and heterotopic treated specimens harvested on day 60 for morphological and molecular analyses (Fig. 11). 60
hTGF-ß3 super activated or untreated coral-derived macroporous bioreactors were intramuscularly implanted as positive controls to correlate the induction of bone formation in treated periodontal sites with de novo induction of bone in the rectus abdominis intramuscular sites. 60 These positive controls were additional to several treated and untreated coral-derived macroporous bioreactors implanted in the rectus abdominis muscle. 87,88,89
sculpture of precisely organized multicellular structures of the bone bone-marrow organ. Of interest, the induction of bone can be initiated by several matrices of mammalian tissues including but not limited to demineralized bone matrices, dentin matrices, uroepithelium and a variety of calcium phosphate-based biomatrices either coral-derived biomimetic bioreactors or sintered crystalline hydroxyapatite macroporous constructs.3
Though the induction of bone is initiated by a variety of matrices as listed above, mechanistically the induction of bone is set into motion by the expression, synthesis and secretion of the bone morphogenetic proteins' genes products, ultimately the initiators of "Bone: Formation by autoinduction". 21
The induction of bone formation by the hTGF-ß3 is a point in context. 86,87,88 The induction of bone formation initiates by expression of profiled bone morphogenetic proteins including BMP-2 with subsequent induction of bone formation by the secreted BMPs gene products upon heterotopic implantation of hTGF-ß3. hTGF-ß3-treated bioreactors set into motion the expression of a variety of BMPs and TGF-ß genes at different time points temporally and spatially regulating the induction of bone formation via Noggin expression. 89
The classic view of a morphogen is that morphogenetic gradients specify gene expression in a distinct spatial order (Research article summary, 90 Tissue Morphogenesis). 90,91 The work of Yang et al. reported in Science presents an alternative pathway to tissue morphogenesis suggesting that morphogens beside modulation of individual cells induce their ultimate functional effects that enable the promotion of distinct supracellular phases that are capable of morphological transformation and organogenesis. 91
Our research data on both the induction of large mineralized corticalized heterotopic ossicles and prominent cementogenesis together with the induction of alveolar bone regeneration in the non-human primate Papio ursinus have indicated that doses of recombinant hTGF-ß3 induce distinct supracellular phases that together with morphological transformation and organogenesis results in the generation of intramuscular mineralized bone organoids with prominent osteoid seams and bone marrow cavities. The generation of transformed periodontal bioreactors into organogenesis of alveolar bone attached to a highly vascularized periodontal ligament system is patterned by collagenic fibers attaching into substantial cementogenesis with capillary sprouting and angioblastic invasion. This results in cementogenesis in angiogenesis with de novo vascularized cementoid formation. 25,56,60
Physiological expression of BMPs genes and gene products upon implantation of hTGF-ß3 may escape the antagonistic expression of Noggin, whereas direct implantation of large doses of hBMPs sets into motion the expression of Noggin tightly controlling the bone induction cascade in humans, as shown by limited effectiveness of hBMPs in clinical contexts.92,93
In his classic Editorial Comment "The reality of a nebulous enigmatic myth" 40 Marshall Urist states that pre-clinical and clinical research on the bone induction principle 30 "are bound to dispel the myth and appreciate the reality of bone induction for the benefit of patients with crippling diseases of the bone and joints". Fifty-seven years later the Bone Research Laboratory not in Los Angeles but in Johannesburg still strongly perceive "The reality of a nebulous enigmatic myth" when reading that several tens of milligrams of recombinant human BMPs are needed to induce an uninspiring bone volume in human patients.
The promise of therapeutic osteoinduction has been recognized during last Century research after pre-clinical and clinical studies. Human bone regeneration and human bone induction have proven to be an elusive target when compared to extraordinary results obtained in pre-clinical studies including non-human primate species. 16,17,92,93,94
The induction of bone formation has dramatically shown that regenerative medicine in clinical context is on a different scale altogether when compared to animal models that may not adequately translate and reproduce morphogen-related therapeutic responses in Homo sapiens. Translation in clinical context of "Tissue Induction" 10 of "Bone: Formation by autoinduction" 21 has however failed, 87,91,92,93 and the promise of human bone induction remains a promise.
As a concluding comment perhaps, it is worth ending verbatim with a statement of a rather controversial manuscript that stated that "the limited morphogens' activity in human patients when compared to different pre-clinical models including the non-human primate Papio ursinus may not indicate the failure of the bone induction principle30 in humans but simply the mere fact that both TGFF-ßs and BMPs are developmentally and biologically not Nature proposal for regeneration of skeletal defects in human patients".92
The above work has highlighted a biological problem rather than a biotechnology problem, i.e. recombinant human morphogens, recombination techniques, doses of recombinant proteins, delivery systems, age of human patients and the like. Both recombinant hBMPs and hTGF-ß3 proteins do indue bone in heterotopic sites of animal models, and the induction of bone formation recapitulates embryonic development. As stated, "In evolutionary molecular biology contexts however, the pleiotropic activity of both proteins' family and the induction of bone formation in heterotopic sites are developmental, and thus not suitable to induce bone when recombinant morphogens are singly implanted in orthotopic skeletal defects, the latter lacking the developmental biological platform". 92
To end, we have thoroughly discussed the biological significant of the heterotopic bioassays that since the last two Centuries have stated to cellular and molecular biologists, and tissue reconstructionists alike, that if a protein and/or any extracellular matrix or matrices initiate the induction of bone in extraskeletal sites, where there is no bone, such protein and/or matrix is per se osteoinductive and as such, it can be used to translate the "bone induction principle" 30 into human skeletal defects. The proteins induce bone where there is no bone via developmental phases of embryonic development, and as such, however, fail the induction of bone formation when applied to human skeletal defects.92
The developments and use of the heterotopic bioassays to test unequivocally the remarkable prerogative of certain proteins and/or matrices to initiate the biological landscape of the induction of bone formation has been essential for the purification, isolation and cloning of several proteins bestowed with the unique capacity or prerogative of initiating the induction of bone formation where there is no bone. It has however sidetracked the clinical translation of the "bone induction principle"30 since it was based on a pleiotropic developmental function without however the capacity of clinical translation into orthotopic bony sites, biologically lacking developmental phenomena.
ACKNOWLEDMENTS
The continuous and focused research experimentation on the induction of bone formation spanned for 30 years, from March 1994 to the end of March 2024 when upon a debatable yet not-negotiable request by the Deanery of the Faculty of Health Sciences, the Bone Research Laboratory ceased to exist, after more than 30 years of important critical research findings in non-human and human primates.
The author of this manuscript still wishes to thank the University of the Witwatersrand, Johannesburg for running the research laboratories from the late eighties at the Dental Research Institute, and from March 1994 at the then inaugurated the Bone Research laboratory, until March this year 2024 and now to the refurbished laboratories of the School of Clinical Medicine, Internal Medicine, jointly with the Laboratories of Molecular and Cellular Biology headed by Raquel Duarte and her team. Together with the Bone Research Laboratory, the molecular biology team resolved molecularly the spontaneous induction of bone formation by macroporous calcium phosphate-based bioreactors, the apparent redundancy of the induction of bone formation by the mammalian transforming growth factor-β isoforms, and the synergistic induction of bone formation, partially touched upon by this manuscript. This contribution to periodontal "Tissue Induction" could not have been possible without the dedication, discipline and expertise of Laura Roden (née Yeates who excelled in purification of naturally derived bone morphogenetic proteins' fractions from baboon bone matrices), Barbara van den Heever and Ruqayya Parak who excelled in cutting undecalcified sections using Reichert' sledge heavy duty microtomes mounted with carbide-tungsten knives and the Exakt diamond saw cutting and grinding technique. Digital images of their sections are now spread in more than 250 publications and in 3 CRC Press Volumes on the induction of bone formation in non-human primates.
A final grateful thank to Alberto and his team of managers at the Superspar at the Islands Broederstroom for continuous support for IT connection and printing of several manuscripts during the preparation of this review.
REFERENCES
1. Romer AS. The "Ancient History" of bone. Ann NY Acad Sci 1963; 109: 168-176. [ Links ]
2. Ripamonti U, Roden L, van den Heever B. Sharks, shark cartilages and shark teeth: A collaborative Africa-USA study to attempt to induce "Bone: formation by autoinduction" in cartilaginous fishes. South Afr Dent J. 2018; 73: 11-21. [ Links ]
3. Ripamonti U. The Induction of Bone Formation and the Osteogenic Proteins of the Transforming Growth Factor-β Supergene family. Pleiotropism and Redundancy. In: U. Ripamonti (ed.) The Geometric Induction of Bone Formation CRC Press, Taylor & Francis Group. Boca Raton FL, USA, 2021, Chapter 3, 51-68. [ Links ]
4. Lim AW. The emerging era of cell engineering: Harnessing the modularity of cells to program complex biological functions. Science 2022, 378: 848-852. [ Links ]
5. Massagué J. The transforming growth factor-beta family. Annu Rev Cell Biol. 1990:6:597-641.doi: 10.1146/annurev.cb.06.110190.003121. [ Links ]
6. Kingsley DM. The TGF-beta superfamily: new members, new receptors, and new genetic tests of function in different organisms Genes Dev. 1994 8(2):133-46. doi: 10.1101/gad.8.2.133. [ Links ]
7. Feng XH, Derynck R. Specificity and versatility in TGF-beta signaling through Smads. Ann Rev Cell Develop Biology 2005; 21: 659-93. http://dx.doi.org/10.1146/annurev.cellbio.21.022404.142018 [ Links ]
8. Pakyari M, Farrokhi A, Maharlooei MK, Ghahary A. Critical Role of Transforming Growth Factor Beta in Different Phases of Wound Healing. Advances in Wound Care Volume 2, 5, 2013 © 2013, Mary Ann Liebert, Inc. https://doi.org/10.1089/wound.2012.0406 [ Links ]
9. Richardson L, Wilcockson SG, Guglielmi L. Hill C. Context-dependent TGFß family signalling in cell fate regulation. Nature Rev Mol Cell Biol. 2023, 24: 876-94 https://doi.org/10.1038/s41580-023-00638-3 [ Links ]
10. Levander G. Tissue induction. Nature 1945: 155: 148-49. [ Links ]
11. Ripamonti U. Biomimetic functionalized surfaces and the induction of bone formation. Invited Expert Opinion Tissue Engineering 2017; 23: 1197-1209. [ Links ]
12. Langer R, Brem H, Kalterman K, Klein M, Folkm an J. Isolations of a cartilage factor that inhibits tumor neovascularization. Science 1976; 193, 4247: 70-72, DOI: 10.1126/science.93585 [ Links ]
13. Lee A, Langer R. Shark cartilage contains inhibitors of tumour angiogenesis. Science 1983; 16;221(4616):1185-7. doi: 10.1126/science.6193581. [ Links ]
14. Moses MA, Sudhalter J, Langer R. Identification of an inhibitor of neovascularization from cartilage. Science 1990. 15;248(4961):1408-10. DOI: 10.1126/science.1694043 [ Links ]
15. Moses MA, Langer R. Inhibitors of angiogenesis. Biotechnology 1991; 9(7): 630-34, doi: 10.1038/nbt0791-630 [ Links ]
16. Ripamonti U. Soluble osteogenic molecular signals and the induction of bone formation. Biomaterials 2006; 27: 807-822. [ Links ]
17. Ripamonti U, Ferretti C, Heliotis M. Soluble and insoluble signals and the induction of bone formation: Molecular therapeutics recapitulating development. J Anat 2006; 209: 447-468. [ Links ]
18. Ripamonti U, Heliotis M, Ferretti C. Bone morphogenetic proteins and the induction of bone formation: From laboratory to patients. Oral Maxillofac Surg Clin North Am. 2007; 19: 575-589. [ Links ]
19. Trueta, J. The role of the vessels in osteogenesis. J Bone Joint Surg. 1963, 45B, 402-18 [ Links ]
20. Ripamonti U. Regenerative Medicine, the Induction of Bone Formation, Bone Tissue Engineering, and the Osteogenic Proteins of the Transforming Growth Factor-ß Supergene Family. In: Ripamonti U (ed.) CRC Press Taylor & Francis, Boca Raton USA, Induction of Bone Formation in Primates. The Transforming Growth Factor- beta3; 2016, Chapter 2, 15-46. [ Links ]
21. Urist MR. Bone: formation by autoinduction. Science 1965 150(3698):893-899 doi: 10.1126/science.150.3698.893. [ Links ]
22. Turing AM. The Chemical Basis of Morphogenesis. Turing Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences, Vol. 237, No. 641. (Aug. 14, 1952), pp. 37-72. [ Links ]
23. Ripamonti U, Ramoshebi LN, Patton J, Matsaba T, Teare J, Renton L. Soluble signals and insoluble substrata: Novel molecular cues instructing the induction of bone. In: EJ Massaro and JM Rogers (Eds.), Chapter 15, The Skeleton. Humana Press, 2004; pp 217-227. [ Links ]
24. Sampath TK, Rashka KE, Doctor JS, Tucker RF, Hoffman FM. Drosophila transforming growth factor beta superfamily proteins induce endochondral bone formation in mammals. Proc Natl Acad Sci. USA 1993; 90(13), 6004-6008 https://doi.org/10.1073/pnas.90.13.6004 [ Links ]
25. Ripamonti U. Developmental patterns of periodontal tissue regeneration. Developmental diversities of tooth morphogenesis do also map capacity of periodontal tissue regeneration? J Periodont Res. 2019; 54: 10-26; doi: 10.1111/jre.12596 [ Links ]
26. Pääbo S. The Human condition-A molecular approach. Cell 2014; 157:216-26. doi:https://doi.org/10.1016/j.cell.2013.12.036 [ Links ]
27. Ripamonti U, Duarte R, Ferretti C, Reddi AH. Osteogenic competence and potency of the Bone induction principle: Inductive substrates that initiate "Bone: Formation by autoinduction". J Craniofac Surg. 2022; 33(3): 971-984. [ Links ]
28. Levander G. A study of bone regeneration. Surg Gynec Obst. 1938; 67(6): 705-14. [ Links ]
29. Reddi AH, Huggins CB. Biochemical sequences in the transformation of normal fibroblasts in adolescent rats. Proc Natl Acad Sci U S A. 1972, 69(6):1601-05. doi: 10.1073/pnas.69.6.1601. [ Links ]
30. Urist MR, Silverman BF, Büring K, Dubuc FL, Rosemberg JM. The bone induction principle Clin Orthop Relat Res. 1967 53: 243-83. [ Links ]
31. Sampath, T. K.; Reddi, A. H. Dissociative extraction and reconstitution of extracellular matrix components involved in local bone differentiation. Proc Natl Acad Sci. USA 1981; 78:7599-7603. [ Links ]
32. Sampath, T. K.; Reddi, A. H. Homology of bone-inductive proteins from human,= monkey, bovine, and rat extracellular matrix. Proc Natl Acad Sci. USA 1983, 80, 6591-95. [ Links ]
33. Ripamonti U, Reddi AH. Bone morphogenetic proteins: Applications in plastic and reconstructive surgery. Adv Plast Reconst Surg. 1995; Vol. 11: 47-65. [ Links ]
34. Urist MR, Strates BS. Bone morphogenetic protein. J Dent Res. 1971; 50: 1392-1406. [ Links ]
35. Urist MR, Hou YK, Brownell AG, Hohl WM, Buyske J, Lietze A, Tempst P, Hunkapiller M, DeLange RJ. Purification of bovine bone morphogenetic protein by hydroxyapatite chromatography. Proc Natl Acad Sci. U S A 1984; 81(2):371-5. doi: 10.1073/ pnas.81.2.371. [ Links ]
36. Sampath TK, Muthukumaran N, Reddi AH. Isolation of ostogenin, an extracellular matrix-associated, bone-inductive protein, by heparin affinity chromatography. Proc Natl Acad Sci. USA 1987; 84(20):7109-13. doi: 10.1073/pnas.84.20.7109. [ Links ]
37. Wang EA, Rosen V, Cordes P, Hewick RM, Kriz MJ, Luxenberg DP, Sibley BS, Wozney JM. Purification and characterization of other distinct bone-inducing factors. Proc Nat Acad Sci USA, 1988, 85 (24): 9484-9488, https://doi.org/10.1073/pnas.85.24.9484 [ Links ]
38. Wozney JM, Rosen V, Celeste AJ, Mitsock LM, Whitters MJ, Kriz RW, Hewick RM, Wang EA. Novel regulators of bone formation: molecular clones and activities. Science. 1988; 42(4885):1528-34. doi: 10.1126/science.3201241.PMID: 3201241 [ Links ]
39. Celeste AJ, Iannazzi JA, Taylor RC, Hewick RM, Rosen V, Wang EA, Wozney JM Identification of transforming growth factor beta family members present in bone-inductive protein purified from bovine bone. Proc Natl Acad Sci, U S A. 1990 Dec;87(24):9843-7. doi: 10.1073/pnas.87.24.9843. [ Links ]
40. Urist MR. The reality of a nebulous, enigmatic myth. Clin Orthop Rel Res. 1968; 59:3-6. 49. [ Links ]
41. Âberg T, Wozney J, Thesleff I. Expression patterns of bone morphogenetic proteins (Bmps) in the developing mouse tooth suggest roles in morphogenesis and cell differentiation. Dev Dyn. 1997; 210:383-3. https://doi.org/10.1002/(SICI)1097-0177(199712)210:4<383::AID-AJA3>3.0.CO;2-C [ Links ]
42. Ripamonti U, Duneas N. Tissue morphogenesis and regeneration by bone morphogenetic proteins. Plast Reconst Surg. 1998; 101: 227-239. [ Links ]
43. Thomadakis G, Crooks J, Rueger D, Ripamonti U. Immunolocalization of bone morphogenetic protein-2, -3 and osteogenic protein-1 during murine tooth morphogenesis and other craniofacial structures. Eur J Oral Sci. 1999; 107: 368-377. [ Links ]
44. Reddi AH. Role of morphogenetic proteins in skeletal tissue engineering and regeneration. Nat Biotechnol 1988; 16(3): 247-52. doi: 10.1038/nbt0398-247. [ Links ]
45. Ripamonti U. Soluble, insoluble and geometric signals sculpt the architecture of mineralized issues. J Cell Mol Med. 2004; 8: 169-180. [ Links ]
46. Ripamonti U, Duneas N, van den Heever B, Bosch C, Crooks J. Recombinant transforming growth factor-ß induces endochondral bone in the baboon and synergizes with recombinant osteogenic protein-1 (bone morphogenetic protein-7) to initiate rapid bone formation. J Bone Miner Res. 1997; 12: 1584-1595. [ Links ]
47. Ripamonti U, Crooks J, Matsaba T, Tasker J. Induction of endochondral bone formation by recombinant human transforming growth factor-ß2 in the baboon (Papio ursinus). Growth Factors 2000; 17: 269-285. [ Links ]
48. Ripamonti U. Osteogenic Proteins of the Transforming Growth Factor-ß Superfamily In: HL Henry and AW Norman (Eds.), Encyclopedia of Hormones. Academic Press, 2003, pp 80-86. [ Links ]
49. Ripamonti U, Ramoshebi LN, Teare J, Renton L, Ferretti C. The induction of endochondral bone formation by transforming growth factory: Experimental studies in the non-human primate Papio ursinus. J Cell Mol Med. 2008; 12: 1029-1048. [ Links ]
50. Ripamonti U. Bone induction by recombinant human osteogenic protein-1 (hOP-1, BMP-7) in the primate Papio ursinus with expression of mRNA of gene products of the TGF-ß superfamily. J Cell Mol Med. 2005; 9: 911-928. [ Links ]
51. Ripamonti U, Heliotis M, van den Heever B, Reddi AH. Bone morphogenetic proteins induce periodontal regeneration in the baboon (Papio ursinus). J Periodont Res. 1994; 29: 439-445. [ Links ]
52. Ripamonti U, Heliotis M, Sampath TK, Rueger D. Induction of cementogenesis by recombinant human osteogenic protein-1 (hOP-1/BMP-7) in the baboon (Papio ursinus). Archives of Oral Biology 1996; 41: 121-126. [ Links ]
54. Ripamonti U, Petit J-C. Bone morphogenetic proteins, cementogenesis, myoblastic stem cells and the induction of periodontal tissue regeneration. Cyt Grow Fact Rev. 2009; 20: 489-499. [ Links ]
55. Ripamonti U, Reddi AH. Periodontal regeneration: Potential role of bone morphogenetic proteins. J Periodont Res. 1994; 29: 225-235. [ Links ]
56. Ripamonti U. The induction of bone formation: From bone morphogenetic proteins to the transforming growth factor-ß3 protein - Redundancy, pleiotropy, and the induction of cementogenesis. The South Afr Dent J. 2021; 76(6): 331-356. [ Links ]
57. Ripamonti U, Crooks J, Petit J-C, Rueger D. Periodontal tissue regeneration by combined applications of recombinant human osteogenic protein-1 and bone morphogenetic protein-2. A pilot study in Chacma baboons (Papio ursinus). Eur J Oral Sci. 2001; 109: 241-248. [ Links ]
58. Ripamonti U, Crooks J, Teare J, Petit J-C, Rueger DC. Periodontal tissue regeneration by recombinant human osteogenic protein-1 in periodontally-induced furcation defects of the primate Papio ursinus. S Afr J Sci. 2002; 98: 361-368. [ Links ]
59. Ripamonti U. Re-defining the induction of periodontal tissue regeneration in primates by the osteogenic proteins of the transforming growth factor-ß supergene family. J Periodont Res. 2016; 51: 699-715. [ Links ]
60. Ripamonti U, Parak R, Klar RM, Dickens C, Dix-Peek T, Duarte R. Cementogenesis and osteogenesis in periodontal tissue regeneration by recombinant human transforming growth factor -ß3: a pilot study in Papio ursinus. J Clin Periodont. 2017; 44: 83-95. [ Links ]
61. Teare J, Ramoshebi LN, Ripamonti U. Periodontal tissue regeneration by recombinant human transforming growth factor-ß3: in Papio ursinus. J Periodont Res. 2008; 43: 1-8. [ Links ]
62. Ripamonti U, Parak R, Petit J-C. Induction of cementogenesis and periodontal ligament regeneration by recombinant human transforming growth factor-ß3: in Matrigel with rectus abdominis responding cells. J Periodont Res. 2009; 44: 141-152. [ Links ]
63. Ripamonti U, Ma S, Cunningham N, Yeates L, Reddi AH. Initiation of bone regeneration in adult baboons by osteogenin, a bone morphogenetic protein. Matrix 1992; 12: 369-380. [ Links ]
64. Coura GS, Garcez RC, Mendes de Agular CBN, Alvarez-Silva M, Magini RS, Trentin AG. Human periodontal ligament: a niche of neuronal crest stem cells. J Periodont Res 2008; 43: 331-36. [ Links ]
65. Han J, Menicanin D, Gronthos S, Bartold PM. Stem cells, tissue engineering and periodontal regeneration. Aust Dent J 2014; 59 Suppl 1:117-30. doi: 10.1111/ adj.12100. Epub 2013 Sep 23. [ Links ]
66. Teare JA, Petit J-C, Ripamonti U. Synergistic induction of periodontal tissue regeneration by binary application of hTGF-B and hOP-1 in Class II furcation defects of Papio ursinus. J Periodont Res. 2011 Dec 6. doi: 10.1111/j.1600-0765.2011.01438.x. [Epub ahead of print]; 2012; 47: 336-344. [ Links ]
67. Ripamonti U, Klar RM, Renton LF, Ferretti C. Synergistic induction of bone formation by hOP- 1, hTGF-ß3 and inhibition by zoledronate in macroporous coral-derived hydroxyapatites. Biomaterials 2010; 31: 6400-6410. [ Links ]
68. Duneas N, Crooks J, Ripamonti U. Transforming growth factor-B^ Induction of bone morphogenetic protein genes expression during endochondral bone formation in the baboon, and synergistic interaction with osteogenic protein-1 (BMP-7). Growth Factors 1998; 15: 259-277. [ Links ]
69. Ripamonti U, Parak R, Klar RM, Dickens C, Dix-Peek T, Duarte R. The synergistic induction of bone formation by the osteogenic proteins of the TGF-ß supergene family. Biomaterials 2016; 104: 279-296. Doi: 10.1016/j.biomaterials.2016.07.018. [ Links ]
70. Ripamonti U, Teare J, Ferretti C. A macroscopic bioreactor super activated by the Recombinnat human transforming growth factor-ß3. Frontiers in Physiology www.frontiersin.org 2012;3:1-18. [ Links ]
71. Ripamonti U, Duarte R, Ferretti C. Re-evaluating the induction of bone formation in primates. Biomaterials 2014; 35: 9407-9422. [ Links ]
72. Ramoshebi LN, Ripamonti U. Osteogenic protein-1, a bone morphogenetic protein, induces angiogenesis in the chick chorioallantoic membrane and synergize with basic fibroblast growth factor and transforming growth factor-ß1. Anat Rec. 2000; 259: 97-107. [ Links ]
73. Kleinman HK, McGan/ey ML, Hassell JR, Star VL, Cannon FB, Laurie GW, Martin GR. Basement membrane complexes with biological activities. Biochemistry 1986; 25(2): 312-318 DOI: 10.1021/bi00350a005 [ Links ]
74. Ripamonti U, van den Heever B, Heliotis M, Dal Mas I, Hahnle U, Biscardi A. Local delivery of bone morphogenetic proteins using a reconstituted basement membrane gel: Tissue engineering with Matrigel. S Afr J Sci. 2002; 98: 429-433. [ Links ]
75. Vukicevic S, Luyten FP, Kleinman HK, Reddi AH. Differentiation of canalicular cell processes in bone cells by basement membrane matrix components: Regulation by discrete domains of laminin. Cell1990, 63, 437-445. [ Links ]
76. Paralkar VM, Nandedkar AK, Pointer RH, Kleinman HK, Reddi AH. Interaction of osteogenin, a heparin binding bone morphogenetic protein, with type IV collagen. J Biol Chem. 1990 5;265(28):17281-4. [ Links ]
77. Vukicevic S, Latin V, Chen P, Batorsky R, Reddi AH, Sampath TK. Localization of osteogenic protein-1 (bone morphogenetic protein-7) during human embryonic development: high affinity binding to basement membranes. Biochem Biophys Res Commun. 1994; 198(2):693-700. doi: 10.1006/bbrc.1994.1100. [ Links ]
78. Paralkar VM, Vukicevic S, Reddi AH. Transforming growth factor beta type 1 binds to collagen IV of basement membrane matrix: implications for development. Dev Biol. 1991;143(2):303-8. doi: 10.1016/0012-1606(91)90081-d. [ Links ]
79. Folkman J, Klagsburn M, Sasse J, Wadzinsky M, Ingber D, Vlodavsky I. A heparin-binding protein - basic fibroblast growth factor - is stored within basement membrane. Am J Path. 1988; 130(2), 393-400 -ncbi.nim.nih.gov [ Links ]
80. Buxboim A, Discher DE. Stem Cells Feel the Difference. Nature Methods 2010, 7 (5), 695-697. [ Links ]
81. Discher DE, Janmey P, Wang Y-L. Tissue Cells Feel and Respond to the Stiffness of Their Substrate. Science 2005, 310, 1139-43. [ Links ]
82. Fu J. Wang, Y-K, Yang MT. Desai RA, Yu X, Liu Z, Chen CS, Mechanical Regulation of Cell Function with Geometrically Modulated Elastomeric Substrates. Nature Methods 2010, doi:10.1033/NMETH.1487. [ Links ]
83. Engler AJ, Sen S, Sweeney L, Discher DE. Matrix Elasticity Directs Stem Cell Lineage Specification. Cell 2006; 126: 677-89. doi10.1016/j.cell.2006.06.044. [ Links ]
84. Ripamonti U. Biomimetism, biomimetic matrices and the induction of bone formation. J Cell Moi Med. 2009; 13 9B: 2953-2972. [ Links ]
85. Luan X, Walker C, Dangaria S, Iti Y, Druzinsky R, Jarosius K, Lesot H, Rieppel O. The mosasaur tooth attachment apparatus as paradigm for the evolution of the gnathostome periodontium. Evol & Develop 2009; https://doi.org/10.1111/j.1525-142X.2009.00327.x [ Links ]
86. Jiménez-Durán K, López-Letay S, Zeichner-David M, Higinio Arzate H. Cloning, expression and biological activity of cementogenin (CMGN): A novel protein from human cementum. Manuscript ID: 1009419, 2022, www.frontiersin.org [ Links ]
87. Klar M R, Duarte R, Dix-Peek T, Ripamonti U. The induction of bone formation by the recombinant human transforming growth factor-ß3 Biomaterials 2014; 35: 2773-2788 http://dx.doi.org/10.1016//.biomatenals.2013.12.062 [ Links ]
88. Ripamonti U, Dix-Peek T, Parak R, Milner B, Duarte R. Profiling bone morphogenetic Proteins and transforming growth factor-ßs by hTGF-ß3 pre-treated coral-derived macroporous constructs: The power of one. Biomaterials 2015; 49: 90-102. [ Links ]
89. Research Article Summary, Tissue Morphogenesis, Editorial Science 382, 902, 2023. [ Links ]
90. Yang S. Palmquist KH, Nathan L, Pfeifer CR, Schultheiss PJ, Sharma A, Kam LC, Miller PW, Shyer AE, Rodrigues AR. Morphogens enable interacting supracellular phases that generate organ architecture. Science 2023; 382, 902. https://doi:10.1126/science.adg5579 [ Links ]
91. Ripamonti U. Global morphogenesis regulating tissue architecture and organogenesis. Biomaterials Advances 2025; 172 214262, https://doi.org/10.1016/j.bioadv.2025.214262 [ Links ]
92. Ripamonti U, Tsiridis E, Ferretti C, Kerawala CJ, Mantalaris A, Heliotis M. Perspectives in regenerative medicine and tissue engineering of bone. J Oral Maxillofac Surg. 2010; doi:10.1016/j.bjoms. [ Links ]
93. Ferretti C, Ripamonti U. Special Editorial: The conundrum of human osteoinduction: Is the bone induction principle failing clinical translation? J Craniofac Surg. 2021; 32: 1287-1289; doi:10.1097/SCS0000000000007429. [ Links ]
94. Ramasamy SK, Kusumbe AP, Adams RH. Regulation of tissue morphogenesis by endothelial cell-derived signals. Trends in Cell Biol. 2015; 25(3): 148-57. https://doi:10.1016/j.tcb.2014.11.007. Epub 2014 Dec 17. [ Links ]
95. Gomez-Salinero JM, Rafii S. Endothelial cell adaptation in regeneration. Science 2018;362(419): 1116-11.https://doi:10.1126/science.aar4800. [ Links ]
 Correspondence:
Correspondence:
Name: Ugo Ripamonti
Email: ugo.ripamonti@wits.ac.za













