Services on Demand
Journal
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Science
On-line version ISSN 1996-7489Print version ISSN 0038-2353
S. Afr. j. sci. vol.121 n.7-8 Pretoria Jul./Aug. 2025
https://doi.org/10.17159/sajs.2025/18999
RESEARCH ARTICLE
Sustainable strategies for sodium reduction in biltong by improving healthiness without impairing safety
Rita OppermanI; Barbara E. van WyngaardI; MacDonald CluffII; Carina BothmaIII; Eileen RoodtI; Celia HugoIV; Arno HugoI
IDepartment of Animal Science, University of the Free State, Bloemfontein, South Africa
IIB.T Enterprises, Johannesburg, South Africa
IIIDepartment of Food Systems and Development, University of the Free State, Bloemfontein, South Africa
IVDepartment of Microbiology and Biochemistry, University of the Free State, Bloemfontein, South Africa
ABSTRACT
Biltong is a popular South African savoury meat snack, which is preserved by drying and adding vinegar and salt to inhibit microbial growth. It is stable at room temperature and can be considered a safe and sustainable local food system. Unfortunately, biltong has exceptionally high sodium (Na) levels, and there are currently no regulations regarding the Na content of biltong. Therefore, research needs to be conducted to determine the effect of Na reduction or replacement on South African biltong's shelf life and chemical and sensory properties. In this study, sodium chloride (NaCl) inclusion levels in biltong models were altered to contain normal or 50% reduced NaCl in combination with various salt replacers - potassium chloride (KCl), organic potassium (K) salts and K-lactate - in different treatments. The treatments with the various levels of Na and replacers were evaluated against the positive control, the normal added NaCl levels and the negative control, which contained 50% of the normal NaCl levels. We found that a 50% reduction in NaCl was possible, and there were no serious adverse effects on the product's chemical, sensory or microbial qualities. The treatments, which contained KCl, organic K salts and K-lactate as replacers, were successful at maintaining the sensory properties of biltong in comparison to the positive control. Overall, the treatment that contained the organic K salts, in conjunction with the K-lactate, was the most effective in inhibiting the growth of Escherichia coli, Staphylococcus aureus and lactic acid bacteria.
SIGNIFICANCE:
Biltong has a very high salt content, but it is essential to ensure that this traditional South African delicacy keeps up and adheres to modern consumers' health and nutritional demands. Our findings indicate that a 50% reduction in salt is possible without adverse effects on biltong's chemical, microbial or sensory quality. These results also enable workers in the spice industry to reformulate spice packs with reduced salt, resulting in a sustainable, healthier product.
Keywords: biltong, sodium reduction, sodium replacement, technological stability, microbial shelf life
Introduction
Sodium (Na) is essential for normal human functioning, such as regulating the extracellular volume, maintaining the acid-base balance, neural transmission, renal function, cardiac output and myocytic contraction.1 However, the current average daily NaCl intake for adults of 14 g far exceeds the 5 g recommended for good health.2 There is a direct and undeniable connection between high levels of salt intake and high blood pressure, which could lead to coronary heart disease and stroke.3 In response to the increase in cardiovascular diseases in South Africa, the South African Department of Health has implemented regulations to lower the salt content of certain food products, with the goal to lower the Na intake of consumers.4
Biltong is a popular South African savoury meat snack. The biltong industry has an estimated annual turnover of ZAR2.4 billion.5 Biltong is preserved by drying and adding vinegar and salt to inhibit microbial growth. It is stable at room temperature and can be considered a safe and sustainable local food system.
Biltong has an extremely high Na content and is currently not included in these regulations. It is, therefore, important to ensure that these traditional South African delicacies keep up and adhere to the health and nutritional demands of the modern consumer.
The aim of this study was to determine the effect of 50% salt reduction and partial salt replacement on the chemical and microbial stability and the sensory quality of biltong.
Materials and methods
Sourcing of lean meat, additives and spices
All the meat used during this project was sourced from an owner-operator meat-processing plant in Bloemfontein, South Africa. The meat was collected less than 24 h before required and transported to the meat-processing facility of the University of the Free State in Bloemfontein. The meat was then spiced and the vinegar was added, and the meat was kept at 4 °C overnight. The spices, Na replacers and additives were obtained from BT Enterprises in Johannesburg, South Africa.
Formulation of the biltong
According to an unpublished survey by this research team, the average %NaCl in biltong samples from various supermarkets and butcher shops is 4.73%. This value was used as the positive control, or Treatment A, and as the basis of the formulation of all the biltong treatments used in this study. The remaining treatments were formulated to contain 50% of the NaCl of Treatment A. The formulations were based on a 60% weight loss, as recommended by the South African Council for Scientific and Industrial Research.6 Table 1 summarises the targeted %NaCl, replacer and mg Na/100 g in the dry sample for each treatment at a 60% weight loss.
Treatments B, C and D were formulated in accordance with 50% of the positive control NaCl inclusion level, in combination with various NaCl replacers. Treatment B was formulated to contain 1237.11 mg Na/100 g (2.365% NaCl) and 2.365% KCl was added as a salt replacer. Treatment C was formulated to contain 1236.52 mg Na/100 g (2.365% NaCl) and 2.365% organic K salts (potassium acetate and potassium diacetate) were added as replacers. Treatment D was formulated to contain 1236.52 mg Na/100 g (2.365% NaCl), and 2.365% K-lactate was added as a replacer. Treatment E, which was the negative control, was formulated with 2.365% NaCl (50% of the positive control) and contained 1236.98 mg Na/100 g. Treatment E did not include any replacers; therefore, the net weight had to be made up with a combination of maltodextrin and cereal binder to ensure that all the batch packs had the same net weight without altering the taste of the biltong. Monosodium glutamate (MSG) was added across all the treatments to override the possible sweetness impact of maltodextrin.
Manufacturing and sampling of the biltong
Three replicates of biltong were manufactured about 3 months apart, with fresh raw materials to compensate for variations in the raw materials, processing and environmental conditions. A single replicate consisted of five 3-kg batches.
To manufacture each replicate, 42 pieces of silver-side beef cuts (Musculus gluteobiceps and Musculus semitendinosus) weighing more or less 300 g were used. Eight pieces of meat were placed in a clean stainless-steel container and spiced, and then the vinegar was added and rubbed into the meat to ensure proper curing. In the case of treatments C and D, the K-lactate was first mixed thoroughly with the vinegar and then added to the meat pieces. The meat was turned every 8 h to ensure adequate curing.
After 16 h of curing, one piece from each treatment was sampled into a sterile bag for microbial analysis. Three pieces from each treatment were weighed and labelled using a sterile tag to be re-weighed daily for assessment of weight loss. The remaining pieces from each treatment were hung in a drying cabinet with a plastic hook until they reached the desired weight loss (60%). A conventional natural drying process was undertaken in a Crown Okto drying cabinet with the extraction fan on but without heating switched on and no humidity control. The temperature in the meat-processing facility was set at 21-23 °C to obtain an average of 22 °C. When the desired weight loss was reached, one piece of dry biltong from each treatment was sampled into a sterile bag for microbial analysis. After drying, four pieces of dry biltong (±400 g in total) per treatment were then chopped to a fine consistency using a 5-L Hobart bowl cutter, sampled into 20 mL plastic cuvettes and stored at -18 °C until analysis was carried out.
Chemical analysis
NaCl, Na and K content
For the determination of NaCl content, the Volhard method was used to volumetrically and quantitatively determine the amount of Cl present in the sample, which was then used to calculate the amount of NaCl present in the sample.7 Na and K contents were determined using atomic absorption spectroscopy. For atomic absorption spectroscopic analysis, the samples were reduced to a mineralised form free from any organic compounds by dry ashing at 525 °C according to the modified method by Nielsen.8 Atomic Absorption spectroscopy was performed using an Agilent Technologies 240FS AA spectrometer (Agilent,USA) fitted with an air-acetylene burner. A Na and K hollow cathode lamp was used as a light source. Sample absorbance values were measured and processed using the Spectra AA version 5.3 Pro software (2015) package.
Water activity
Water activity (aw) was determined using a Novasina Thermoconstanter TH 200 (Labotec, Johannesburg, South Africa) water activity meter. After equilibrium was reached with deionised distilled water, quadruplicate measurements per treatment group and replicate were made at 25 °C. The results were reported as percentage relative humidity (%RH) and converted to aw values by dividing each value by a factor of 100.
Microbial analysis
A 10 g sample from each product was aseptically weighed and placed into a sterile 207 mL WhirlPakTM bag (Lasec, Bloemfontein, South Africa), after which 90 mL of sterile 0.1 M buffered peptone water solution was added to create a 10-1 dilution. The sample was homogenised using a stomacher (AME Stomacher Lab-Blender 400, Johannesburg, South Africa) for 1 min. Further dilutions (10-2 to 10-6) were made by adding 1 mL of the 100 mL sample (10-1 dilution) in McCartney bottles containing 9 mL sterile 0.1 M phosphate buffer solution.9 One millilitre of each dilution was then plated on different selective media using the pour plate technique, except for Staphylococcus aureus determination, which employed the surface plating technique. Unless otherwise indicated, all media were sourced from ThermoFisher (Pty) Ltd (Johannesburg, South Africa).
Total bacterial count
Standard plate count agar (Oxoid CM0463) was used to enumerate the total viable counts, and the plates were incubated at 32 °C for 48 h. After incubation, the colonies were enumerated using a colony counter.
Escherichia coli and coliforms
Violet red bile agar + 4-methylumbelliferyl-p-D-glucuronide (VRBM; Oxoid CM0978) was used for total coliform counts and detecting E. coli. The VRBM plates were incubated at 37 °C for 48 h, and fluorescence under ultraviolet light (366 nm, CAMAG Universal UV Lamp) was used to indicate the presence of E. coli.
Staphylococcus aureus
Baird Parker agar (Oxoid CM0275), with egg yolk tellurite supplement (Oxoid SR0054), was used to detect S. aureus. Incubation was at 37 °C for 24 h. Staphylococcus aureus typically forms colonies that are 1.01.5 mm in diameter, black, shiny, convex with a narrow white margin and surrounded by clear zones, extending 2-5 mm into the opaque medium.9
Yeasts and moulds
Yeast and mould enumeration were carried out using Rose-Bengal chloramphenicol agar (Oxoid CM0549), with chloramphenicol supplement (Oxoid SR0078), incubated at 25 °C for 4 days.
Listeria monocytogenes
RAPID'L. mono (Biorad 356-4293; AEC-Amersham; Johannesburg, South Africa) was used with two supplements, namely, reconstituted RAPID'L. mono Supplement 1 (Biorad 3564294) and RAPID' Listeria Supplement 2 (Biorad 3564746) to enumerate L. monocytogenes. Incubation was at 37 °C for 24 h, and light blue colonies were regarded as L. monocytogenes colonies.
Enterobacteriaceae
Violet red bile glucose agar (CM0485) was used to detect Enterobacteriaceae. A double layer of agar was used to ensure anaerobic growth. Incubation was at 37 °C for 24 h.
Lactic acid bacteria
Man, Rogosa and Sharpe (MRS) agar consisting of MRS Broth (Oxoid CM0359) with 1.50% w/v agar bacteriological (Oxoid Agar No. 1, LP0011) was used to enumerate lactic acid bacteria. Incubation was at 37 °C for 24 h.
Consumer sensory evaluation
The samples of the five treatments were sliced up and placed in ziplock bags until they were evaluated. A 100-member panel of students and staff from the University of the Free State, men and women aged 20-60 years, evaluated the biltong samples and gave their opinion on the acceptability of the different biltong treatments. The nine-point hedonic scale was used, ranging from 1 (dislike extremely) to 9 (like extremely). The evaluation was carried out in separate booths, under red lights, to mask any colour differences. Diluted apple juice was provided as a palette cleanser.
Statistical analyses
An analysis of variance10 was used to determine any effects that salt reduction and replacement levels had on various quality parameters of the biltong treatments. The Tukey-Kramer multiple comparison test (α = 0.05) was carried out to identify significant differences between the treatment means.10
Results and discussion
Moisture content and water activity
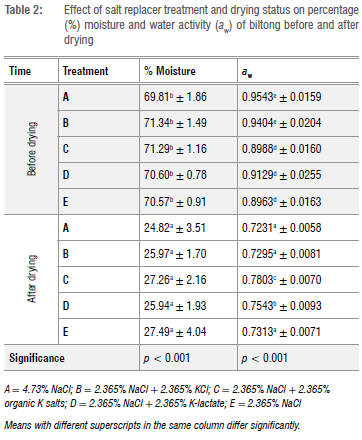
There were significant differences (p < 0.001) in the moisture content for all treatments before and after drying (Table 2). Fresh beef has a moisture content of 65-80%.11 Treatments did not differ significantly in moisture content before drying or after drying. Dried biltong has a moisture content of 20-50%.12 Wet and dry biltong from all treatments in this study were within this range (Table 2). Treatments A and B had a significantly (p < 0.001) higher aw than Treatments C, D and E before drying (Table 2). Treatments C and D had a significantly (p < 0.001) higher aw than Treatments A, B and E after drying. NaCl is known to reduce aw by drawing out water from the meat tissue through osmosis, as in the study conducted on the effect of salt on the aw of dry-cured sausages.13 It was clear that the treatments with the higher Na content had a lower aw, after drying, but the organic K salts were not as successful in lowering the aw. Petit et al.14 grouped biltong into two groups, namely dry biltong, which has a moisture content between 21.5% and 25.3% and an aw range from 0.65 to 0.68, and moist biltong, which has a moisture content between 35.1% and 42.8% and an aw range from 0.85 to 0.89. The biltong manufactured in this study had a moisture content and aw closer to the ranges classified as dry biltong.
Ash, NaCl, Na and K content
Each treatment consisted of a different formulation, with a different added NaCl content, that explained the different (p < 0.001) percentages of ash content. Treatment B (3.84%) differed significantly (p < 0.001) in ash content from Treatment E (2.78%) for the biltong samples before drying. After drying, the percentage of ash was approximately three times higher than that of the biltong samples before drying (Table 3); this is because the mixture of salt and replacers diffused slowly into the muscle tissue, reduced the free water and increased the protein density.15
The ash content of the five biltong treatments differed significantly (p < 0.001) in response to the added NaCl and replacers. The ash percentage of treatments A and B did not differ significantly after drying because the inorganic matter that remained was made up of metal oxides, which are enhanced by the amount of NaCl and KCl added.15 Treatments C and D had a significantly (p < 0.001) lower ash percentage than Treatments A and B after drying. Treatments C and D contained K-lactate, which has a lower mineral content than NaCl. Treatment E generally had a significantly (p < 0.001) lower percentage ash because it contained lower levels of NaCl and had no replacers. The cereal binder and maltodextrin were removed during the solvent wash for the ashing process.
Treatment A, the positive control, was the baseline for all the formulations. The end target for the %NaCl for Treatment A in the formulations after drying was 4.73% NaCl (Table 3), and the actual average %NaCl for Treatment A was 5.76% (Table 3). It is challenging to formulate the salt content of a product such as biltong because the NaCl is mixed in a brine solution, making it difficult to determine how much NaCl is absorbed within the meat sample and how much the NaCl content will increase after drying.
Table 3 indicates that, both before and after drying, the %NaCl of Treatments A and B differed significantly (p < 0.001) from all the other treatments, but Treatments C, D and E did not differ significantly (p < 0.001) from one another. Treatments B to E were formulated to contain half the %NaCl of Treatment A, but they also included replacers (except Treatment E), which is why Treatment A had a higher %NaCl than Treatments C-E (Table 1). The higher %NaCl in Treatment B was because the Volhard method, used to determine the amount of NaCl in a sample, also picked up the Cl from the KCl used as a replacer in Treatment B. The Volhard method determined the amount of chloride in the sample and because Treatment B contained KCl, the Cl- of KCl was also detected during the NaCl determination. If KCl is in the future used as a salt replacer in biltong, an alternative method of salt determination will have to be used. However, individual Na and K determinations are more important than NaCl determinations as far as Na reduction is concerned.
Although Treatment A had a numerically higher Na content than Treatments B-E, there were no significant differences in the Na content between the treatments before drying. However, the %NaCl indicated that there should be significant differences because the %NaCl differed significantly (p < 0.001). This lack of significant difference in Na content was probably due to the high moisture content of the products before drying, which caused a dilution effect of the Na. The Na content of Treatment A was significantly (p < 0.001) higher compared to the Na content from the other treatments after drying (Table 3), with nearly double the Na content, compared to Treatments B-E. Treatments A and E also had significantly (p < 0.001) lower K contents than Treatments B, C and D after drying. Treatments A and E were the only two formulations that did not contain added K, which is why, both before and after drying, they had the lowest K content.
All the treatments, except Treatment D, had a lower actual Na content than what was formulated for in the wet product (before drying), which can be attributed to the possibility that NaCl was not properly distributed through the biltong before hanging it to dry. The formulated Na content was calculated for the dry biltong sample at a 60% weight loss. Biltong from all dried treatments also had less Na than the formulated levels. However, they mainly differed only by 200-300 mg/100 g throughout all the treatments (Table 3). Treatment B was the only treatment with a lower actual K content than what was formulated for before and after drying. All the other treatments had higher K contents than what was formulated before and after drying. It is difficult to formulate a specific mineral content in a product like biltong that is dried because it is difficult to predict the absorption of brine.
Microbiological analyses
Although ready-to-eat meat products are produced under microbial-limiting steps, several spoilage and pathogenic bacteria have been associated with these products. Surveys of commercial biltong have shown total viable counts (TVC) of up to 7 log cfu/g, Enterobacteriaceae and coliforms up to 4 log cfu/g, yeasts up to 7 log cfu/g, moulds up to 5 log cfu/g, lactic acid bacteria (LAB) up to 8 log cfu/g, E. coli up to 1 log cfu/g, S. aureus up to 8.5 log cfu/g and L. monocytogenes were prevalent at very low incidence.16 Throughout the discussion, the main focus will be on the biltong samples after drying, because biltong is consumed in its dry state and should, therefore, be safe to eat.
There was a significant (p < 0.001) trend which indicated that the Enterobacteriaceae and coliform counts decreased after drying (Table 4). There were no significant differences in Enterobacteriaceae counts between the treatments before drying. Treatment A (0.83 log cfu/g) was significantly (p < 0.001) more effective in reducing Enterobacteriaceae counts in dry biltong compared to Treatments B, D and E. All the biltong samples before and after drying had Enterobacteriaceae counts less than the suggested 4 log cfu/g limit set by the Health Protection Agency.17
There was a decrease in coliform counts after drying in all the treatments. There were no significant differences in coliform counts between treatments before drying as well as after drying.
Although not significant, Treatment B, containing 4.75% NaCl (Table 3), was not as effective against the inhibition of coliforms as Treatment A, which contained 5.76 % NaCl.18 The detection of coliforms is used as a general indicator of sanitary conditions in the food-processing environment.19 According to Jones and co-workers12, commercial biltong may have Enterobacteriaceae and coliform counts of up to 4 log cfu/g, indicating that biltong manufactured from all treatments in this study was produced very hygienically with Enterobacteriaceae and coliform counts of the dry biltong generally below log 2 cfu/g.
Treatment B had significantly (p = 0.001) higher E. coli counts before drying than Treatments A, C and D (Table 4). There were no significant differences in E. coli counts between treatments after drying.
There were no significant differences (p = 0.752) in S. aureus counts between treatments of raw and dry biltong (Table 4). Commercial biltong could have up to 8.5 log cfu/g of S. aureus but should have less than 1 log cfu/g of E. coli.12 The S. aureus counts and E. coli presence for different treatments in this study conformed to these requirements.
No significant differences were found in the TVCs of biltong before drying and also after drying. Before and after drying, the TVCs of the biltong samples indicate that the overall microbial quality of the manufactured biltong was good because they fell within the acceptable range of 6-7 log cfu/g.17 When comparing the drying status, there was a significant (p < 0.001) increase in TVC from before drying to after drying.
No significant differences were observed in LAB counts for different treatments within the raw biltong samples, as well as within the dry biltong samples (Table 4). When comparing the drying status, there was a significant (p < 0.001) increase in LAB count from before drying to after drying. Although not statistically significant, Treatment C (5.10 log cfu/g) was the most effective, and Treatment D (5.98 log cfu/g) was the least effective in inhibiting LAB after drying (Table 4). According to Taormina20, potassium lactates, in conjunction with NaCl, at the correct concentrations, have been shown to inhibit LAB. The effects of lactate may be enhanced when used with other organic salts, such as diacetate salts21, which could explain why Treatment C, which contained 0.34% K-lactate with diacetate salts, was the most effective, and Treatment D, which contained 1.21% K-lactate without diacetate salts, was the least effective in inhibiting the LAB. A study conducted by Aaslyng et al.22 also found that Na reduction did not affect the growth of LAB. According to Jones et al.12, commercial biltong may contain up to 8 log cfu/g LAB. None of the wet or dry biltong treatments exceeded these counts.
There were no significant differences in yeast counts between the treatments before drying (Table 4). Although not always statistically significant, there was a trend towards lower yeast counts after drying compared to before drying. Treatments A, C and E had a significantly (p < 0.001) lower yeast count after drying in comparison to Treatment D. Treatment D had the same yeast count before and after drying (2.32 log cfu/g). The treatment with no replacers (E) and the treatment with organic K salts (C) were the most effective at inhibiting the growth of yeasts after drying.
No significant differences were observed in mould counts between the different biltong treatments before drying. After drying, Treatment A had significantly (p < 0.01) lower mould counts compared to Treatments B, D and E (Table 4), which indicates that the positive control was more effective at inhibiting moulds than the negative control, and the KCl and K-lactate replacers. Commercial biltong has been found to contain yeast counts of up to log 7 cfu/g and mould counts of up to log 5 cfu/g.12 None of the biltong treatments in this study exceeded these yeast and mould counts before or after drying.
Since the listeriosis outbreak of 2018, producers of ready-to-eat meat products have focused on the detection of L. monocytogenes in these products. Listeria monocytogenes is the most common bacterial contaminant present in ready-to-eat meat products post-processing.
Throughout all three replications of biltong manufactured, no Listeria monocytogenes was present before or after drying.
Sensory analysis
The 100-member panel consisted of 70% female participants, and the range in age was between 20 and 60 years, with 50% of panel members in the age bracket of 20-29 years.
No significant differences were detected in the aroma of biltong from different treatments (Table 5). Consumers preferred (p = 0.008) the texture of Treatments A and C over E (negative control). Consumers gave the positive control group samples a higher score (p = 0.024) for overall liking when compared to the negative control group (Table 5). Although the positive control got the highest ranking for aroma, texture, aftertaste and overall liking, it did not differ significantly from the treatment with replacers B, C and D. This implies that reduced salt in combination with all the replacers used in this study can be used without the consumer detecting a significant difference. Saltiness received a statistically (p < 0.046) higher ranking for Treatment A compared to all other treatments. With a slower reduction in salt content (stealth principle), the difference in saltiness could possibly not be picked up by consumers.
Panellists could detect a small but significant difference between the positive and negative controls regarding saltiness, texture, aftertaste and overall liking. Although not statistically significant, Treatment C scored the second-highest ranking for aroma, texture and aftertaste.
Conclusion
We found that a 50% reduction in NaCl and replacement of NaCl were possible, and with no significant adverse effects on the product's chemical, microbial or sensory qualities. Therefore, reducing the extremely high Na content of biltong, a popular South African shelf-stable meat snack is possible. Not only will the Na content of biltong be reduced, but also the K content will increase. The biltong will be chemically, microbiologically and sensorily acceptable if the NaCl is reduced by 50% and salt replacers are used. The meat industry can only benefit from reducing the Na content of biltong, even if there is currently no legislation that limits the Na content.
Funding
We acknowledge funding from Red Meat Research and Development -South Africa.
Data availability
The data supporting the results of this study are available upon request to the corresponding author.
Authors' contributions
R.O.: Methodology, data collection, sample analysis, writing - the original draft. B.E.v.W.: Methodology, validation, student supervision. M.C.: Methodology, validation, student supervision. C.B.: Methodology, data collection, validation. E.R.: Methodology, validation. C.H.: Conceptualisation, methodology, validation, student supervision, project leadership, project management. A.H.: Conceptualisation, methodology, data analysis, validation, student supervision, project leadership, project management, funding acquisition. All authors read and approved the final manuscript.
Declarations
We have no competing interests to declare. We have no AI or LLM use to declare. Ethical approval was provided by the General/Human Research Ethics Committee of the University of the Free State (UFS-hsd2020/1561/0911).
References
1. Dötsch M, Busch J, Batenberg M, Liem G, Tareilus E, Mueller R, et al. Strategies to reduce sodium consumption: A food industry perspective. Crit Rev Food Sci Nutr. 2009;49(9):841-851. https://doi.org/10.1080/10408390903044297 [ Links ]
2. Bhat S, Marklund M, Henry ME, Appel LJ, Croft KD, Neal B, et al. A systematic review of the sources of dietary salt around the world. Adv Nutr. 2020;11(3):677-686. https://doi.org/10.1093/advances/nmz134 [ Links ]
3. De Marco ER, Navarro JL, León AE, Steffolani ME. Sodium chloride replacement by potassium chloride in bread: Determination of sensorial potassium threshold and effect on dough properties and breadmaking quality. Int J Gastron Food Sci. 2022;27, Art. #100486. https://doi.org/10.1016/j.ijgfs.2022.100486 [ Links ]
4. South African Department of Health (DoH). Regulations relating to the reduction of sodium in certain foodstuffs and related matters: Amendment. Foodstuffs, Cosmetics and Disinfectants Act, 1972 (act 54 of 1972). Government Gazette No. 42496, 31 May 2019. Government Notice No. 812. [ Links ]
5. Coleman A. 'Biltong must secure geographical indication' - economist. Farmers Weekly. 2019 September 30. [ Links ]
6. Council for Scientific and Industrial Research (CSIR). The preparation of biltong. Pretoria: CSIR; 1994. p. 5-16. [ Links ]
7. Association of Official Analytical Chemists (AOAC). Official methods of analysis. 18th ed. Washington DC: AOAC; 2005. [ Links ]
8. Nielsen SS. Sodium and potassium determinations by atomic absorption spectroscopy and inductively coupled plasma-atomic emission spectroscopy. In: Nielsen SS, editor. Food analysis laboratory manual. 2nd ed. New York: Springer; 2010. p. 87-93. https://doi.org/10.1007/978-1-4419-1463-7 [ Links ]
9. Harrigan WF. Laboratory methods in food microbiology. 3rd ed. San Diego, CA: Academic Press; 1998. [ Links ]
10. NCSS LLC. NCSS 11 statistical software. Kaysville, UT: NCSS LLC; 2018. [ Links ]
11. Ren Sun D. Monitoring of moisture contents and rehydration rates of microwave vacuum and hot air dehydrated beef slices and splits using hyperspectral imaging. Food Chem. 2022;382, Art. #132346. https://doi.org/10.1016/j.foodchem.2022.132346 [ Links ]
12. Jones M, Arnaud E, Gouws P Hoffman LC. Processing of South African biltong - A review. S Afr J Anim Sci. 2017;47(6):743-757. https://doi.org/10.4314/sajas.v47i6.2 [ Links ]
13. Patarata L, Fernandez L, Silva JA, Fraqueza MJ. The risk of salt reduction in dry-cured sausage assessed by the influence on water activity and the survival of Salmonella. Foods. 2022;11(3), Art. #444. https://doi.org/10.3390/foods11030444 [ Links ]
14. Petit T, Caro Y, Petit A, Santchurn SJ, Collignan A. Physicochemical and microbiological characteristics of biltong, a traditional salted dried meat of South Africa. Meat Sci. 2014;96(3):1313-1317. https://doi.org/10.1016/j.meatsci.2013.11.003 [ Links ]
15. Honikel KO. The use and control of nitrate and nitrite for processing of meat products. Meat Sci. 2008;78(1-2):68-76. https://doi.org/10.1016/j.meatsci.2007.05.030 [ Links ]
16. Naidoo K. The microbial ecology of biltong in South Africa during production and at point-of-sale [dissertation]. Johannesburg: University of the Witwatersrand; 2010. [ Links ]
17. Health Protection Agency. Guidelines for assessing the microbiological safety of ready-to-eat foods placed in the market. London: Health Protection Agency; 2009. [ Links ]
18. Kurlansky M. Salt: A world history. London: Vintage Books; 2003. [ Links ]
19. Fung DYC. Microbial hazards in food: Food-borne infections and intoxicants. In: Toldrá F, editor. Handbook of meat processing. Ames, lA: Blackwell Publishing; 2010. p. 481-500. https://doi.org/10.1002/9780813820897.ch28 [ Links ]
20. Taormina PJ. Implications of salt and sodium reduction on microbial food safety. Crit Rev Food Sci Nutr. 2010;50(3):209-227. https://doi.org/10.1080/10408391003626207 [ Links ]
21. Verma AK, Banerjee R. Low-sodium meat products: Retaining salty taste for sweet health. Crit Rev Food Sci Nutr. 2012;52(1):72-84. https://doi.org/10.1080/10408398.2010.498064 [ Links ]
22. Aaslyng MD, Vestergaard C, Koch AG. The effect of salt reduction on sensory quality and microbial growth in hotdog sausages, bacon, ham and salami. Meat Sci. 2014;96(1):47-55. https://doi.org/10.1016/j.meatsci.2013.06.004 [ Links ]
 Correspondence:
Correspondence:
Arno Hugo
Email: hugoa@ufs.ac.za
Received: 28 June 2024
Revised: 25 Mar. 2025
Accepted: 02 Apr. 2025
Published: 11 Aug. 2025
Editors: Annchen Mielmann, Leslie Swartz
Funding: Red Meat Research and Development - South Africa













