Services on Demand
Journal
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Surgery
On-line version ISSN 2078-5151Print version ISSN 0038-2361
S. Afr. j. surg. vol.62 n.2 Cape Town 2024
https://doi.org/10.36303/SAJS.00465
HPB SURGERY
Effect of preoperative biliary drainage on intraoperative biliary cultures and surgical outcomes after pancreatic resection
MM BernonI; S SteenkampI; S DlaminiII; M MillerIII; S SobnachI; N LeechI; U KotzeI; JEJ KrigeI; EG JonasI
ISurgical Gastroenterology and HPB Surgical Unit, Department of Surgery, Groote Schuur Hospital, University of Cape Town, South Africa
IIDivision of Infectious Diseases & HIV Medicine, Department of Medicine, Groote Schuur Hospital, University of Cape Town, South Africa
IIIDepartment of Anaesthesia and Perioperative Medicine, Groote Schuur Hospital, University of Cape Town, South Africa
ABSTRACT
BACKGROUND: Prolonged obstructive jaundice (OJ), associated with resectable pancreatic pathology, has many deleterious effects that are potentially rectifiable by preoperative biliary drainage (POBD) at the cost of increased postoperative infective complications. The aim of this study is to assess the impact of POBD on intraoperative biliary cultures (IBCs) and surgical outcomes in patients undergoing pancreatic resection.
METHODS: Data from patients at Groote Schuur Hospital, Cape Town, between October 2008 and May 2019 were analysed. Demographic, clinical, and outcome variables were evaluated, including perioperative morbidity, mortality, and 5-year survival.
RESULTS: Among 128 patients, 69.5% underwent POBD. The overall perioperative mortality in this study was 8.8%. The POBD group had a significantly lower perioperative mortality rate compared to the non-drainage group (5.6% vs. 25.6%). POBD patients had a higher incidence of surgical site infections (55.1% vs. 23.1%), polymicrobial growth from IBCs and were more likely to culture resistant organisms. Five-year survival was similar in the two groups.
CONCLUSION: POBD was associated with a high incidence of resistant organisms on the IBCs, a high incidence of surgical site infections and a high correlation between cultures from the surgical site infection and the IBCs.
Keywords: pancreaticoduodenectomy, preoperative biliary drainage, surgical site infection, complications, pancreatic cancer, obstructive jaundice
Introduction
Obstructive jaundice (OJ) is a common presenting symptom in patients with periampullary tumours who may need surgical resection, which is the only curative-intended treatment for patients with periampullary cancers.1,2 Unrelieved and prolonged OJ is associated with malnutrition, coagulopathy, impaired immune and renal function and poor wound healing.3 Infective complications occur in up to 38% of patients after pancreaticoduodenectomy and are associated with increased length of hospital stay, increased cost of treatment, need for additional invasive procedures, post-pancreatectomy bleeding, delay in commencing adjuvant chemotherapy and diminished disease-free survival.4 Endoscopic or percutaneous preoperative biliary drainage (POBD) will correct the physiological abnormalities associated with OJ.5 Endoscopic stenting with placement of self-expanding metal stents (SEMS) is favoured.6,7 Routine POBD, however is associated with an increase in postoperative infective complications and has generally been reserved for patients with cholangitis, malnutrition, very high bilirubin levels and those requiring neo-adjuvant therapy.8-12
Perioperative antimicrobial therapy has been shown to reduce surgical site infections. Evidence suggests that patients who undergo POBD biliary drainage may benefit from more prolonged antimicrobial therapy.13 Antibiotics used for prophylaxis should be tailored to the antibiotic sensitivities of the most prevalent organisms cultured in bile of patients following POBD at the specific institution.
The aim of this study was to evaluate the effect of POBD on the intraoperative biliary cultures (IBCs) of patients undergoing pancreaticoduodenectomy (PD) and to assess a possible correlation between IBCs, postoperative infective complications and mortality.
Patients and methods
Patients who underwent a PD or total pancreatectomy at Groote Schuur Hospital, Cape Town, between 1 October 2008 and 31 May 2019 and were followed up for at least five years were identified from a Human Research Ethics Committee approved registry (HREC R016/2016). Only patients with malignant pathology who had IBCs performed during the pancreatic resection were included in the study. Perioperative variables analysed included demographic data, preoperative blood tests, operation performed, details of POBD, prophylactic antibiotic use, morbidity and management of complications, length of ICU and hospital stay, tumour stage and mortality (perioperative and 5-year overall survival).
Perioperative morbidity and mortality were defined as complications or death occurring within 30 days of surgery or during the index hospital admission. Postoperative bleeding, pancreatic leaks and delayed gastric emptying were defined and graded according to the International Study Group of Pancreatic Surgery definitions.14 Sites of infection and surgical site infections were classified according to the Centre for Disease Control and Prevention guideline for the prevention of surgical site infection15 and the severity of postoperative complications were graded according to the Clavien-Dindo classification.16
Intraoperative biliary cultures (IBCs) in patients who underwent POBD were compared to those without drainage. Sub-analyses were performed in patients with positive and negative IBCs in the two groups comparing length of ICU and hospital stay, tumour stage, and morbidity and mortality. In patients with infective complications, postoperatively acquired microbiological specimens were compared to IBCs. In addition, prophylactic antibiotics use was evaluated.
Statistical analysis was performed using IBM SPSS Statistics version 29.0 software. Demographic and clinical characteristics were described as frequencies and percentages or as a median and range. Survival data were analysed using the Kaplan-Meier method. Differences between the POBD and non-drained groups were evaluated using the Fisher exact test for categorical data and the t-test for continuous data. The statistical significance level was set at 0.05 and all p-values were two sided.
Ethical approval for the study was obtained from the University of Cape Town Human Research Ethics Committee (HREC 065/2021).
Results
One hundred and twenty-eight patients met the inclusion criteria of whom 89 (69.5%) had POBD (Figure 1). Seventy-three (82.0%) patients in the POBD group had endoscopically placed biliary stents, 12 (13.5%) had percutaneously placed stents and four (4.5%) had combined endoscopic and percutaneous procedures.
The POBD and non-drainage groups had similar demographic and preoperative blood results with the only significant difference being a higher preoperative total bilirubin in the POBD group (Table I). Sixty-six patients (51.6%) had pancreatic ductal adenocarcinoma (PDAC), 35 (27.3%) ampullary adenocarcinoma and 21(16.4%) distal cholangiocarcinoma. A higher proportion of patients in the POBD group had PDAC (66.7% vs 45%) although this result was not significant (Table I).
One hundred and twenty-one patients (94.5%) had a pylorus preserving pancreaticoduodenectomy, three (2.3%) a classic pancreaticoduodenectomy and four (3.1%) a total pancreaticoduodenectomy. Eighty-one (63.3%) patients had T3 tumours, 35 (27.3%) had T2 tumours, nine (7.0%) had T1 tumours and three (2.3%) had T4 tumours. Prophylactic antibiotic use varied with no standardised policy for patients undergoing pancreatic surgery (Table I).
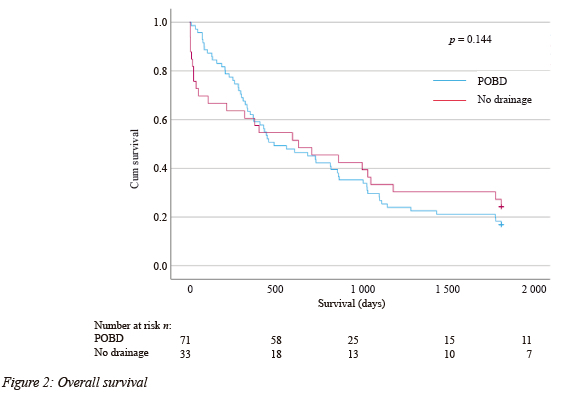
The overall perioperative mortality rate in this study was 8.8%. The perioperative mortality rate in the non-drainage group was significantly higher than the POBD group (25.6% versus 5.6%). The deaths in the non-drainage group occurred earlier than the deaths in the POBD group (median 5 vs. 17 days post-surgery) and were more frequently related to non-surgical complications (Table II). One hundred and four (81.3%) patients were followed up until death or at least five years. The overall 5-year survival was 19.2% with no difference between the two groups (Figure 2).
The culture results in patients who underwent POBD were compared to those who did not have drainage. The upfront surgical group were significantly more likely to have no growth on the IBC compared to the POBD group (38.5% vs 6.7%). Patients in the POBD were more likely to grow multiple organisms. The most common bacteria cultured from IBC were Enterobacter cloacae, Klebsiella pneumonia, Escherichia coli and Enterococcus faecalis (Table III). Escherichia coli was significantly more common in the POBD drainage group (22.47% vs 2.56%). Candida spp were cultured in 9 (10.11%) patients in the POBD drainage group and none of the non-drainage group. The organisms cultured in the POBD were more likely to be resistant to commonly used antibiotics (Supplementary Tables I and II).
Surgical site infections occurred in 58 (45.3%) patients and pancreatic fistula in 29 (22.7%) of patients and were significantly more likely to occur in the POBD group (Table II). Fifteen (11.7%) patients with surgical site infections required percutaneous drainage of intra-abdominal collections. Post-pancreatectomy bleeding occurred in 11 (8.5%) patients of whom seven required angiography with stenting or coiling of the bleeding vessel (Table II).
Forty-five (50.6%) patients in the POBD group had at least one positive postoperative culture versus 21 (53.8%) in the non-drainage group. These cultures were significantly more likely to correlate with the IBC in the POBD group (40.0% vs 19.0%).
Discussion
Preoperative biliary drainage is known to result in colonisation of the biliary tract and has been shown in several meta-analyses to be associated with an increased incidence of surgical site infections.4,17,18 These findings were confirmed in this study, with the non-drainage group significantly more likely to have no growth on the IBC compared to the POBD group. Patients undergoing POBD were also more likely to culture resistant organisms and have polymicrobial growth from the IBC. Candida were also more commonly cultured in the POBD group. The IBCs in our cohort were similar to studies from other parts of the world (Tables III and IV).19-22
Prolonged systemic perioperative antimicrobial therapy compared to perioperative prophylactic antimicrobial therapy only at the time surgery has been shown to decrease the incidence of deep surgical site infections.13,23,24 In this study patients in the POBD group were more likely to develop surgical site infections and have a correlation between IBCs and postoperative cultures. As POBD results in biliary contamination with organisms that are often resistant to conventional prophylactic antibiotics, antimicrobial therapy should be guided by local hospital surveillance data based on routine IBCs during pancreatic resections.25-30 The IBCs in this study support the use of piperacillin and tazobactam, ceftriaxone, ciprofloxacin, bactrim (sulfamethoxazole and trimethoprim), aminoglycosides and carbapenems. A high proportion of the cultured organisms were resistant to co-amoxiclav and cefuroxime.
The overall perioperative mortality rate in this study was 8.8% which is comparable to other studies.31 Unexpectedly there was a significantly higher perioperative mortality in the non-drainage group (25.6% versus 5.6%) in contrast to most other studies. These findings support routine POBD in our patient population. Many of the patients in this study were from low socio-economic backgrounds with extensive comorbidities and very high bilirubin levels (> 250 umol/l) at presentation. This cohort of patients may benefit more from POBD and prehabilitation than patients from better resourced environments with fewer comorbidities and lower bilirubin levels at presentation.32-34 The perioperative deaths in the non-drainage group occurred earlier and were mostly due to multiple organ failure. The deaths in the POBD drainage occurred later and were frequently associated with surgical site infections, pancreatic fistulas and post-pancreatectomy bleeding. Despite the significant differences in perioperative deaths there was no significant difference in the 5-year survival between the two groups.
Although neo-adjuvant chemotherapy (NAC) has primarily been used in patients with borderline resectable PDAC, there is an increasing trend towards offering neo-adjuvant chemotherapy to patients with upfront resectable PDAC to allow improved assessment of tumour biology and prehabilitation prior to surgery.35-38 This has resulted in more patients requiring POBD. This trend may be associated with an increase in surgical site infections.
Limitations of this study include the variable use of prophylactic antibiotics and the lack of a defined antibiotic policy making assessment of prophylactic antibiotic use difficult. The study was also conducted in a single centre necessitating a long study period to include an adequate number of patients. IBCs were not performed routinely in all patients during the study period with 33 (19.3%) patients being excluded because they did not have an IBC.
In conclusion, this is one of a few studies from sub-Saharan Africa assessing the effect of POBD on IBCs and surgical outcomes after pancreatic resection. POBD was associated with a high incidence of resistant organisms on the IBCs, a high incidence of surgical site infections and a high correlation between cultures from the surgical site infection and the IBCs. This supports prolonged antibiotic therapy after pancreatic surgery guided by local hospital surveillance data based on routine IBCs during pancreatic resections.
Conflict of interest
The authors declare no conflict of interest.
Funding source
None.
Ethical approval
Ethical approval for the study was obtained from the University of Cape Town Human Research Ethics committee (HREC 065/2021).
ORCID
MM Bernon https://orcid.org/0000-0002-7967-8548
M Miller https://orcid.org/0000-0001-6137-7723
S Sobnach https://orcid.org/0000-0002-4456-2115
N Leech https://orcid.org/0009-0002-6973-8241
U Kotze https://orcid.org/0000-0003-1405-474X
JEJ Krige https://orcid.org/0000-0002-7057-9156
EG Jonas https://orcid.org/0000-0003-0123-256X
REFERENCES
1. Schneider M, Hackert T, Strobel O, Büchler MW. Technical advances in surgery for pancreatic cancer. Br J Surg. 2021;108(7):777-85. https://doi.org/10.1093/bjs/znab133. [ Links ]
2. Strobel O, Neoptolemos J, Jäger D, Büchler MW. Optimising the outcomes of pancreatic cancer surgery. Nat Rev Clin Oncol. 2019;16(1):11-26. https://doi.org/10.1038/s41571-018-0112-1. [ Links ]
3. Clarke DL, Pillay Y, Anderson F, Thomson SR. The current standard of care in the periprocedural management of the patient with obstructive jaundice. Ann R Coll Surg Engl. 2006;88(7):610-6. https://doi.org/10.1308/003588406X149327. [ Links ]
4. Fang Y, Gurusamy KS, Wang Q, et al. Preoperative biliary drainage for obstructive jaundice. Cochrane Database Syst Rev. 2012;9(9):CD005444. https://doi.org/10.1002/14651858.CD005444.pub3. [ Links ]
5. Lai EC, Lau SH, Lau WY. The current status of preoperative biliary drainage for patients who receive pancreaticoduodenectomy for periampullary carcinoma: A comprehensive review. Surgeon. 2014;12(5):290-6. https://doi.org/10.1016/j.surge.2014.02.004. [ Links ]
6. Satoh D, Matsukawa H, Shiozaki S. The optimal type and management of biliary drainage in patients with obstructive jaundice who undergo pancreaticoduodenectomy. In Vivo. 2022;36(1):391-7. https://doi.org/10.21873/invivo.12716. [ Links ]
7. Pang L, Wu S, Kong J. Comparison of efficacy and safety between endoscopic retrograde cholangiopancreatography and percutaneous transhepatic cholangial drainage for the treatment of malignant obstructive jaundice: A systematic review and meta-analysis. Digestion. 2023;104(2):85-96. https://doi.org/10.1159/000528020. [ Links ]
8. Blacker S, Lahiri RP, Phillips M, et al. Which patients benefit from preoperative biliary drainage in resectable pancreatic cancer? Expert Rev Gastroenterol Hepatol. 2021;15(8):855-63. https://doi.org/10.1080/17474124.2021.1915127. [ Links ]
9. Sauvanet A, Boher JM, Paye F, et al; French Association of Surgery. Severe jaundice increases early severe morbidity and decreases long-term survival after pancreaticoduodenectomy for pancreatic adenocarcinoma. J Am Coll Surg. 2015;221(2):380-9. https://doi.org/10.1016/j.jamcollsurg.2015.03.058. [ Links ]
10. Van der Gaag NA, Rauws EA, van Eijck CH, et al. Preoperative biliary drainage for cancer of the head of the pancreas. N Engl J Med. 2010;362(2):129-37. https://doi.org/10.1056/NEJMoa0903230. [ Links ]
11. Lu B, Chen Y, Qin S, Chen J. Value of preoperative biliary drainage in pancreatic head cancer patients with severe obstructive jaundice: A multicentre retrospective study. Saudi J Gastroenterol. 2023. https://doi.org/10.4103/sjg.sjg_296_23. [ Links ]
12. Van Gils L, Verbeek R, Wellerdieck N, et al; RegionalAcademic Cancer Centre Utrecht (RAKU). Preoperative biliary drainage in severely jaundiced patients with pancreatic head cancer: A retrospective cohort study. HPB (Oxford). 2022;24(11):1888-97. https://doi.org/10.1016/j.hpb.2022.05.1345. [ Links ]
13. Degrandi O, Buscail E, Martellotto S, et al. Perioperative antibiotherapy should replace prophylactic antibiotics in patients undergoing pancreaticoduodenectomy preceded by preoperative biliary drainage. J Surg Oncol. 2019;120(4):639-45. https://doi.org/10.1002/jso.25622. [ Links ]
14. Bassi C, Marchegiani G, Dervenis C, et al; International Study Group on Pancreatic Surgery (ISGPS). The 2016 update of the International Study Group (ISGPS) definition and grading of postoperative pancreatic fistula: 11 years after. Surgery. 2017;161(3):584-91. https://doi.org/10.1016/j.surg.2016.11.014. [ Links ]
15. Berríos-Torres SI, Umscheid CA, Bratzler DW, et al; Healthcare Infection Control Practices Advisory Committee. Centers for Disease Control and Prevention Guideline for the Prevention of Surgical Site Infection, 2017. JAMA Surg. 2017;152(8):784-91. https://doi.org/10.1001/jamasurg.2017.0904. [ Links ]
16. Clavien PA, Barkun J, de Oliveira ML, et al. The Clavien-Dindo classification of surgical complications: five-year experience. Ann Surg. 2009;250(2):187-96. https://doi.org/10.1097/SLA.0b013e3181b13ca2. [ Links ]
17. Sewnath ME, Karsten TM, Prins MH, et al. A meta-analysis on the efficacy of preoperative biliary drainage for tumors causing obstructive jaundice. Ann Surg. 2002;236(1):17-27. https://doi.org/10.1097/00000658-200207000-00005. [ Links ]
18. Pisters PW, Hudec WA, Hess KR, et al. Effect of preoperative biliary decompression on pancreaticoduodenectomy-associated morbidity in 300 consecutive patients. Ann Surg. 2001;234(1):47-55. https://doi.org/10.1097/00000658-200107000-00008. [ Links ]
19. Bilgiç Ç, Keske Ş, Sobutay E, et al. Surgical site infections after pancreaticoduodenectomy: Preoperative biliary system interventions and antimicrobial prophylaxis. Int J Infect Dis. 2020;95:148-52. https://doi.org/10.1016/j.ijid.2020.04.005. [ Links ]
20. Krüger CM, Adam U, Adam T, et al. Bacterobilia in pancreatic surgery - conclusions for perioperative antibiotic prophylaxis. World J Gastroenterol. 2019;25(41):6238-47. https://doi.org/10.3748/wjg.v25.i41.6238. [ Links ]
21. Maatman TK, Weber DJ, Qureshi B, et al. Does the microbiology of bactibilia drive postoperative complications after pancreatoduodenectomy? J Gastrointest Surg. 2020;24(11):2544-50. https://doi.org/10.1007/s11605-019-04432-5. [ Links ]
22. Müssle B, Hempel S, Kahlert C, et al. Prognostic impact of bacterobilia on morbidity and postoperative management after pancreatoduodenectomy: A systematic review and meta-analysis. World J Surg. 2018;42(9):2951-62. https://doi.org/10.1007/s00268-018-4546-5. [ Links ]
23. Donald GW, Sunjaya D, Lu X, et al. Perioperative antibiotics for surgical site infection in pancreaticoduodenectomy: Does the SCIP-approved regimen provide adequate coverage? Surgery. 2013;154(2):190-6. https://doi.org/10.1016/j.surg.2013.04.001. [ Links ]
24. Sourrouille I, Gaujoux S, Lacave G, et al. Five days of postoperative antimicrobial therapy decreases infectious complications following pancreaticoduodenectomy in patients at risk for bile contamination. HPB (Oxford). 2013;15(6):473-80. https://doi.org/10.1111/hpb.12012. [ Links ]
25. Herzog T, Belyaev O, Akkuzu R, et al. The impact of bile duct cultures on surgical site infections in pancreatic surgery. Surg Infect (Larchmt). 2015;16(4):443-9. https://doi.org/10.1089/sur.2014.104. [ Links ]
26. Stecca T, Nistri C, Pauletti B, et al. Bacterobilia resistance to antibiotic prophylaxis increases morbidity after pancreaticoduodenectomy: A monocentric retrospective study of 128 patients. Updates Surg. 2020;72(4):1073-80. https://doi.org/10.1007/s13304-020-00772-z. [ Links ]
27. Sugimachi K, Iguchi T, Mano Y, et al. Significance of bile culture surveillance for postoperative management of pancreatoduodenectomy. World J Surg Oncol. 2019;17(1):232. https://doi.org/10.1186/s12957-019-1773-7. [ Links ]
28. Windisch O, Frossard JL, Schiffer E, et al. Microbiologic changes induced by biliary drainage require adapted antibiotic prophylaxis during duodenopancreatectomy. Surg Infect (Larchmt). 2019;20(8):677-82. https://doi.org/10.1089/sur.2019.088. [ Links ]
29. Kondo K, ChijiiwaK, Ohuchida J, et al. Selection of prophylactic antibiotics according to the microorganisms isolated from surgical site infections (SSIs) in a previous series of surgeries reduces SSI incidence after pancreaticoduodenectomy. J Hepatobiliary Pancreat Sci. 2013;20(3):286-93. https://doi.org/10.1007/s00534-012-0515-9. [ Links ]
30. Mohan A, Gupta R, Yadav TD, et al. Association of intraoperative bile culture with postoperative complications after pancreaticoduodenectomy. Surg Infect (Larchmt). 2022;23(4):351-6. https://doi.org/10.1089/sur.2021.215. [ Links ]
31. Baum P, Diers J, Lichthardt S, et al. Mortality and complications following visceral surgery: A nationwide analysis based on the diagnostic categories used in German hospital invoicing data. Dtsch Arztebl Int. 2019;116(44):739-46. https://doi.org/10.3238/arztebl.2019.0739. [ Links ]
32. West MA, Jack S, Grocott MPW. Prehabilitation before surgery: Is it for all patients? Best Pract Res Clin Anaesthesiol. 2021;35(4):507-16. https://doi.org/10.1016/j.bpa.2021.01.001. [ Links ]
33. Trestini I, Cintoni M, Rinninella E, et al. Neoadjuvant treatment: A window of opportunity for nutritional prehabilitation in patients with pancreatic ductal adenocarcinoma. World J Gastrointest Surg. 2021;13(9):885-903. https://doi.org/10.4240/wjgs.v13.i9.885 [ Links ]
34. Bundred JR, Kamarajah SK, Hammond JS, et al. Prehabilitation prior to surgery for pancreatic cancer: A systematic review. Pancreatology. 2020;20(6):1243-50. https://doi.org/10.1016/j.pan.2020.07.411. [ Links ]
35. Lindemann J, du Toit L, Kotze U, et al. Survival equivalence in patients treated for borderline resectable and unresectable locally advanced pancreatic ductal adenocarcinoma: A systematic review and network meta-analysis. HPB (Oxford). 2021;23(2):173-86. https://doi.org/10.1016/j.hpb.2020.09.022. [ Links ]
36. Jonas E, Krige J, Bernon M. Letter to the editor: Comment on outcomes of patients with borderline resectable pancreatic cancer treated with combination chemotherapy. J Gastrointest Cancer. 2021;52(3):1188-9. https://doi.org/10.1007/s12029-021-00656-4. [ Links ]
37. Jonas E, Lindemann J, Bernon M, Krige J. Letter comments re: Multi-agent neoadjuvant chemotherapy improves survival in early-stage pancreatic cancer: A NCDB analysis. Eur J Cancer. 2021;158:36-7. https://doi.org/10.1016/j.ejca.2021.09.001. [ Links ]
38. Kamarajah SK, Chatzizacharias N, Hodson J, et al. Intention to treat outcomes among patients with pancreatic cancer treated using International Study Group on Pancreatic Surgery recommended pathways for resectable and borderline resectable disease. ANZ J Surg. 2021;91(7-8):1549-57. https://doi.org/10.1111/ans.16643. [ Links ]
 Correspondence:
Correspondence:
email: mm.bernon@uct.ac.za
Supplementary files available online
The supplementary file is available in pdf: [Supplementary file]













