Services on Demand
Journal
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
SAMJ: South African Medical Journal
On-line version ISSN 2078-5135Print version ISSN 0256-9574
SAMJ, S. Afr. med. j. vol.115 n.3 Pretoria Apr. 2025
https://doi.org/10.7196/SAMJ.2025.v115i3.2696
RESEARCH
High frequency of SARS-CoV-2 infection in children admitted to academic hospitals in central South Africa
O P KhaliqI; S C BrownII; B PitsoIII; N E TabaneIII
IMMedSc, PhD; Department of Paediatrics and Child Health, School of Clinical Medicine, Faculty of Health Sciences, University of the Free State, Bloemfontein, South Africa
IIMMed (Paeds), PhD; Department of Paediatrics and Child Health, School of Clinical Medicine, Faculty of Health Sciences, University of the Free State, Bloemfontein, South Africa
IIIMB ChB, MMed (Paeds); Department of Paediatrics and Child Health, School of Clinical Medicine, Faculty of Health Sciences, University of the Free State, Bloemfontein, South Africa
ABSTRACT
BACKGROUND. During the COVID-19 pandemic, the Omicron variant was highly infectious in children, and resulted in high hospital admission rates in this population compared with other SARS-CoV-2 variants.
OBJECTIVE. To investigate the prevalence of SARS-CoV-2 infection in children during the Omicron variant wave in Free State Province, South Africa (SA).
METHODS. This prospective cross-sectional study was conducted from August 2022 to April 2023 in the Free State, SA. A total of 320 children admitted at Pelonomi Tertiary Hospital and Universitas Academic Hospital between the ages of 0 and 12 years were recruited. All participants were tested for SARS-CoV-2 using the nucleocapsid antibody rapid test. Parent and caregiver vaccination history was also collected.
RESULTS. In our study, 46.8% of the children tested positive for SARS-CoV-2. The highest (60%) infection rate was observed in neonates. Children were admitted for various reasons, and none were screened for suspected SARS-CoV-2 on admission. Of the infected population, 28% were premature, 12.6% had gastrointestinal tract infections, 12% had respiratory conditions and 10% had central nervous system conditions. A total of 43.3% of the infected children were from vaccinated parents or caregivers.
CONCLUSION. Our study showed that a high number of hospitalised children tested positive for SARS-CoV-2 while admitted for conditions unrelated to COVID-19. Most, if not all, children did not exhibit COVID-19-specific symptoms, and this may be due to the Omicron variant, which was highly infectious but less virulent, and was associated with mild disease.
Keywords: SARS-CoV-2, Omicron variant, antibodies, children, admission diagnosis
The COVID-19 pandemic affected the globe from 2020 to 2023, following its discovery in December 2019 in Wuhan, China.[1] Globally, >2.6 million cases and 17 000 deaths were reported from 17 April 2023 to 14 May 2023.[2] The virus has mutated rapidly since its discovery, resulting in different variants worldwide.[3] However, vaccinations for COVID-19 were manufactured to protect individuals from severe complications after infection.[4] The safety of these vaccines in children was unknown. Therefore, children under the age of 12 years were not vaccinated in South Africa (SA) until July 2023.
SA has had four variants since the emergence of COVID-19, and this study focused on the fourth variant, Omicron.[3] The Omicron variant was first discovered in SA and Botswana in November 2021.[5] This variant was reported to spread more rapidly than previous variants.[6] Interestingly, infections were noted even in environments where COVID-19 safety protocols, such as wearing of masks, were observed.[7] The Omicron variant was also reported to have high asymptomatic carriage, with mild disease patterns, making it difficult to detect and control.[8]
During the emergence of the Omicron variant, the virus spread rapidly in adults and children, with higher admission numbers noted in paediatric wards in Germany.[9] Another increase of paediatric admissions was noted in the USA.[10] In Romania, paediatric cases also increased in children (>10%).[11] Countries such as China, Korea, Japan and SA experienced increased paediatric admissions during the time of the Omicron variant.[12-15]
The Omicron variant is involved with lower antibody neutralisation and lower vaccine effectiveness, resulting in risk of re-infection.[6] Moreover, antibody neutralisation is higher in children with mild infections than in adults with mild infections, and even higher in children <10 years of age than older children.[16] Cloete et al.[5] hypothesised that increased paediatric admissions were attributed to decreased frequency of mask use in children, and lower vaccination rates, as children <12 years were not vaccinated at the time of their study.[15]
Children wore masks less frequently than adults because it was believed that they had a lower expression level of angiotensin-converting enzyme II (ACE2) and transmembrane protease serine 2 (TMPRSS2) (binding sites for the SARS-CoV-2 virus) than adults.[17-19] The low expression of ACE2 receptors is suspected to result in mild symptoms in children. At the same time, other findings suggest that this could be due to children having fewer or no underlying conditions.[20,21]
The immune response in children and adults is also reported to differ. Chou et al.[22] have shown that children have stronger interferon (IFN) responses to SARS-CoV-2 and more T-cells than adults. The stronger IFN response to infection results in a lower cytokine storm, leading to mild infection.[21] Furthermore, cytokine levels in children with mild COVID-19 were similar to those of healthy children, indicating that children only have a mild inflammatory response during infection.[23] Children with mild COVID-19 infections are usually asymptomatic, and where not, have shorter hospitalisation stays than those with severe COVID-19 disease.[24]
Wearing masks and performing COVID-19 screening before admission was no longer a standard procedure during 2022. At this point, children were not yet vaccinated in SA. This study aimed to investigate the prevalence of COVID-19 infection in children aged 0 - 12 years during the Omicron variant wave in the Free State Province of SA, as this age group of children was not vaccinated until July 2023.
Methods
Study design and study population
A total of 320 participants aged 0 - 12 years were included in the study. No admissions were done primarily for COVID-19. No patients were excluded. The study occurred in Bloemfontein, at the Pelonomi and Universitas Academic hospitals, from August 2022 to April 2023. Ethical permission was obtained from the University of the Free State before the commencement of the study (ref. no. UFS-HSD2022/1456/2609) in accordance with the Declaration of Helsinki. Informed consent was obtained from parents and legal guardians, and assent was obtained from children >7 years of age. This prospective cross-sectional study consisted of children admitted for various reasons; interestingly, none were admitted for suspected SARS-CoV-2 infection. Participants were randomly recruited from general paediatric wards and specialised wards such as paediatric oncology, cardiology and paediatric intensive care units at both hospitals. Their COVID-19 status was unknown during recruitment as SARS-CoV-2 screening upon admission was no longer standard practice during the period that the study was conducted. The children were categorised into different age groups (<1 month, 1 - 12 months, 13 - 60 months, 61 - 144 months). Babies who were born prematurely and were admitted for >1 month were categorised in the 1-month - 12-month group. Children were tested for SARS-CoV-2 antibodies following consent from their parents or caregivers. The nucleocapsid antibody testing technique (Roche) was used to measure the presence of antibodies (IgG/IgM rapid COVID test) in children. A drop of blood was obtained on admission during their routine daily blood withdrawal procedures. SARS-CoV-2 infection in children was confirmed by the presence of both immunoglobulin G (IgG) and immunoglobulin M (IgM) antibodies. The results include all children tested, with and without COVID-19 infection.
Data analysis
Data were analysed using a descriptive-quantitative approach. The frequencies and percentages for categorical data, mean, standard deviation, medians and percentiles for numerical data were analysed using SAS Software, version 9.4 (SAS Institute Inc., USA).
Results
This study occurred during the fourth wave of COVID-19 (Omicron), which started in December 2021. A total of 320 children were included in the study during the period from August 2022 to April 2023. The COVID-19 status of all recruited children according to the various age groups and parent/caregiver COVID-19 vaccination status is depicted in Table 1.
Overall, 46.8% (n=150) of the children had both IgM and IgG antibodies, indicating active or recent infection. The infection rate was higher (60%) in neonates (<1 month) than in other age groups. In infants aged 1 - 12 months, 21.3% were infected; in toddlers between 13 and 60 months, 11.3% were infected; while 8% of the children aged 61 - 72 months were infected (Table 1).
The COVID-19 vaccination history of all parents or caregivers included in the study showed that 61.3% had received the COVID-19 vaccination. Almost half (43.4%) of all children who tested positive for COVID-19 came from vaccinated parents or caregivers, while 41% came from unvaccinated ones.
In neonates, prematurity was the most common reason that children were admitted in both infected (32.2%) and uninfected groups (37.3%). However, slightly more infected children tended to have gastrointestinal tract (GIT) infection diagnoses (14.4%) than in the uninfected group (9.3%). Interestingly, almost double the number of neonates in the uninfected group were diagnosed with respiratory conditions compared with the infected group (24.0% v. 11.1%) (Table 2).
More of these infants were admitted owing to complications related to their prematurity (40.6%) in the infected group than the uninfected group (5.4%). These babies were born prematurely and had been admitted for >1 month. GIT conditions were accountable for only 16.5% of admissions in the infected group, compared with 43.2% in the uninfected group (Table 2).
In the 13 - 60-month cohort, 18.6% of the infected children were diagnosed with respiratory conditions, compared with 14.6% in the uninfected group. The most common respiratory conditions in the infected group were asthma, diffuse alveolar haemorrhage and recurrent chest infections. In the 61 - 144-month group, the most common diagnoses in infected children were related to the central nervous system (CNS) and respiratory conditions (33.3%). In contrast, in the uninfected group, the most common reason for admission was haematological and oncological conditions (52.6%) (Table 2). The most common CNS conditions were seizures, while respiratory conditions were asthma and lower respiratory tract infections.
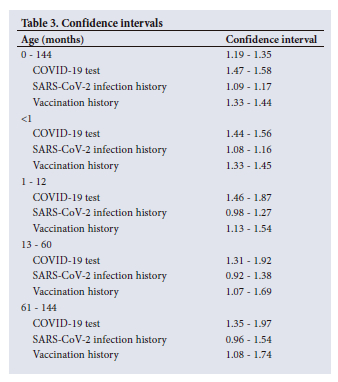
Confidence intervals were calculated to determine the relation between age in months, parents/caregiver infection history and parent/caregiver vaccination history. Table 3 shows that in all the different age groups, there is no relationship between SARS-CoV-2 infection history of parents/caregivers and the infection of the children. However, there is a relationship between the age of the children and their SARS-CoV-2 infection, as well as the vaccination history of the parents/caregivers and infection in the children (Table 3).
Discussion
This study aimed to investigate the prevalence of SARS-CoV-2 infection in unvaccinated children during the Omicron variant (fourth wave). Results show that just under half of the children included in the study had both IgM and IgG antibodies present, indicating current or recent active infection. However, they were admitted for other conditions. All positive cases found in the study were incidental, as none of these children were tested for SARS-CoV-2 upon admission, or admitted due to infection.
The highest infection rates were noted in neonates <1 month (60%), followed by infants aged 1 - 12 months and toddlers, aged 13 - 60 months, and the lowest infection rates were observed in children between 61 - 144 months. These results differ from a multicentre observational study conducted in SA in the Tshwane district that investigated clinical profiles of all children infected with SARS-CoV-2 (6 287 children <19 years of age) during the fourth wave (Omicron variant). Of the infected children, 14% were aged 0 - 4 years, 20% were aged 5 - 9 years, 32% were aged 10 - 14 years, and 34% were aged 15 - 19 years.[15] The infection rates increased with age while in our study, the rates decreased with age. Furthermore, of the three COVID-19 waves, the highest rates of infection were noted in the fourth wave (Omicron).[22]
According to the Centers for Disease Control and Prevention update on COVID-19 in children and adolescents <18 years, 11.8% of children were infected during the fourth wave of COVID-19 in January 2022 in SA.[25] Furthermore, a paediatric registry in the USA reported that 24% of children aged between 5 and 11 years tested positive for SARS-CoV-2.[16]
The infection rate of the neonates in the current study is also high compared with a study published in Iran, which had an infection rate of 6.1% in neonates.[26] A study in the UK reported that the incidence of SARS-CoV-2 was 5.6 per 10 000 live births. Of these neonates, 42% had severe SARS-CoV-2 infection.[27] Our results may have had a higher infection rate in neonates because the majority of the study population comprised neonates (51.6%). The presence of SARS-CoV-2 antibodies in circulation determined infection.
The presence of both antibodies represents a recent infection, as research shows that the IgM antibody is detected 4 days following infection and reaches its peak on the 20th day, after which it drops. Conversely, IgG rises 7 days after infection until the 25th day, but remains detectable 4 weeks after infection.[28] IgM is the first line of defence against viruses, while IgG maintains long-term immunity after viral infection.[29] A study conducted in China in 2020 showed that in patients with SARS-CoV-2 infection, IgM antibodies appear early and peak before IgG. IgM was reported to linger in the system for 3 weeks, while IgG antibodies remained in the system longer.[28]
Children included in the study were admitted for reasons other than suspected SARS-CoV-2 infection, and none were tested before admission; therefore, their infection status was unknown upon admission. In December 2021, the paediatric population of hospital admissions due to SARS-CoV-2 infection was ~18% (462/2 550) in the Tshwane district.[15] Of these admissions, the most common conditions associated with positive antibodies were seizures (20%), acute gastroenteritis (20%), respiratory infections (14%) and bronchopneumonia (15%). The current study's admission diagnoses differed among the different age groups. In these, the reasons for admission were prematurity (28%), GIT conditions (12.6%), respiratory conditions (12%) and CNS conditions (10%). However, whether the admission diagnoses were due to SARS-CoV-2 infection in these children is unknown, as this study was only focused on infection rates. This study adds new knowledge because most SA studies underreport neonatal data.[1,2]
Another study recruited 97 SARS-CoV-2-positive children admitted to a hospital in India.[30] Some of the children were admitted due to COVID-19, while others were admitted for other reasons but tested positive for SARS-CoV-2 upon admission. The study population was aged 1 - 18 years. The total sample size of the study was lower (n=97) than the current study (n=320). The study found that 30.93% of the children were asymptomatic, while 68.04% were symptomatic. These results differ from the current study as all 46.8% (n=150/320) SARS-CoV-2 infected children were asymptomatic, indicating mild disease.
Studies show that pregnant women infected with SARS-CoV-2 are at a higher risk of preterm deliveries.[31-34] Another multicentre study conducted in Saudi Arabia described the clinical outcomes of mothers infected with SARS-CoV-2 and their neonatal outcomes. The authors found that out of 204 neonates, 15.5% (n=31) were born prematurely.[35] In the current study, 32.2% of the SARS-CoV-2-infected babies were premature. These babies may have been infected via vertical transmission before birth, or the infection may have been acquired in hospital. There are different ways in which vertical transmission can occur, leading to preterm birth.[36] SARS-CoV-2 is transmitted through faecal contamination of the birth canal, which can infect the neonate during labour. Vertical transmission via the ACE2 receptors found in the placenta can also affect gaseous exchange and lead to other complications, such as intrauterine death and perinatal asphyxia.[35] However, the likelihood of transmission to the fetus is minimal due to the presence of the placental barrier.[37] Some researchers state that vertical transmission to the fetus might be possible through other receptors (dipeptidyl peptidase-4 inhibitor (DDP4) and cluster of differentiation (CD147) and proteases (Furin).[38,39]
In a study by Allotey et al.,[32] the rate of preterm births in women infected with COVID-19 was 17%, which is lower than in the current study. Furthermore, in the current study, mothers were not tested for SARS-CoV-2; therefore, their SARS-CoV-2 status was unknown. However, their maternal COVID-19 vaccination history was collected, and 61.8% of the mothers with premature deliveries were vaccinated. The rates of COVID-19-vaccinated adults in the Tshwane district were lower than in this study (32%).[15] Our results align with reports published by the SA National Department of Health, which found that 65 - 80% of the adult population had a positive result due to a previous infection or COVID-19 vaccination.[40] A study conducted in Israel from April 2020 to March 2021 revealed that antenatal vaccination with the Pfizer vaccine demonstrated elevated IgG titers, and these antibodies were transferred to the fetus through the placenta.[41] Maternal transfer of anti-SARS-CoV-2 antibodies was found in neonatal circulation 2 weeks after the first dose, indicating neonatal immunity during pregnancy. Equally, another study showed that the transfer of SARS-CoV-2 IgG and IgM was possible via breastmilk. Notably, both antibodies in neonatal circulation may indicate neonatal immunity more than an active infection.[18] However, in the current study, information on breastfeeding was not collected to confirm this finding.
According to Beharier et al.,[41] it is difficult to confirm whether antibodies found in neonates following delivery may be due to the placental transfer of a seropositive mother or to immune response following the COVID-19 vaccine. The study reported no significant difference between maternal and neonatal antibody titers in vaccinated mothers compared with previously infected mothers who had recently recovered.
Strengths
As far as we are aware, this is the first prevalence study of SARS-CoV-2 infection in children in SA. All studies published on COVID-19 in children include all children with known SARS-CoV-2 infection, and they all report on symptoms and outcome. The current study, however, included all admissions regardless of SARS-CoV-2 infection status. None of the children included had a known COVID-19 status.
Limitations
Our study included all children admitted to a hospital ward between 0 and 12 years old. Our results showed the highest SARS-CoV-2 infection rate in the <1 month population, which is often under-reported. However, the mothers of the neonates were not tested for SARS-CoV-2 infection on admission. Therefore, the study cannot confirm whether the antibodies detected were due to vertical transmission. Furthermore, COVID-19 vaccination status was obtained verbally, and the timing of the vaccination was not recorded. Feeding practices, such as breastfeeding, were not recorded. The results may be under-reported because the study only included inpatients, and the majority of the children were neonates. This may also introduce some bias. There is no evidence that the reasons for admission were related to COVID-19.
Conclusion
A substantial number of hospitalised children were infected with SARS-CoV-2. All infected children did not have COVID-19-specific symptoms and were admitted for conditions entirely unrelated to COVID-19. This may be due to the Omicron variant, which is highly infectious, less virulent and associated with mild disease. Noting the high infection rates in the study, vaccination of all children for COVID-19 is recommended, as vaccinating parents alone does not protect the children from infection.
Data availability. The data that support the findings of this study are available on request from the corresponding author. The data are not publicly available owing to ethical restrictions.
Declaration. None.
Acknowledgements. We would like to acknowledge the Department of Paediatrics and Child Health at the University of the Free State, with special thanks to Dr Ahmad Jassen and Dr Dorothy Russell for their tremendous support of the project. We would also like to thank the staff members from the two hospitals for their co-operation and patience during the study. Lastly, we would like to give sincere thanks to Dr Omololu Aluko for his assistance with statistical analysis of the data.
Author contributions. OPK: conceptualisation, investigations, data collection, writing and editing. BP: results interpretation, writing and editing. SB: results interpretations, reading and editing. NT: conceptualisation, reading and editing.
Funding. This study was funded by the National Research Foundation (NRF). Opinions expressed and conclusions arrived at are those of the author and not necessarily attributed to NRF (SA, grant no. 149140).
Conflicts of interest. None.
References
1. Wang H, Paulson KR, Pease SA, et al. Estimating excess mortality due to the COVID-19 pandemic: A systematic analysis of COVID-19-related mortality, 2020 - 21. Lancet 2022;399(10334):1513-1536. https://doi.org/10.1016/s0140-6736(21)02796-3 [ Links ]
2. World Health Organization. COVID-19 weekly epidemiological update 2023. https://www.who.int/publications/m/item/weekly-epidemiological-update-on-covid-19---18-may-2023 (accessed 22 November 2023). [ Links ]
3. Wolter N, Jassat W, Walaza S, et al. Early assessment of the clinical severity of the SARS-CoV-2 omicron variant in South Africa: A data linkage study. Lancet 2022;399(10323):437-446. https://doi.org/10.1016/s0140-6736(22)00017-4 [ Links ]
4. Rubin R. COVID-19 vaccines vs variants - determining how much immunity is enough. JAMA 2021;325(13):1241-1243. https://doi.org/10.1001/jama.2021.3370 [ Links ]
5. Tegally H, Wilkinson E, Giovanetti M, et al. Detection of a SARS-CoV-2 variant of concern in South Africa. Nature 2021;592(7854):438-443. https://doi.org/10.1038/s41586-021-03402-9 [ Links ]
6. South Africa COVID-19 Modelling Consortium. COVID-19 modelling update: Considerations for a potential fourth wave. Johannesburg: National Institute for Communicable Diseases, 2021. https://www.nicd.ac.za/wp-content/uploads/2021/11/SACMC-Fourth-wave-report-17112021-final.pdf (accessed 27 November 2024). [ Links ]
7. Rostami A, Sepidarkish M, Leeflang MM, et al. SARS-CoV-2 seroprevalence worldwide: A systematic review and meta-analysis. Clin Microbiol Infect 2021;27(3):331-340. https://doi.org/10.1016/j.cmi.2020.10.020 [ Links ]
8. Garrett N, Tapley A, Andriesen J, et al. High asymptomatic carriage with the omicron variant in South Africa. Clin Infect Dis 2022;75(1):e289-e292. https://doi.org/10.1093/cid/ciac237 [ Links ]
9. Bittmann S, Luchter E, Moschuring-Alieva E, Bittmann L, Villalon G. What is new with omicron variant of SARS-CoV-2 in children? J Clin Med Res 2022;14(2):108-109. https://doi.org.10.14740/jocmr4671 [ Links ]
10. Marks KJ. Hospitalisation of infants and children aged 0 - 4 years with laboratory-confirmed COVID-19. COVID-NET, 14 states, March 2020 - February 2022. Morbidity and Mortality Weekly Report 2022;71(11);429-436. https://doi.org/10.15585/mmwr.mm7111e2 [ Links ]
11. Streinu-Cercel A, Sãndulescu O, Miron VD, et al. Undetected Omicron transmission in Romania -report of the first detected case of locally acquired omicron infection and complete epidemiological investigation. Diagnostics 2022;12(2):348. https://doi.org/10.3390/diagnostics12020348 [ Links ]
12. Shen N, Wu Y-F, Chen Y-W, et al. Clinical characteristics of pediatric cases infected with the SARS-CoV-2 Omicron variant in a tertiary children's medical center in Shanghai, China. World J Pediatrics 2023;19(1):87-95. https://doi.org/10.1007/s12519-022-00621-6 [ Links ]
13. Han MJ, Heo JH, Hwang JS, Jang Y-T, Lee M, Kim SJ. Incidence of febrile seizures in children with COVID-19. J Clin Med 2023;12(3):1076. https://doi.org/10.3390/jcm12031076 [ Links ]
14. Iijima H, Kubota M, Ogimi C. Change in seizure incidence in febrile children with COVID-19 in the era of omicron variant of concern. J Pediatric Infect Dis Soc 2022;11(11):514-517. https://doi.org/10.1093/jpids/piac085 [ Links ]
15. Cloete J, Kruger A, Masha M, et al. Paediatric hospitalisations due to COVID-19 during the first SARS-CoV-2 omicron (B. 1.1. 529) variant wave in South Africa: A multicentre observational study. Lancet Child Adolesc Health 2022;6(5):294-302. https://doi.org/10.1016/S2352-4642(22)00027-X. [ Links ]
16. Pediatric COVID-19 Case Registry. Pediatric COVID-19 US Registry. Collaboration is our strategy. Children are our priority. PCCR, 2022. https://www.pedscovid19registry.com/ (accessed 23 July 2024). [ Links ]
17. Berni Canani R, Comegna M, Paparo L, et al. Age-related differences in the expression of most relevant mediators of SARS-CoV-2 infection in human respiratory and gastrointestinal tract. Front Pediatr 2021:28(9):697390. https://doi.org/10.3389/fped.2021.697390.eCollection_2021 [ Links ]
18. Bunyavanich S, Do A, Vicencio A. Nasal gene expression of angiotensin-converting enzyme 2 in children and adults. JAMA 2020;323(23):2427-2429. https://doi.org/10.1001/jama.2020.8707 [ Links ]
19. Patel AB, Verma A. Nasal ACE2 levels and COVID-19 in children. JAMA 2020;323(23):2386-2387. https://doi.org/10.1001/jama.2020.8946 [ Links ]
20. She J, Liu L, Liu W. COVID-19 epidemic: Disease characteristics in children. J Med Virol 2020;92(7):747-54. https://doi.org/10.1002/jmv.25807 [ Links ]
21. Colonna C, Monzani NA, Rocchi A, Gianotti R, Boggio F, Gelmetti C. Chilblain-like lesions in children following suspected COVID-19 infection. Pediatr Dermatol 2020;37(3):437-440. https://doi.org/10.1111/pde.14210 [ Links ]
22. Chou J, Thomas PG, Randolph AG. Immunology of SARS-CoV-2 infection in children. Nature Immunol 2022:23;177-185. https://doi.org/10.1038/s41590-021-01123-9 [ Links ]
23. Chung E, Chow EJ, Wilcox NC, et al. Comparison of symptoms and RNA levels in children and adults with SARS-CoV-2 infection in the community setting. JAMA Pediatr 2021;175(10):e212025-e. https://doi.org/10.1001/jamapediatrics.2021.2025 [ Links ]
24. Pierce CA, Preston-Hurlburt P, Dai Y, et al. Immune responses to SARS-CoV-2 infection in hospitalised pediatric and adult patients. Sci Translat Med 2020;12(564):eabd5487. https://doi.org/10.1126/scitranslmed.abd5487 [ Links ]
25. Coronavirus Disease (COVID-19) Pandemic. Update of COVID-19 in children and adolescents <18 years, South Africa, 1 March 2020 - 15 January 2022. Pretoria: National Institute for Communicable Diseases, 2022. https://www.nicd.ac.za/wp-content/uploads/2022/01/Update-of-COVID-19-in-children-and-adolescents-%E2%89%A418.pdf (accessed 22 July 2024). [ Links ]
26. Babaei R, Bokharaei-Salim F, Khanaliha K, et al. Prevalence of SARS-CoV-2 infection in neonates born to mothers or relatives with COVID-19. BMC Infect Dis 2022;22(1):730. https://doi.org/10.1186/s12879-022-07688-6 [ Links ]
27. Gale C, Quigley MA, Placzek A, et al. Characteristics and outcomes of neonatal SARS-CoV-2 infection in the UK: A prospective national cohort study using active surveillance. Lancet Child Adolescent Health 2021;5(2):113-121. https://doi.org/10.1016/S2352-4642(20)30342-4 [ Links ]
28. Liu X, Wang J, Xu X, Liao G, Chen Y, Hu C-H. Patterns of IgG and IgM antibody response in COVID-19 patients. Emerg Microbes Infect 2020;9(1):1269-1274. https://doi.org/10.1080/22221751.2020.1773324 [ Links ]
29. Li G, Chen X, Xu A. Profile of specific antibodies to the SARS-associated coronavirus. N Engl J Med 2003;349(5):508-509. https://doi.org/10.1056/NEJM200307313490520 [ Links ]
30. Singh P, Singh SN, Singh K, et al. Epidemiological and clinical features among COVID-19-positive children in Eastern Uttar Pradesh (India): An observational study. Pediatr Infect Dis 2024;6(3):73-79. https://doi.org/10.5005/jp-journals-10081-1431 [ Links ]
31. Zambrano LD, Ellington S, Strid P, et al. Update: characteristics of symptomatic women of reproductive age with laboratory-confirmed SARS-CoV-2 infection by pregnancy status - United States, January 22 - October 3, 2020. Morbidity and Mortality Weekly Report 2020;69(44):1641. https://doi.org/10.15585/mmwr.mm6944e3 [ Links ]
32. Allotey J, Fernandez S, Bonet M, et al. Clinical manifestations, risk factors, and maternal and perinatal outcomes of coronavirus disease 2019 in pregnancy: Living systematic review and meta-analysis. BMJ 2020;1:370:m3320. https://doi.org/10.1136/bmj.m3320 [ Links ]
33. Metz TD, Clifton RG, Hughes BL, et al. Disease severity and perinatal outcomes of pregnant patients with coronavirus disease 2019 (COVID-19). Obstet Gynecol 2021;137(4):571-580. https://doi.org/10.1097/AOG.0000000000004339 [ Links ]
34. Villar J, Ariff S, Gunier RB, et al. Maternal and neonatal morbidity and mortality among pregnant women with and without COVID-19 infection: The INTERCOVID multinational cohort study. JAMA Pediatr 2021;175(8):817-826. https://doi.org/10.1001/jamapediatrics.2021.1050 [ Links ]
35. Al-Matary A, Almatari F, Al-Matary M, et al. Clinical outcomes of maternal and neonate with COVID-19 infection - multicenter study in Saudi Arabia. J Infect Public Health 2021;14(6):702-708. https://doi.org/10.1016/j.jiph.2021.03.013 [ Links ]
36. Ryan L, Plötz FB, van den Hoogen A, et al. Neonates and COVID-19: State of the art: Neonatal sepsis series. Pediatr Res 2022;91(2):432-439. https://doi.org/10.1038/s41390-021-01875-y [ Links ]
37. Peng Z, Zhang J, Shi Y, Yi M. Research progress in vertical transmission of SARS-CoV-2 among infants born to mothers with COVID-19. Future Virol 2022;17(4):211-214. https://doi.org/10.2217/fvl-2021-0213 [ Links ]
38. Ulrich H, Pillat MM. CD147 as a target for COVID-19 treatment: Suggested effects of azithromycin and stem cell engagement. Stem Cell Rev Rep 2020;16(3):434-440. https://doi.org/10.1007/s12015-020-09976-7 [ Links ]
39. Li Y, Zhang Z, Yang L, et al. The MERS-CoV receptor DPP4 as a candidate binding target of the SARS-CoV-2 spike. Iscience 2020;23(6):101160. https://doi.org/10.1016/j.isci.2020.101160 [ Links ]
40. National Department of Health, South Africa. COVID-19 public dashboard. Pretoria: NDoH, 2021. https://www.nicd.ac.za/diseases-a-z-index/disease-index-covid-19/surveillance-reports/ (accessed 12 August 2024). [ Links ]
41. Beharier O, Mayo RP, Raz T, et al. Efficient maternal to neonatal transfer of antibodies against SARS-CoV-2 and BNT162b2 mRNA COVID-19 vaccine. J Clin Investigation 2021; 131(13):e150319. https://doi.org/10.1172/JCI150319 [ Links ]
 Correspondence:
Correspondence:
O Khaliq
khaliqop@ufs.ac.za
Received 8 October 2024
Accepted 14 January 2025













