Services on Demand
Journal
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Animal Science
On-line version ISSN 2221-4062Print version ISSN 0375-1589
S. Afr. j. anim. sci. vol.55 n.8 Pretoria 2025
https://doi.org/10.17159/sajas.v55i8.02
RESEARCH ARTICLES
Heritability coefficients of growth traits of rainbow trout from a national selection programme
V. GolubovićI, #; M. StankovićI; D. VukojevićI; D. StanojevićI; R. DjedovićI; S. MarićII; Z. MarkovićI
IInstitute of Zootechnics, Faculty of Agriculture, University of Belgrade, Serbia
IIInstitute of Zoology, Faculty of Biology, University of Belgrade, Serbia
ABSTRACT
In order to create a selection programme aimed at improving the production characteristics of rainbow trout, broodstocks from eight fish farms in the Republic of Serbia were collected in 2010. The spawning of broodstocks, hatching of eggs, and rearing of offspring from different families of known parentage, until marking at five months of age, were carried out in individual tanks under strictly controlled conditions. After tagging, the fish were reared in a flow-through system on a commercial fish farm, where their growth was further monitored. The study of the phenotypic variability and heritability coefficients of the broodstock was carried out on a sample of 6565 individuals. The body weights and total lengths of the individuals were measured at five, 12, 24, and 36 months of age. Factors such as the year of spawning and sex proved to be highly statistically significant for the observed growth traits, while the heritability values of the analysed traits were moderate to high (0.22-0.68). The estimated heritability values provide an excellent basis for the continuation of the selective breeding programme, which will ultimately lead to the improvement of salmonid aquaculture and the profitability of rainbow trout production in the Republic of Serbia.
Keywords: body weight, genetic variability, total length, trout
Introduction
The rainbow trout (Oncorhynchus mykiss; Walbaum, 1792) is a salmonid species that is successfully bred all over the world. The success of fish farming depends on the environmental conditions provided by a well-designed production system, the use of a well-balanced diet, the implementation of health protection systems, and the existence of selection programmes to improve economically important traits (Gjedrem et al., 2012). The evaluation of genetic parameters for the trait of heritability and knowledge of the genetic correlations between traits and the interactions between genotypes and the environment, are prerequisites for the successful implementation of breeding programmes (Gjedrem, 2010; Yáñez et al., 2015). A high growth rate is one of the most important traits in trout breeding; however, a high growth rate cannot be maintained under unfavourable farming conditions (Sae-Lim et al., 2013, 2015). It is estimated that only 10% of global aquaculture utilises genetically improved fish lines (Gjedrem et al., 2012). Nonetheless, the genetic gain achieved in rainbow trout selection is estimated to be around 14% (10%-20%) per generation, which is significantly more than for other farmed animals (Gjedrem, 2010; Hung et al., 2013; Leeds et al., 2016).
Studies on improving the production of cultured species of organisms in aquaculture most commonly investigate genetic programmes based on body weight (Gunadi et al., 2021). Growth traits are good selection criteria because of their economic importance, easy measurability, broad coefficients of variation, and moderate to high heritability values (Yáñez et al., 2014; Houston et al., 2020). In the genetic improvement of body weight, the body weight traits of individuals at 10 months of age are typically included in the selection programme (Leeds et al., 2016), as are the coefficients of thermal growth of individuals at 10 to 13 months of age (Robledo et al., 2019). In addition to body weight, morphological characteristics of individual growth, such as total length, height, and width, are also the result of phenotypic and genetic correlations (Sae-Lim et al., 2013). Furthermore, an increase in body weight can be achieved by an increase in the width and height of an individual, rather than an increase in that individual's total length (Nguyen, 2016).
The growth of fish is characterised by a positive allometry of the muscles in relation to the organs (Vo et al., 2016). In contrast to mammals and birds, fish exhibit unlimited growth, with the most dynamic growth occurring in the first phases of life. A rapid growth rate in farmed fish is desirable as it shortens the growth period until the desired body weight is reached and reduces water consumption, which has an overall positive effect on production costs (Houston et al., 2020). For this reason, growth performance is particularly important for rainbow trout producers, and growth rate is one of the most crucial traits that should be improved through selection programmes (Sae-Lim et al., 2015). Improving growth performance through various selection programmes in rainbow trout, as in other farmed salmonid species, is both efficient and economically justified (Leeds et al., 2016; Janssen et al., 2016). The selection of parents to produce offspring for aquaculture breeding has the positive effect of enhancing the desired traits in the progeny (Gunadi et al., 2021).
The growth rate of male fish is faster than that of females, because males do not need to prepare for yolk formation in oocytes or for oocyte maturation (Gunadi et al., 2021). In addition to the effect of sex on fish growth, growth differences can also result from social interactions between fish (competition for food and space), and variations in the living environment (water) (Aksungur et al., 2007; Johnsson et al., 2014). Consequently, the growth of salmonids is a complex physiological process that depends on both genetic factors and the aquatic environment, which means that one genotype is not necessarily superior in all environments (Robledo et al., 2019). In salmonids, the average heritability of body weight per year is 0.24 (0.10-0.52) (Kause et al., 2007; Silverstein et al., 2009; Hu et al., 2013). Such moderate to high heritability values indicate the possibility of further genetic improvement of trout (Sae-Lim et al., 2013).
The aim of this work was to estimate the additive genetic variance and heritability of the physical developmental characteristics of rainbow trout. The results obtained will serve as a basis for the development of breeding programmes to improve growth traits through selection, in order to increase the productivity and economic profitability of farming this species.
Materials and methods
The experiments were conducted in accordance with the ethical conditions approved by the Serbian Ministry of Agriculture, Forestry and Water Management (authorisation number: 0001986332024 14841 002 000 000 001).
Sexually mature individuals of three to four years of age were used for the selection programme, with the aim of improving the production traits of rainbow trout families of known parentage. To start the selection programme, fish were collected from six fish farms in different locations in the Republic of Serbia in 2010 (Table 1) and brought to the Mali Dunav Centre for Fisheries and Applied Hydrobiology at the Radmilovac experimental farm of the Faculty of Agriculture of the University of Belgrade, where the selection programme was carried out. The initial criterion for the selection of mature individuals was that no fish from other farms had been introduced into the fish farms from which the individuals were taken in the last 15 years.

After broodstock collection, a crossing scheme was drawn up, on the basis of which spawning and crossing was carried out. The resultant eggs were then incubated according to standard procedures (Markovic & Mitrovic-Tutundzic, 2003). In the first crossing scheme, males from one farm were crossed with females from another farm, taking care to avoid inbreeding. In the following generations, similar criteria were applied. Following these rules, the individuals with the best production within each family were selected to serve as future parents. To implement the selection programme, fish spawning, family formation, and body performance measurements were carried out every year from 2011 to 2016. The selection programme for generation G0 and generation G1 was carried out every year under the same experimental conditions and using the same methodology. Generation G0 was formed from the spawns of the years 2011, 2012, and 2013, and generation G1 from the spawns of the years 2014, 2015, and 2016.
The spawning of the fish and the formation of families was carried out on one day each year. The eggs of all families were incubated in separate incubators until the larvae began to swim. The larvae hatched at 30-32 days post fertilisation, after incubation at a water temperature of 9-11 °C, and started swimming at 15 days post hatching. A few days after the larvae began to swim, 100 larvae were randomly selected from each family and transferred to individual tanks with a volume of approximately 120 L, where their rearing continued. Rearing in tanks meant rearing the fish under controlled and similar environmental conditions (water temperature: 9-11 °C, dissolved oxygen: 7-9 ml/L).
In order to monitor the production traits of each family, the offspring were marked by injecting passive integrated transponder (PIT) tags (Norway) into the abdominal cavities of the fish at the age of five months, i.e. when they had reached a size that allowed the injection of a PIT tag (over 1.75 g). Of the initially-separated 100 individuals per family, up to 50 of the largest specimens were selected for tagging. The largest fish were identified by measuring the body weights of 100 fish from each tank/family. It was assumed that the fish had grown sufficiently by this time to inject the chip without affecting the performance of other vital functions. Each tagged individual was given an identification number representing a unique combination of 10 symbols (letters and numbers), which was entered into a database (BKTAGCOM, Norway) together with other production traits. Prior to tagging, the trout were anaesthetised in a clove solution (produced by Probotanika) to reduce stress and to facilitate the measurement of body weight. Body weight was measured using a precision scale (RADWAG THB-600, max 600 g, d = 0.01 g, Poland), while the total length was measured using an ichthyometer.
A few days after tagging, the young trout were transported from the Mali Dunav Centre for Fisheries and Applied Hydrobiology to the RM Stubica fish farm (Sisevac, Republic of Serbia; 43.956405, 21.585439), where they were further reared and where additional production indicators were monitored. In order to keep the genetic variability as high as possible, only mild selection was carried out during further rearing. For the first seven months, all individuals, regardless of whether they came from different families, were reared together in a small pool (dimensions 3x0.70x0.50 m). At one year of age, the trout were moved to a larger pool (25x5x1 m), where they were reared further.
The trout were fed a high-quality protein feed produced by Skreting (crude protein: 44.50%, crude fat: 22%, crude fibre: 3%, crude ash: 7.50%, phosphorus: 1.20%, sodium: 0.30%, calcium: 1.90%), following the manufacturer's recommendations. At the end of each year (in December), the body weights and total lengths of the fish were measured, and the values recorded in the database to continue the selection programme and identify the smallest individuals based on body performance (body weight and total length). At two years of age, in addition to the basic data mentioned above, information on the sex of each fish and on sexual maturity, i.e. readiness to spawn, was entered into the database. This monitoring continued the following year.
Between 2011 and 2016, a total of 6565 fish from 195 families, formed in the G0 and G1 generations, were reared. Families of the G0 generation were formed in 2011, 2012, and 2013, and families of the G1 generation were formed from the G0 fish in 2014, 2015, and 2016. The formation of the families was based on the principle of selecting the best individuals from each family of known parentage, observing the rule that no inbreeding should occur. When preparing the crossing scheme, families with a higher average body weight were given priority for participation in the spawning season. The families formed had no common ancestors.
The study included the measurement of body weight and total length at five, 12, 24, and 36 months of age. During the implementation of the selection programme, the results of which are presented in this paper, the number of individuals decreased from year to year, so that the number of individuals monitored was 6565 at five months of age and 1196 at 36 months of age. At each measurement, individuals were eliminated according to the criterion of obtaining the individuals with the best phenotypic and productive traits for further breeding, by excluding individuals with inferior traits from the selection programme.
The P-value was used to determine whether the different factors investigated (year of spawning, sex, and their interaction) influenced the studied characteristics. The basic statistical indicators of phenotypic expression and variability of the body weight and total body length traits were calculated using standard statistical procedures, based on the PROC MEANS procedure of the SAS software package, version 9.4, 2013 (SAS Institute Inc., Cary, North Carolina, USA).
Data preparation, or coding, for the calculation of genetic and phenotypic variances was performed using the PEST program package (Groeneveld et al., 1990). The variance components were calculated using the restricted maximum likelihood procedure within the program package VCE v6 (Groeneveld et al., 2010), using the following mixed model:
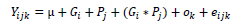
where:
Yijk = the phenotypic expression of the examined trait,
μ= the general population average,
Gi = the fixed effect of the ith year of spawning,
Pj = the fixed effect of the jth sex of the individual fish,
Gi*Pj = interaction factor,
Ok = the random effect of the kth father of the individual fish, and eijk = random error.
The calculated variance components were used to calculate the heritabilities of the investigated traits using the following formula:
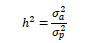
where:
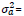 = the additive genetic variance, and
= the additive genetic variance, and
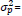 = the total phenotypic variance, consisting of the additive genetic variance, the dominance variance, the epistatic variance, and the error variance
= the total phenotypic variance, consisting of the additive genetic variance, the dominance variance, the epistatic variance, and the error variance 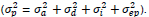
Based on the values calculated for the variance of the father, the additive variance, the environmental variance, and the phenotypic variance, the heritability coefficients (heritabilities) were determined for the investigated traits. The heritability coefficient is a genetic parameter of the population and does not refer to individuals. By calculating the heritability value, information is obtained on whether and to what extent genetic variability exists in the population. The heritability value determines the possibility of genetic improvement through selection.
Results and discussion
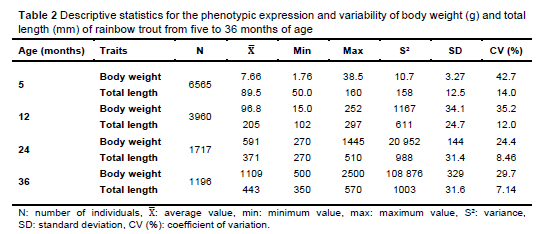
Table 2 contains the average values and variability measures of the investigated traits. The comparison of the initial and final values of the expressed characteristics showed that the average body weight of the five-month-old fish was 7.66 g and that of the 36-month-old fish was 1109 g. As for the average total length, the fish at five months of age had an average total length of 89.5 mm, while the average total length of the fish at 36 months of age was 443 mm. The coefficient of variation for body weight was 42.7% at five months of age and 29.7% at 36 months of age, while the coefficient of variation for total length was 14.0% at five months of age and 7.14% at 36 months of age. The body weight and body length values confirmed a clear pattern of positive growth dynamics within the observed rainbow trout population, which is consistent with results published for other salmonid species (Janampa-Sarmiento et al., 2020). The observed variance, standard deviation, and coefficient of variation values in G0 were high (Table 2), indicating a good potential for selection and genetic improvement. Yoshida et al. (2019) came to the same conclusion in their study. The high coefficients of variation and ranges of values found for body weight and body length are typical of trout, salmon (Sae-Lim et al., 2016), and tilapia (Yoshida et al., 2019). Continuous monitoring of phenotypic parameters such as the standard deviation and coefficient of variation is necessary to monitor the selection response and adjust the breeding strategy to achieve further production progress. Improving growth performance while maintaining genetic diversity ensures the long-term sustainability of the selection programme (D'Ambrosio et al., 2019; Nœve et al., 2022).
The effects of the investigated factors (year of spawning and sex) are shown in Table 3.
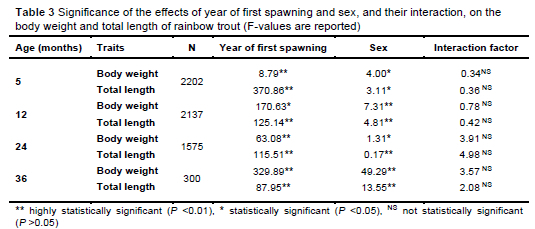
From the results in Table 3, it can be seen that the effects of the factors studied (year of first spawning and sex) on the observed characteristics were significant. Thus, year of first spawning had a highly statistically significant effect on the body weight and total length measured at five months, 24 months, and 36 months, and on the total length at 12 months, and had a statistically significant effect on the body weight at 12 months after spawning. Sex had a statistically significant effect on body weight measured at 5 months and 24 months of age, and on total length measured at five months of age. Sex had a highly statistically significant effect on body weight and total length at 12 months of age, total length at 24 months of age, and body weight and total length at 36 months of age. The interaction of the two factors had no statistically significant effects on either the body weights or total lengths of the individuals studied, suggesting that these traits can be considered independently and improved by selection. The influence of spawning year and sex on growth has been demonstrated in other studies (EL-Bab et al., 2024).
As can be seen in Tables 2 and 3, the number of individuals in generations G0 and G1 decreased significantly from year to year, because of the selection process. Through selection based on phenotypic and productive indicators, the best individuals were retained and further cultivated, while those with poor production results were excluded from the selection programme. This practice maximises genetic progress per generation and improves production efficiency. A decrease in the number of individuals with increasing age is to be expected, and is a normal process in selection programmes, as certain families or individuals are excluded from reproduction for selection reasons (Gjedrem et al., 2012). The number of individuals listed in Table 3 also depends on the number of fish of known sex at a given age. The different sizes of the observed populations, depending on the age of the individuals and the known sex, is a common phenomenon when it comes to research in the field of aquaculture. The incomplete determination of sex in the juvenile fish stages often leads to the exclusion of individuals from analysis, because of the unknown sex of the individual (Hamzah et al., 2014). This highlights the importance of using reliable methods to determine sex, especially in selection programmes. By controlling these factors as part of a selection programme, breeders can optimise growth performance and maintain the uniformity of production traits (Gjedrem, 2012).
From the variance and heritability values obtained (Table 4), it can be concluded that the heritability was moderate to high for the observed traits of the fish measured at five, 12, 24, and 36 months of age. In this case, the heritability indicates a strong link between the phenotype and the genotype of the individuals, which allows further selection based on phenotype in the form of individual selection (Sae-Lim et al., 2013).
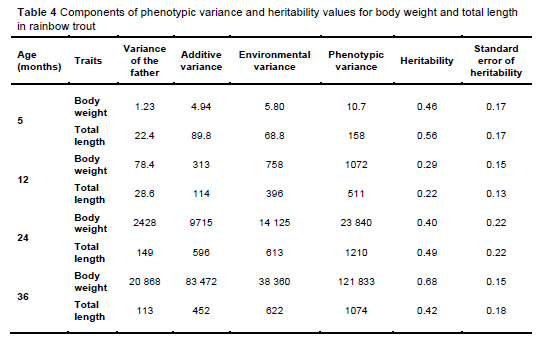
High heritability values are evidence that heterozygosity prevails in the observed population (Serbezov et al., 2010; Houston et al., 2020). Moderate to high heritability values indicate that both genetic and environmental factors have a significant influence on the body weight and total length of rainbow trout. Traits with high heritability due to a low environmental influence and a high genetic effect significantly influence the results of selection (Gunadi et al., 2021). The heritability values for body weight and total length found in this study are approximately the same, which is to be expected, considering that body weight and total length are highly correlated, as reported by Hu et al. (2013). The heritability values determined in this study for the growth traits are similar to the results published by Kause et al. (2002, 2007), Silverstein et al. (2009), Hu et al. (2013), Thodesen et al. (2013), and Robledo et al. (2019), who reported heritability values between 0.10 and 0.52. The strong positive correlation between body weight and body length is a crucial point, as it enables the concurrent improvement of both traits and maximises the genetic gain per generation (Robledo et al., 2019). Maintaining moderate to high heritability for body weight traits is possible if the population is large enough to maintain genetic variability and avoid inbreeding depression (Houston et al., 2020).
Considering that rainbow trout reach market size at 12 to 18 months of age, the genetic improvement of growth traits is particularly important within this age range (Jobling, 2003). The body weights and total lengths of 12-month-old fish were monitored in this study, to determine the effects of the selection performed on these traits (Figure 1).
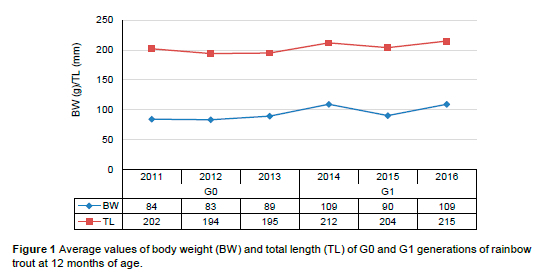
A comparative analysis of the growth performances of the rainbow trout from generations G0 (2011-2013) and G1 (2014-2016) clearly shows the differences between the fish at 12 months of age. The increase in average body weight and total length values in the G1 generation compared to the G0 generation confirms the value of implementing a selection programme (Leeds et al., 2016; Gunadi et al., 2021). The improvement in body growth, and especially body weight, is a good indicator of the success of the selective breeding of rainbow trout, especially considering the increasing need to achieve the most economical production possible (Silverstein et al., 2009), while maintaining the quality of farmed fish meat.
When examining the production traits by sex of all the observed individuals from both generations (G0 and G1), differences in the growth traits of males and females were observed at 12 months of age (Figure 2).
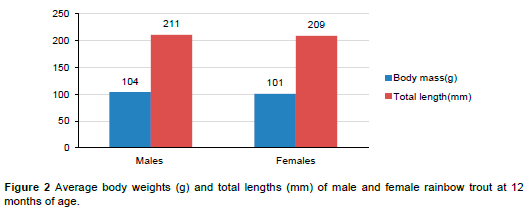
The results shown in Figure 2 demonstrate the well-known biological differences in energy distribution between the sexes in rainbow trout. Males allocate a greater proportion of their energy intake to somatic growth, which leads to greater body weight and length, while females allocate a significant proportion of their energy to gonadal development (Aksungur et al, 2007; Gunadi et al, 2021). Fluctuations in body mass associated with fish sex become more pronounced as fish approach their first spawning event (Barson et al., 2015). Sex is therefore considered an important factor in selection programmes to increase growth and production efficiency.
Conclusions
Significant differences in the phenotypic expressions and variabilities of body weight and total length were found within the studied families of rainbow trout in the Republic of Serbia. Rainbow trout families with high genetic variability enable the selection of individuals with desirable traits for further breeding. This variability increases the adaptive potential of the species and thus improves resistance to diseases and environmental stress factors. The introduction of such families into selection programmes in the Republic of Serbia can significantly improve the quality and productivity of rainbow trout aquaculture. The determined heritability values, which were measured at five, 12, 24, and 36 months of age and ranged from 0.22 to 0.68, indicated that there was moderate to high heritability of these traits in the different families. These heritability values indicate the possibility of genetic improvement through a selection programme. As rainbow trout in the Republic of Serbia usually reach market size at 12 to 18 months of age, the genetic improvement of growth traits is primarily important for this age range. We thus conducted a comparative analysis of the growth performances of 12-month-old fish from the G0 generation (2011-2013) and their offspring from the G1 generation (2014-2016), with the results suggesting an improvement in the traits measured. The estimated heritability provides an excellent basis for the continuation of the selective breeding programme, which will ultimately lead to the improvement of salmonid aquaculture and the profitability of rainbow trout production in Republic of Serbia.
Acknowledgements
This paper reports the results of research supported by the contract on the realisation and financing of scientific research between the Faculty of Agriculture and the Ministry of Science, Technological Development, and Innovation of the Republic of Serbia (contract registration number: 451-03-137/2025-03/200116).
Authors' contributions
This paper is the result of the joint work of all the authors. V.G., M.S., and D.S. collected data, analysed the data, interpreted the results, and wrote the manuscript. Z.M., R.D.J., and S.M. designed and assessed the study. D.V. works at the Centre for Fisheries and Applied Hydrobiology. All authors read and approved the final manuscript.
Conflict of interest declaration
The authors declare that they have no conflicts of interest.
References
Aksungur, N., Aksungur, M., Akbulut, B., & Kutlu, I., 2007. Effects of stocking density on growth performance, survival and food conversion ratio of Turbot (Psetta maxima) in the net cages on the southeastern coast of the Black Sea. Turkish Journal of Fisheries and Aquatic Sciences, 7(2):147-152. [ Links ]
Barson, N.J., Aykanat, T., Hindar, K., Baranski, M., Bolstad, G.H., Fiske, P., Jacq, C., Jensen, A.J., Johnston, S.E., Karlsson, S., Kent, M., Moen, T., Niemelä, E., Nome, T., Nœsje, T.F., Orell, P., Romakkaniemi, A., Sœgrov, H., Urdal, K., Erkinaro, J., Lien, S., & Primmer, C.R., 2015. Sex-dependent dominance at a single locus maintains variation in age at maturity in salmon. Nature, 528:405-408. DOI: https://doi.org/10.1038/nature16062 [ Links ]
D'Ambrosio, J., Phocas, F., Haffray, P., Bestin, A., Brard-Fudulea, S., Poncet, C., Quillet, E., Dechamp, N., Fraslin, C., Charles, M., & Dupont-Nivet, M., 2019. Genome-wide estimates of genetic diversity, inbreeding and effective size of experimental and commercial rainbow trout lines undergoing selective breeding. Genetics Selection Evolution, 51(1):26. DOI: https://doi.org/10.1186/s12711-019-0468-4 [ Links ]
EL-Bab, A. F. F., El-Saiegh, S. S., Abd-Elghany, M. F., Khalifa, G. A. & Naiel, M. A. E. 2024. Influence of spawning year and size on reproductive parameters in adult European sea bass (Dicentrarchus labrax). Discover Animals, 1:13. https://doi.org/10.1007/s44338-024-00016-z. [ Links ]
Gjedrem, T., 2010. The first family-based breeding program in aquaculture. Reviews in Aquaculture, 2:2-15. DOI: https://doi.org/10.1111/j.1753-5131.2010.01011.x [ Links ]
Gjedrem, T., Robinson, N., & Rye, M., 2012. The importance of selective breeding in aquaculture to meet future demands for animal protein: a review. Aquaculture, 350-353:117-129. DOI: https://doi.org/10.1016/j.aquaculture.2012.04.008 [ Links ]
Groeneveld, E., Kovac, M., & Mielenz, N., 2010. VCE6 User's Guide and Reference. Institute of Farm Animal Genetics, Neustadt, Germany. [ Links ]
Groeneveld, E., Kovac, M., & Wang, T., 1990. PEST, a general purpose BLUP package for multivariate prediction and estimation. In: 4th World Congress on Genetics Applied to Livestock Production, Edinburgh, Scotland, 13:488-491. [ Links ]
Gunadi, B. & Robisalmi, A., 2021. Improving genetic quality of cultivated aquatic species under a breeding program: Case study of first generation (G1) population of red tilapia (Oreochromis spp). IOP Conference Series: Earth and Environmental Science, 744: 012020. DOI: 10.1088/1755-1315/744/1/012020 [ Links ]
Hamzah, A., Ponzoni, W. R., Nguyen, H. N., Khaw, L. H., Yee, Y. H., & Nor, A. M. S. 2014. Genetic evaluation of the Genetically Improved Farmed Tilapia (GIFT) strain over ten generations of selection in Malaysia. Pertanika Journal of Tropical Agriculture Science, 37 (4): 411-429. [ Links ]
Houston, R.D., Bean, T.P., Macqueen, D.J., Gundappa, M.K., & Houston, L., 2020. Harnessing genomics to fast-track genetic improvement in aquaculture. Nature Reviews Genetics, 21(7):389-409. DOI: https://doi.org/10.1038/s41576-020-0227-y [ Links ]
Hu, G., Gu, W., Bai, Q., & Wang, B., 2013. Estimation of genetic parameters for growth traits in a breeding program for rainbow trout (Oncorhynchus mykiss) in China. Genetics and Molecular Research, 12(2):1457-1467. DOI: https://doi.org/10.4238/2013.april.26.7 [ Links ]
Hung, D., Vu, N., Nguyen, N., Ponzoni, R., Hurwood, D., & Mater, P., 2013. Genetic response to combined family selection for improved mean harvest weight in giant freshwater prawn (Macrobrachium rosenbergii) in Vietnam. Aquaculture, 412-413:70-73. DOI: 10.1016/j.aquaculture.2013.07.015 [ Links ]
Janampa-Sarmiento, P.C., Takata, R., De Freitas, T.M., De Sá Freire, L., De Britto Pereira, M.M., Lugert, V., Heluy, G.M., & Pereira, M.M., 2020. Modeling the weight gain of freshwater-reared rainbow trout (Oncorhynchus mykiss) during the grow-out phase. Revista Brasileira de Zootecnia, 49: e20190028. DOI: https://doi.org/10.37496/rbz4920190028 [ Links ]
Johnsson, J.I., Brockmark, S., & Näslund, J., 2014. Environmental effects on behavioural development consequences for fitness of captive-reared fishes in the wild. Journal of Fish Biology, 85(6):1946-1971. DOI: https://doi.org/10.1111/jfb.12547 [ Links ]
Kause, A., Paananen, T., Ritola, O., & Koskinen, H., 2007. Direct and indirect selection of visceral lipid weight, fillet weight and fillet percentage in a rainbow trout breeding program. Journal of Animal Science, 85(12):3218-3227. DOI: https://doi.org/10.2527/jas.2007-0332 [ Links ]
Kause, A., Ritola, O., Paananen, T., Mäntysaari, E., & Eskelinen, U., 2002. Coupling body weight and its composition: a quantitative genetics analysis in rainbow trout. Aquaculture, 211(1 -4):65-79. DOI: https://doi.org/10.1016/S0044-8486(01)00884-5 [ Links ]
Leeds, T.D., Vallejo, R.L., Weber, G.M., Gonzalez-Pena, D., & Silverstein, J.T., 2016. Response to five generations of selection for growth performance traits in rainbow trout (Oncorhynchus mykiss). Aquaculture, 465(1):341-351. DOI: https://doi.org/10.1016/j.aquaculture.2016.08.036 [ Links ]
Marković, Z. & Mitrović-Tutundzić, V., 2003. Gajenje riba. Zaduzbina Andrejevic. pp.129. (in Serbian). [ Links ]
Nœve, I., Korsvoll, S.A., Santi, N., Medina, M., & Aunsmo, A., 2022. The power of genetics: Past and future contribution of balanced genetic selection to sustainable growth and productivity of the Norwegian Atlantic salmon (Salmo salar) industry. Aquaculture, 553: 738061. DOI: https://doi.org/10.1016/j.aquaculture.2022.738061 [ Links ]
Nguyen, N.H., 2016. Genetic improvement for important farmed aquaculture species with a reference to carp, tilapia and prawns in Asia: Achievements, lessons and challenges. Fish and Fisheries, 17(2):483-506. https://doi.org/10.1111/faf.12122 [ Links ]
Robledo, D., Gutiérrez, A.P., Barría, A., Lhorente, J.P., Houston, R.D., & Yáñez, J.M., 2019. Discovery and functional annotation of quantitative trait loci affecting resistance to sea lice in Atlantic salmon. Frontiers in Genetics, 10:56. DOI: https://doi.org/10.3389/fgene.2019.00056 [ Links ]
Sae-Lim, P., 2016. Climate change and selective breeding in aquaculture. Journal of Animal Science, 94(suppl 5):195-196. DOI: https://doi.org/10.2527/jam2016-0403 [ Links ]
Sae-Lim, P., Kause, A., Janhunen, M., Vehviläinen, H., Koskinen, H., Gjerde, B., Lillehammer, M., & Mulder, A.H., 2015. Genetic (co)variance of rainbow trout (Oncorhynchus mykiss) body weight and its uniformity across production environments. Genetics Selection Evolution, 47:46. DOI: https://doi.org/10.1186/s12711-015-0122-8 [ Links ]
Sae-Lim, P., Komen, H., Kause, A., Martin, K.E., Crooijmans, R., van Arendonk, J.A.M., & Parsons, J.E., 2013. Enhancing selective breeding for growth, slaughter traits and overall survival in rainbow trout (Oncorhynchus mykiss). Aquaculture, 372-375:89-96. DOI: https://doi.org/10.1016/j.aquaculture.2012.10.031 [ Links ]
SAS, 2013. SAS Version 9.1.3, SAS Institute Inc. Cary, NC, USA. [ Links ]
Serbezov, D., Bernatchez, L., Olsen, E., & Vøllestad, L., 2010. Quantitative genetic parameters for wild stream-living brown trout: heritability and parental effects. Journal of Evolutionary Biology, 23:1631-1641. DOI: https://doi.org/10.1111/j.1420-9101.2010.02028.x [ Links ]
Silverstein, J.T., Vallejo, R.L., Palti, Y., Leeds, T.D., Rexroad, III C.E., Welch, T.J., Wiens, G.D., & Ducrocq, V., 2009. Rainbow trout resistance to bacterial cold-water disease is moderately heritable and is not adversely correlated with growth. Journal of Animal Science, 87(3):860-867. DOI: https://doi.org/10.2527/jas.2008-1157 [ Links ]
Thodesen, J., Rye, M., Yu-Xiang, W., Kong-Song, Y., Bentsen, B.H., & Gjedrem, T., 2013. Genetic improvement of tilapias in China: genetic parameters and selection responses in growth, survival and external color traits of red tilapia (Oreochromis spp.) after four generations of multi-trait selection. Aquaculture, 416-417:354-366. DOI: https://doi.org/10.1016/j.aquaculture.2013.09.047 [ Links ]
Vo, T.A., Galloway, T.F., Bardal, T., Halseth, C.K., Øie, G., & Kjørsvik, E., 2016. Skeletal muscle growth dynamics and the influence of first-feeding diet in Atlantic cod larvae (Gadus morhua L.). Biology Open, 5(11):1575-1584. DOI: https://doi.org/10.1242/bio.018556 [ Links ]
Yáñez, J.M., Houston, R.D., & Newman, S., 2014. Genetics and genomics of disease resistance in salmonid species. Frontiers in Genetics, 5:415. DOI: https://doi.org/10.3389/fgene.2014.00415 [ Links ]
Yáñez, J.M., Newman, S., & Houston, R.D., 2015. Genomics in aquaculture to better understand species biology and accelerate genetic progress. Frontiers in Genetics, 6:128. DOI: https://doi.org/10.3389/fgene.2015.00128 [ Links ]
Yoshida, G.M., Bangera, R., Correa, K., Soto, J., Salas, D., & Yáñez, J.M., 2019. Genomic prediction accuracy for growth and fillet traits in Nile tilapia (Oreochromis niloticus). G3: Genes, Genomes, Genetics, 9(9):2597-2607. DOI: https://doi.org/10.1534/g3.119.400225 [ Links ]
Submitted 11 December 2024
Accepted 16 July 2025
Published 20 August 2025
# Corresponding author: vukosavg@agrif.bg.ac.rs













