Services on Demand
Journal
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
Journal of the South African Veterinary Association
On-line version ISSN 2224-9435Print version ISSN 1019-9128
J. S. Afr. Vet. Assoc. vol.96 n.1 Pretoria 2025
https://doi.org/10.36303/jsava.679
Ovine balanoposthitis: observations on the microbiome and immunoglobulin response
L Fernhout; H Lambrechts; JHC van Zyl
Department of Animal Sciences, Faculty AgriSciences, Stellenbosch University, South Africa
ABSTRACT
The incidence of ulcerative balanoposthitis (UB) in rams contributes to significant economic losses in the national sheep population. Understanding the immune response in the reproductive tract can aid in developing preventive measures. This study aimed to characterise the immunoglobulin profiles and microbial diversity in the reproductive tract of rams affected by UB, providing insight into the immune responses to this disease. Serum and glans tissue samples from healthy and UB-affected rams were analysed using an ELISA approach to quantify IgG, IgA, and IgM levels. The microbial diversity in nasal, penile swabs, and smegma samples was assessed using an ARISA approach. Significant differences (p < 0.001) were observed between the systemic and localised immune responses. UB-affected rams exhibited higher IgG and IgM levels but lower IgA levels in both serum and glans tissue compared to healthy rams. The study confirmed UB-induced shifts in the microbiome, with significant differences in microbial diversity indices in nasal, penile, and smegma samples. Specifically, there were changes in the nasal fungi Shannon (p = 0.047) and Simpson (p = 0.038) indices, the penile Shannon (p = 0.015) and Simpson (p = 0.006) bacterial indices, and the smegma bacterial species number index (p = 0.042). Correlations between microbial populations and immunoglobulin profiles indicated an interactive immune response in different health statuses. This study highlights the need to understand the immune system of the lower reproductive tract and its interaction with commensal organisms to develop therapeutic immunomodulators for preventing UB in sheep.
Keywords: ARISA, ELISA, sheep, immune response, venereal
Introduction
Ulcerative balanoposthitis (UB) is a venereal disease that occurs iwhich has been classified as an economically important disease in South Africa (Kidanemariam et al. 2005; Ali 2012; Courchay 2017). Symptoms of UB in South African Dorper rams include hyperaemia and inflammation of the mucosal surface of the penis, which then progresses into small scattered papulo-vesicular lesions which can become erosions or ulcerations causing the penis (glans penis, prepuce/foreskin, and urethral process) to be covered with fibrinous or mucopurulent exudate (Trichard et al. 1993; Kidanemariam et al. 2005; Rutten 2012; Courchay 2017). The disease leads to severe economic losses due to decreased fertility, increased veterinary care costs, and the culling of affected animals (Trichard et al. 1993; Doe 2018). The welfare of the animals is compromised due to pain, discomfort, and secondary infections that aggravate the condition (Smith et al. 2019). Addressing UB is therefore crucial for providing economic stability for farmers and ensuring better care for the animals, with the goal of more effective productivity and sustainability of sheep farming.
Since 1993 researchers have been working towards identifying the aetiological agent of this disease, primarily focused on the clinical manifestations and basic epidemiology of UB, with limited insights into the underlying immunological mechanisms and microbiome alterations (Trichard et al. 1993; Kidanemariam et al. 2005; Ali 2012; Courchay 2017; Brown et al. 2020). Immunoglobulins, such as IgG, IgA, and IgM, play vital roles in the immune response, yet their specific roles in the context of UB remain poorly understood, which complicates the formulation of proper management and therapeutic strategies (Dibarrat et al. 2007; Johnson 2021). The re-occurrence of the disease in specific animals also indicates that the acquired immune response is ineffective to retain a memory of UB infection, that the initial infection is never completely resolved and flares up when certain unknown conditions change (Trichard et al. 1993; Aristizábal & González 2013; Courchay 2017; Smith et al. 2019; Lambrechts 2021). Additionally, the microbiome of the reproductive tract and its interaction with the immune system during UB infection is an underexplored area. Thus, an improved understanding of the response of the immune components and the microbiome in the male reproductive tract in sheep, once infected with UB, is needed.
The aims and objectives of this study were, therefore, to characterise the immune response of UB-affected rams by measuring the change in the IgG, IgA, and IgM profile of the serum and the glans penile tissue, in order to provide insights into the localised and systemic immune responses to UB, contributing to the development of effective preventive and therapeutic strategies.The bacterial and fungal population dynamics of the upper respiratory- and lower reproductive tract was also characterised in UB-affected rams, to determine the correlations between immunoglobulin profiles and microbial populations in different health statuses of rams.
Methods and design
This research utilised a cross-sectional study design to evaluate the immune response and microbial diversity in the lower reproductive tract of rams affected by UB compared to healthy control animals. The study was conducted at the Sheep Section on the Welgevallen Experimental Farm of Stellenbosch University, South Africa. This setting provided a controlled environment where all rams were housed in individual disinfected pens and received a uniform diet ad lib, with free access to drinking water throughout the experimental period.
The study involved a total of 22 adult rams, comprising 10 Dohne Merino rams and 12 Dorper rams. The average live weight of the rams was approximately 25 kg. Inclusion criteria required rams to be either healthy or affected by UB. The UB status of each ram was classified by the absence or presence of ulcerations on the mucosal membrane of the glans penis only, or on the prepuce only, or on both the mucosal membrane of the glans penis and the prepuce. The classification system for UB was developed based on previously described clinical signs (Trichard et al. 1993: 29-37; Kidanemariam et al. 2005; Ali 2012; Courchay 2017), and the severity was ranked in terms of the ulcerations present on the glans penis and prepuce area.
These animals were part of a research flock (ACU-2022-24709) designated for slaughter. The reproductive tracts of each slaughtered animal were collected during the process.
Data collection involved several procedures. A5mL blood sample was collected from each ram in serum-separating tubes (SST) (Lasec, South Africa) using venepuncture. Blood samples were transported on ice (4 °C) to the laboratory, and were allowed to stand overnight at 4 °C allowing natural clotting and separation of the serum. The supernatant was transferred into a clean sterile Eppendorf tube and stored at -80 °C until further analysis (Cusabio Sheep Immunoglobulin ELISA Kit Protocol).
Smegma samples were collected using an intra-uterine lavage catheter for bovines (~ 20 cm; Lakato [Pty] Ltd, South Africa) (Dibarrat et al. 2007). A disposable syringe introduced 10 mL of pre-warmed (37 °C) sterile PBS through the catheter, and the prepuce was gently massaged for about one minute before the sample was extracted and transferred to a sterile vial (Dibarrat et al. 2007). Each catheter was flushed with fresh pre-warmed PBS to ensure that the entire smegma sample was obtained, and the samples were transported on ice (4 °C) to the laboratory and stored at -20 °C until further analysis.
Penile sheath swabs of each ram were collected using a regular Copan FLOQswab™ (HCPN519CS01; Lasec, South Africa) by rolling the swab over the entire inner membrane of the area for about 20 seconds (Courchay 2017). Each swab was then placed in a conical tube containing 3 mL universal transport medium (UTM™) (HCPN330C; Lasec, South Africa). The UTM tubes with the swab, were placed on ice (4°C) and transported to the laboratory and stored frozen at -20 °C until further analyses.
Each ram was restrained for the collection of nasal swabs for the estimation of microbial diversity and community composition analysis. Nasal swabs were collected using a regular Copan FLOQswab™ (HCPN519CS01; Lasec, South Africa) by rolling the swab inside the entire nasal cavity for about 20 seconds (Courchay 2017). Each swab was then transferred to a conical tube containing 3 mL universal transport medium (UTM™) (HCPN330C; Lasec, South Africa). These swabs were placed in a conical tube containing 3 mL universal transport medium (UTM™) (HCPN330C; Lasec, South Africa). The UTM tubes with the swab, were placed on ice (4 °C) and transported to the laboratory and stored frozen at -20 °C until further analyses.
All rams were slaughtered at a commercial abattoir, where the reproductive tracts of all the animals were collected on the slaughter line and transported on ice (4 °C) to the laboratory. At the laboratory, all reproductive tracts were inspected for ulcers on the sheath/prepuce area as well as on the glans penis, the central penis, and the preputial ring fold. The penis was then extracted from the sheath and cut off at the posterior end. A tissue sample of the glans penis area was collected, rinsed with phosphate-buffered saline (PBS), homogenised in 1 mL PBS, and stored overnight at -20 °C in 15 mL PBS. The tissue homogenates were subjected to two freeze-thaw cycles to break the cell membranes, whereafter the homogenates were centrifuged for five minutes at 5 000 RCF, at 8 °C. The supernatant was removed, and the aliquot was stored at -20 °C until further analysis (CUSABIO; ELISA Protocol).
Data analysis involved immunoglobulin quantification using an enzyme-linked immunosorbent assay (ELISA) to quantify IgG, IgA, and IgM levels, microbial diversity analysis was assessed using automated ribosomal intergenic spacer analysis (ARISA), and statistical comparison using various statistical tests.
The serum and glans penis tissue concentration of immunoglobulins IgG, IgA, and IgM were determined using an ELISA approach. Commercial sheep-specific ELISA kits (CUSABIO, Sheep Immunoglobulin G, A, and M ELISA Kit; Cat# CSBE14400Sh, CSB-E13681Sh, and CSB-E13682Sh; Biocom Africa [Pty] Ltd.) were used according to the manufacturer's instructions for sheep. The tissue homogenate supernatant was used without any dilutions, and all tissue sample concentrations were divided by the sample weight to ensure correction for sample weight.
ARISA was used to quantify the bacterial and fungal diversity of the smegma, penile- and nasal- swab samples. Total genomic DNA was extracted from the samples with the use of the QuickDNA Fecal/Soil Microbe Miniprep Kit (Zymo Research). The extracted DNA was then used in the ARISA polymerase chain reaction (PCR). With the use of ARISA PCR, the diversity and community composition of the penile and nasal environment were estimated by amplifying the Internal Transcribed Spacer (ITS) region of both bacteria and fungi. The variability in length of the ITS regions infers diversity, whereby different lengths represent different operational taxonomic units (OTUs, or number of species). The PCRs were done by using fluorescent-labelled forward primers, ITSF-FAM (5'GTCGTAACAAGGTAGCCGTA-3') for bacteria, and ITS5 (5'-GGAAGTAAAAGTCTAACAAGG-3') for fungi, and the reverse primers ITSreub (5'-GCCAAGGCATCCACC-3') and ITS4 (5'TCCTCCGCTTATTGATATGC-3') for bacteria and fungi, respectively. Each PCR reaction mixture consisted of 5 μL KapaTaq Readymix, 10 pM forward primers, 10 pM reverse primers, 0.5 μL DNA template, and 4.1 μL double distilled water, to a final volume of 10 μL. The PCR amplification took place under the following conditions: initial denaturation at 95 °C for 5 min, denaturation of 36 cycles at 95 °C for 45 seconds, annealing at 56 °C for 50 seconds, and extension at 72 °C for 70 seconds for bacteria. For fungi, the initial denaturation was 95 °C for 5 min, denaturation for 36 cycles at 95 °C for 45 seconds, annealing at 54 °C for 50 seconds, and extension at 72 °C for 70 seconds. A final extension was completed at 72 °C for 7 minutes for both microbes. Each sample was amplified in triplicate and pooled before being sent to the Central Analytical Facility (CAF) for analysis. An automated Genetic Analyzer ABI 3100 was used for capillary analysis using Liz1200 as the size standard. The raw data was analysed using GeneMapper v5, and the genotypes table was used in further analyses.
The statistical analysis of data was performed using XLSTAT (version 2022.1). All alpha- and beta-diversity calculations, as well as statistical calculations (PERMANOVA), were analysed in R statistical software (version 4.1.3), with packages: vegan, ggplot2, ellipse, ggdendro, grid, dplyr, dendextend, and factoextra. Data were assessed for normality and homoscedasticity with the use of a Shapiro-Wilks test and Levene's test, respectively. Not normally distributed data were transformed with the use of a Log-function. This data was studied with an analysis of variance (ANOVA) test, with a two-way interaction included accounting for possible interactions between the variables (health status and sample type). Where unable to use ANOVA due to non-normal and non-homoscedastic data, a non-parametric Kruskal-Wallis test was used in XLSTAT. Effects were considered significant at a p < 0.05.
Ethical clearance was obtained from the Stellenbosch University Research Ethics Committee for Animal Care and Use, with the permit number ACU-2022-22342. All procedures adhered to ethical standards for animal research.
Results
Table I presents the serum and tissue immunoglobulin concentrations measured for the healthy (i.e. no UB) and UB-positive rams in the study. No significant differences were observed between the healthy and the UB-positive group (IgG p = 0.293; IgA p = 0.937; IgM p = 0.170), neither were there any significant interactions observed between the health status and the sample type (IgG p = 0.452; IgA p = 0.051; IgM p = 0.093).
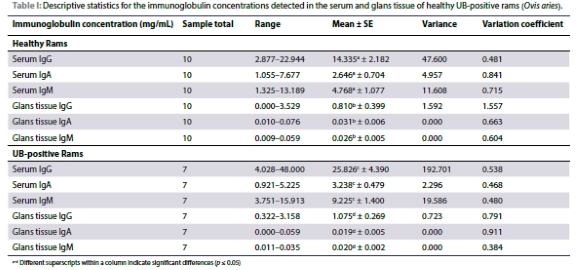
All ARISA results were expressed with the following indexes. The Shannon index considers the number of species living in a habitat (richness) and their relative abundance (evenness), with > 2.5 as high, 1.5 as average, and < 1 as low. A high value of Shannon would be representative of a diverse and equally distributed community and lower values represent a less diverse community (Conradie 2022). The Simpson index is a measure of diversity which considers the number of species present, as well as the relative abundance of each species. A value closer to 1 means there is an equal distribution of all species within the sample, and a value closer to 0, indicates that there are dominant species present. The number of species index, indicates the number of species present within the sample (Conradie, 2022). Table II presents descriptive statistics for the microbial and fungal population of the healthy and UB- infected group.
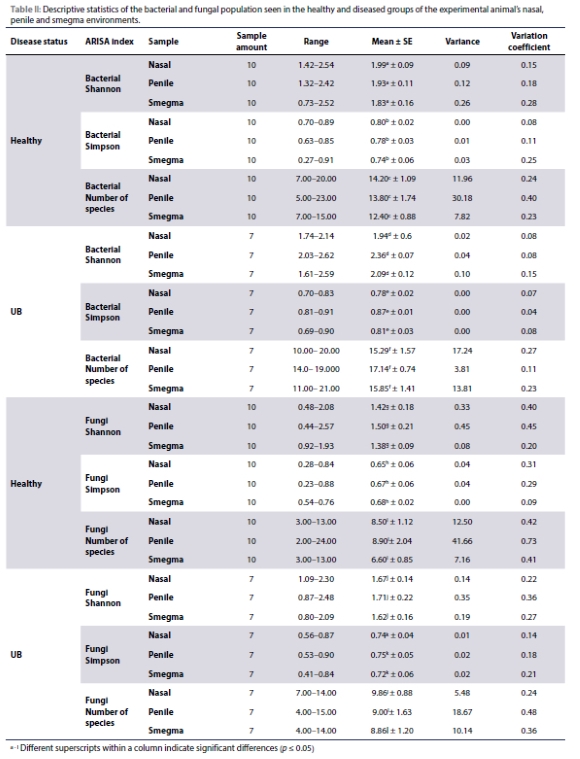
Figure 1 represent non-metric multidimensional scaling (NMSD) ordination plots of the bacterial and fungal beta diversities for the comparison of community compositions between the three different sample types. No significant differences were observed between the three different sample types (nasal swab, penile swab, and smegma) for the bacterial or fungi indexes, irrespective of the presence of disease (Figure 1).
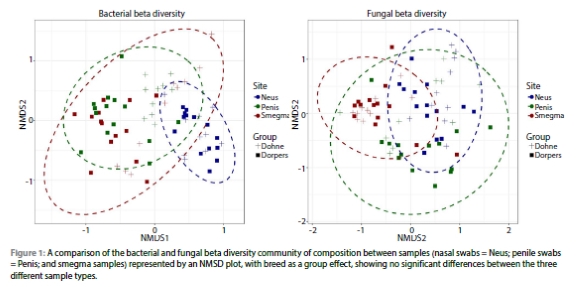
With the use of a Kruskal-Wallis test, the relationship between the healthy and UB group, and the microbial population was investigated to see if any of the health conditions cause a change in the microbial population of the three sampling locations (nasal swab, penile swab, and smegma samples).
When the overall influence of the different health statuses on the microbial population of the nasal environment is considered, no significant differences were observed for the bacterial Shannon index (p = 0.103), the number of bacterial species index (p = 0.250), and the number of fungal species index (p = 0.218). With the consideration of health status on the nasal bacterial Simpson index, a tendency towards significance exists (p = 0.080). Significant differences were caused by health status in the fungal population of the nasal environment (fungi Shannon p = 0.047 and fungal Simpson p = 0.038).
When the overall influence of the different health statuses on the microbial population of the penile environment is considered, no significant differences have been detected for the bacterial Shannon index (p = 0.087), the number of bacterial species index (p = 0.367), fungi Shannon index (p = 0.400), the fungi Simpson index (p = 0.270), or the number of fungal species index (p = 0.714). Significant differences were caused by health status in the bacterial population of the penile environment (bacterial Simpson p = 0.050). The penile Shannon (p = 0.015) and the penile Simpson (p = 0.006) bacterial indexes of the UB-infected group compared to the healthy group, showed significant differences.
When the overall influence of the different health statuses on the microbial population of the smegma sample composition is considered, no significant differences were observed for the bacterial Shannon index (p = 0.497), the bacterial Simpson index (p = 0.760), fungi Shannon index (p = 0.205), the fungi Simpson index (p = 0.212), or the number of fungal species index (p = 0.229). With the consideration of health status on the smegma number of bacterial species index, a tendency towards significance exists (p = 0.082). A significant difference was seen between the smegma bacterial species number (p = 0.042) of the UB-infected group of rams compared to the healthy group.
Figure 2 presents a comparison of the averages of the three ARISA indexes for bacterial and fungal populations of the nasal swab, penile swab, and smegma samples obtained from the healthy and diseased rams.
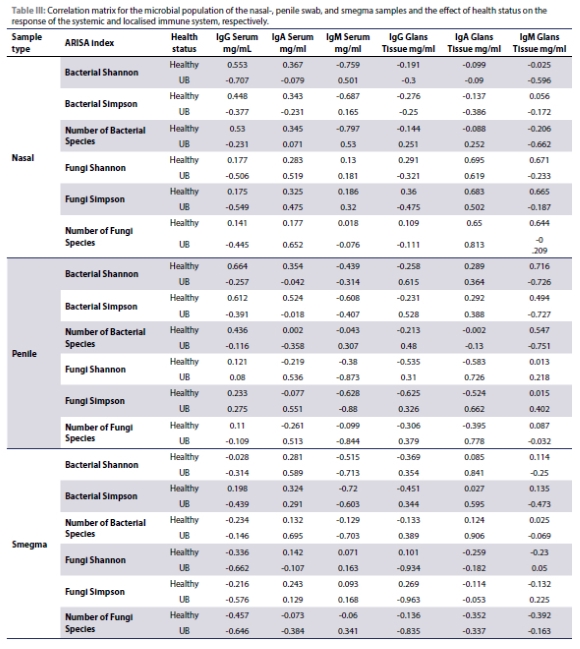
The effect of a changing microbial population and the relationship seen in the immune response was determined with the use of a correlation matrix for the healthy and diseased groups. Table III indicates the correlation coefficients between the microbial population and the immunoglobulin profile, and how the immune system will react to the changing microbial population of the different health status groups.
 .
.
Discussion
In the current study, immunoglobulin concentrations (i.e. IgG, IgA, and IgM) were determined in serum and tissue samples collected from healthy rams (i.e. not affected by UB), and rams affected by UB. No literature could be found on the immunoglobulin profile of the lower reproductive tract of rams, suggesting that the current study, therefore, presents novel information in this regard. The immunoglobulin levels reported for the serum and glans tissue samples in the current study will therefore be used as reference values to contextualise and discuss the immunoglobulin levels detected in the UB affected rams (Table I).
In this study, serum immunoglobulin concentrations were significantly higher than those in the glans tissue samples of healthy rams, which was expected as antibodies make up a substantial portion of blood plasma proteins (Grattendick & Pross 2007). This difference highlights the distinct roles of the localised immune system in the glans tissue, which responds to pathogens at the site, versus the systemic immune system circulating throughout the body (Berneman et al. 1998; Dibarrat et al. 2007; National Library of Medicine 2022). Systemic immunity, involving antigen-presenting cells and the retention of memory cells, contrasts with localised immunity, which focuses on limiting infection spread and tissue repair (Gnanasampanthan 1993; Berneman et al. 1998; Rynkiewicz et al. 2019; Alexandre et al. 2020). The study underlines that interpreting systemic immune responses to understand localised infections can be misleading, emphasising the need to differentiate between these two immune system functions for accurate analysis of immune responses, particularly in mucosal surfaces where immunological memory is less clear (Gnanasampanthan 1993; Garulli et al. 2004; Nagaoka et al. 2017).
Although not significant, rams positive for UB displayed higher overall serum immunoglobulin levels for IgG and IgM, but lower levels for IgA, compared to UB-negative rams. When the tendency of interaction between UB incidence and sample type is considered, serum samples of the UB-positive rams were characterised by higher IgG, IgA, and IgM levels when compared to that of the healthy rams. IgM is the first immunoglobulin to respond to infections, offering short-term protection, while IgG is abundant and crucial for fighting bacterial and viral infections and retaining memory of antigens (Janeway et al. 2001; National Library of Medicine 2022). Serum IgA provides resistance against infections and neutralises viruses (Woof & Kerr 2006). The increased levels of these immunoglobulins in UB-positive rams suggest an active systemic immune response to UB infection.
In the glans tissue, UB-positive rams had higher IgG levels, whereas healthy rams had higher IgA and IgM levels. The elevated IgG in the glans tissue indicates an active localised immune response to the UB infection, suggesting that IgG production can be localised to the penile tissue independently of the systemic immune system (Berneman et al. 1998). Secretory IgA, important for defending mucosal surfaces, was observed to be lower in this study. Overall, the study demonstrates both localised and systemic immune responses to UB infection in rams, with notable increases in immunoglobulin levels indicating immune activity.
Previous studies have suggested that microbial pathogens might play a role in the prevalence of UB in rams. Analysis revealed 1 084 OTUs for bacteria and 622 OTUs for fungi across three sampling environments, indicating significant bacterial and fungal diversity in both the upper respiratory tract and the lower reproductive tract of rams. There were no significant differences in bacterial and fungal indexes among the sample sites, regardless of the rams' disease status. Some venereal diseases are commonly transmitted through oral-nasal routes rather than venereal routes, suggesting that comparing microbial indexes from these different sites could indicate potential non-venereal transmission routes for UB (Camacho et al. 2005). The absence of significant differences in microbial indexes supports the hypothesis that microbes can migrate along mucosal surfaces, linking the oral-nasal and reproductive areas (Aich & Dwivedy 2011). This implies that microorganisms in the upper respiratory tract could contribute to diseases in the lower reproductive tract.
This study examined fungal communities in the lower respiratory and reproductive tracts of rams and found no significant differences between UB-infected and healthy groups. However, the fungal Shannon and Simpson indexes of smegma samples indicated a trend toward significance, with UB-infected rams displaying more diverse and equally distributed fungal communities. This suggests that UB infection may alter fungal species distribution, potentially influencing rams' susceptibility to UB.
Based on the microbial responses observed in healthy and diseased rams, further investigation was conducted to explore the correlation between changes in the natural microbiome and the immunoglobulin profile. The study revealed several associations: serum and glans tissue IgA levels increased with higher bacterial species numbers in UB-infected animals, while serum IgM decreased with increased bacterial species in smegma. UB infection correlated with increased glans tissue IgG with higher penile bacterial diversity, yet decreased glans tissue IgM with greater bacterial richness and evenness. The penile Simpson index showed that serum IgG, IgA, and IgM decreases with an increasing Simpson index caused by UB. The correlations between this index and the IgG and IgM of the glans tissue suggests that both tissue IgG and IgM increases with the increase of the Simpson index, once infected with UB. Nasal fungal Shannon indexes showed that higher fungal species richness and evenness correlated with increased serum IgA and IgM, decreased glans tissue IgG, and slightly increased glans tissue IgA and IgM, indicative of disease.
Overall, these changes highlight the complex interaction between commensal microbes in the lower respiratory and reproductive tracts and immune system responses (Kotwal 1997). Understanding these dynamics could aid in developing novel immunomodulatory therapies for managing disease outbreaks in veterinary settings, emphasising the critical role of mucosal sites in distinguishing between commensal and pathogenic organisms for effective immune protection (Entrican & Wheelhouse 2006; Belkaid & Hand 2014).
Conclusion
This study emphasises the need for a deeper understanding of immune responses in the genital tract to mitigate the impact of sexually transmitted diseases like UB in Dorper rams. It identifies research gaps compared to ewe reproductive tract studies, highlighting the ram's immunological competence in initiating innate and acquired immune responses to pathogens. The study provides baseline values for immunoglobulin G, A, and M in healthy sheep serum and reproductive tissue, stressing the distinct roles of systemic and localised immunity. Understanding these interactions is crucial, as UB alters microbial populations in the ram's reproductive tract, emphasising the importance of mucosal and systemic immunity for effective prevention and treatment strategies.
Conflict of interest
The authors declare no conflict of interest.
Funding source
None.
Ethical approval
Ethical clearance was obtained from the Stellenbosch University Research Ethics Committee for Animal Care and Use, with the permit number ACU-2022-22342. All procedures adhered to ethical standards for animal research
ORCID
L Fernhout https://orcid.org/0009-0002-4333-9031
H Lambrechts https://orcid.org/0000-0001-7853-406X
JHC van Zvl https://orcid.org/0000-0001-712-2728
References
Aich, P., Dwivedy, P., 2011, Importance of innate mucosal immunity and the promises it holds, International Journal of General Medicine 4, 299-31 1. https://doi.org/10.2147/IJGM.S17525. [ Links ]
Alexandre, Y.O., Devi, S., Park, S.L., et al., 2020, Systemic inflammation suppresses lymphoid tissue remodelling and B cell immunity during concomitant local infection. Cell Reports 33(13), 108567. https://doi.org/10.1016/j.celrep.2020.108567. [ Links ]
Ali, H., 2012, Molecular characterization of Mycoplasmas species isolated from the genital tract of Dorper sheep in South Africa. Master's Thesis. Pretoria: University of Pretoria. [ Links ]
Aristizábal, B., González, Á., 2013, Autoimmunity: From Bench to Bedside 1st ed. Vol. 1. Bogota: El Rosario University. [ Links ]
Belkaid, Y., Hand, T.W., 2014, Role of the microbiota in immunity and inflammation, Cell 157(1), 121-41. https://doi.org/10.1016/j.cell.2014.03.011. [ Links ]
Berneman, A., Belec, L., Fischetti, V.A. et al., 1998, The specificity patterns of human immunoglobulin G antibodies in serum differ from those in autologous secretions, Infection and Immunity 66(9), 4163-8. https://doi.org/10.1128/IAI.66.9.4163-4168.1998. [ Links ]
Brown, R., Johnson, T., Lee, S., 2020, Clinical manifestations and epidemiology of ulcerative balanoposthitis, Veterinary Pathology 35(4), 455-66. [ Links ]
Camacho, A., Guitian, J., Pallas, E., et al., 2005, Serum protein response and renal failure in canine Babesia annae infection, Veterinary Research 36(5-6), 713-22. https://doi.org/10.1051/vetres:2005026. [ Links ]
Conradie, T.A., 2022, Personal Communication. [2022 29 June] Stellenbosch. [ Links ]
Courchay, A.M.M., 2017, Molecular characterization of the penile microbiome of Dorper rams (Ovis aries). Master's Thesis. Stellenbosch: Stellenbosch University. [ Links ]
Dibarrat, J.A., Aparicio, E.D., Gutierrez, V.R.T., et al., 2007, Determination of pathological changes in the reproductive tract IgG, IgM and Ig antibodies in blood, seminal plasma and smegma of rams inoculated with Actinobacillus seminis, Journal of Animal and Veterinary Advances 6(1), 105-113. [ Links ]
Doe, J., 2018, Impact of ulcerative balanoposthitis on sheep farming in South Africa, Journal of Veterinary Medicine 45(3), 234-45. https://doi.org/10.31588/2413-4201-1883-234-2-45-49. [ Links ]
Entrican, G., Wheelhouse, N.M., 2006, Immunity in the female sheep reproductive tract, Veterinary Research 37(3), 295-309. https://doi.org/10.1051/vetres:2006002. [ Links ]
Garulli, B., Kawaoka, Y., Castrucci, M.R., 2004, Mucosal and systemic immune responses to a human immunodeficiency virus type 1 epitope induced upon vaginal infection with a recombinant influenza A virus, Journal of Virology 78(2):1020-5. https://doi.org/10.1128/JVI.78.2.1020-1025.2004. [ Links ]
Gnanasampanthan, G., 1993, Immune responses of sheep to rumen ciliates and the survival and activation of antibodies in the rumen fluid. Doctoral Thesis. Adelaide: University of Adelaide. [ Links ]
Grattendick, K., Pross, S., 2007, Immunoglobulins. In: S.J Enna and David B. Bylund, eds, xPharm: The Comprehensive Pharmacology Reference 1st ed. Amsterdam: Elsevier. https://doi.org/10.1016/B978-008055232-3.60239-9. [ Links ]
Janeway, C.J., Travers, P., Walport, M., et al., 2001, The mucosal immune system. In: Immunobiology: The Immune System in Health and Disease. 5th ed. New York. Available: https://www.ncbi.nlm.nih.gov/books/NBK10757/ [2022/10/29]. [ Links ]
Johnson, T., 2021, Immunoglobulin responses in livestock diseases, Immunology Today 31(5), 112-9. [ Links ]
Kidanemariam, A., Gouws, J., van Vuuren, M., et al., 2005, Ulcerative balanitis and vulvitis of Dorper sheep in South Africa: A study on its aetiology and clinical features, Journal of the South African Veterinary Association 76(4), 197-203. https://doi.org/10.4102/jsava.v76i4.426. [ Links ]
Kotwal, G.J., 1997, Microorganisms and their interaction with the immune system, Journal of Leukocyte Biology 62(4), 415-29. https://doi.org/10.1002/jlb.62A415. [ Links ]
Lambrechts, H., 2021, Personal Communication. [2021 May 3] Stellenbosch. [ Links ]
Nagaoka, Y., Nosaka, N., Yamada, M., et al., 2017, Local and systemic immune responses to influenza A virus infection in pneumonia and encephalitis mouse models, Disease Markers, https://doi.org/10.1155/2017/2594231. [ Links ]
National Library of Medicine, 2022. Immunoglobulins Blood Test [Online]. Available: https://medlineplus.gov/lab-tests/immunoglobulins-blood-test/ [2022 November 28]. [ Links ]
Rutten, M., 2012, Outbreak of ulcerative vulvitis in a sheep flock caused by Ovine Herpesvirus Type 2, Journal of Comparative Pathology 146(1), 71. https://doi.org/10.1016/j.jcpa.2011.11.103. [ Links ]
Rynkiewicz, E.C., Clerc, M., Babayan, S.A., et al., 2019, Variation in local and systemic pro-inflammatory immune markers of wild wood mice after anthelmintic treatment, Integrative and Comparative Biology 59(5), 1190-202. https://doi.org/10.1093/icb/icz136. [ Links ]
Smith, A., Brown, B., Clark, C., 2019, Secondary infections in ulcerative balanoposthitis, Animal Health Research Reviews 28(2), 150-62. [ Links ]
Smith, N.C., Rise, M.L., Christian, S.L., 2019, A comparison of the innate and adaptive immune systems in cartilaginous fish, ray-finned fish and lobe-finned fish, Frontiers in Immunology 10, 2292. https://doi.org/10.3389/fimmu.2019.02292. [ Links ]
Trichard, C.J., Jordaan, P., Prozesky, L., et al., 1993, The identification of Mycoplasma mycoides mycoides LC as the aetiological agent of balanoposthitis and vulvovaginitis in sheep in South Africa, Onderstepoort Journal of Veterinary Research 60(1), 29-37. PMID: 8392679. [ Links ]
Woof, J.M., Kerr, M.A., 2006, The function of immunoglobulin A in immunity, Journal of Pathology 208(2), 270-82. https://doi.org/10.1002/path.1877. [ Links ]
 Correspondence:
Correspondence:
email: helet@sun.ac.za













