Services on Demand
Journal
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
Southern African Journal of Critical Care (Online)
On-line version ISSN 2078-676XPrint version ISSN 1562-8264
South. Afr. j. crit. care (Online) vol.41 n.1 Pretoria Apr. 2025
https://doi.org/10.7196/SAJCC.2025.v41i1.2049
RESEARCH
Critical pertussis infection in the paediatric intensive care unit: A case series in an outbreak
H CrichtonI; N NkadoII; B MorrowIII; S SalieIV; C ProcterV
IMB ChB FCPaed; Department of Paediatrics and Child Health, Red Cross War Memorial Children's Hospital, Cape Town, South Africa
IIMB ChB; Department of Paediatrics and Child Health, Red Cross War Memorial Children's Hospital and University of Cape Town, Cape Town, South Africa
IIIPhD; Department of Paediatrics and Child Health, University of Cape Town, Cape Town, South Africa
IVFCPaed, Cert Critical Care; Department of Paediatrics and Child Health, Red Cross War Memorial Children's Hospital and University of Cape Town, Cape Town, South Africa
VMB BS, Cert Critical Care; Department of Paediatrics and Child Health, Red Cross War Memorial Children's Hospital and University of Cape Town, Cape Town, South Africa
ABSTRACT
BACKGROUND: Bordetella pertussis is a highly infectious disease associated with increased mortality in young infants. Critical pertussis is defined as pertussis disease resulting in paediatric intensive care unit admission or death
OBJECTIVE: This study describes the clinical profile, morbidity, mortality and management of children with critical pertussis admitted to a South African paediatric intensive care unit (PICU
STUDY DESIGN: A retrospective analysis of children with critical pertussis admitted to the PICU over 1 year (January 2022 - December 2022
RESULTS: There were 1 273 PICU admissions during the study period, of which 101 (7.9%) were tested and 19 (1.5%) tested positive for B. pertussis. Of those, 13 (68.4%) were completely unimmunised, mainly owing to young age (n=9). The median (interquartile range) total duration of ventilation was 7.0 (3.0 - 21.0) days, with a maximum of 35 days. Mortality was 36.8% (n=7) in those with B. pertussis; with more deaths in HIV-exposed patients (n=3/7,42.9% v. n=3/12, 25%; p<0.001) and preterm infants (n=4/7,57.1% v. n=4/12, 33.3%; p<0.001), as well as those with septic shock (n=7/7, 100% v. n=4/7, 33.3%; p = 0.02) and acute kidney injury (n=6/7, 85.7% v. n=2/12, 16.7%; p<0.001). The maximum white cell count was significantly lower in survivors (27.75 v. 76.32; p<0.001). None of the variables was independently associated with mortality on multivariable regression
CONCLUSION: Critical B. pertussis infection is associated with significant morbidity and mortality in this setting. The low rate of vaccination is a concern, and advocacy is needed to promote access to vaccination during the third trimester of pregnancy as well as strengthening current immunisation programmes. Further research is required to identify the prevalence of critical pertussis and risk factors for poor outcomes in resource-limited settings
Keywords: Bordetella pertussis; critical pertussis; outcomes
We recommend further research regarding these markers of disease severity as well as therapies offered such as leukoreduction or exchange transfusion.
Pertussis, commonly known as 'whooping cough', is a highly infectious disease caused by the Gram-negative bacterium Bordetella pertussis. The Global Pertussis Initiative states that about 67% of pertussis cases occur in children less than three months old.[1] Pertussis has persisted and resurged despite the widespread use of vaccination against pertussis, with outbreaks occurring worldwide every 3 - 5 years.[2,3] The burden of this disease has been found to be greatest in low- and middle-income countries (LMICs), such as South Africa.[3-5] Precise data from LMICs are lacking owing to variable case definitions, overlap in respiratory clinical presentation and inadequate testing which results in underreporting. In September 2022, the National Institute for Communicable Diseases of South Africa (NICD) reported an increase of pertussis cases in South Africa, with most cases (42%) being reported in the Western Cape.[6] Consequently, there was an increase in the number of pertussis cases being admitted to the Red Cross War Memorial Hospital (RCWMCH) paediatric intensive care unit (PICU) in 2022.
Pertussis disease that requires admission to an intensive care unit (ICU) has been defined as critical pertussis.[7,8] Hyperleukocytosis and pulmonary hypertension are commonly reported risk factors associated with increased mortality in infants with critical pertussis.[9-11] Pertussis toxin- nduced leukocytosis results in aggregates of leukocytes in small vessels in the lungs, leading to irreversible pulmonary hypertension, refractory hypoxaemia, and death in infants.[9,12]
The efficacy of available therapies to treat pulmonary hypertension, such as nitric oxide or leukoreduction therapy by exchange transfusion or plasmapheresis, remains uncertain.[13] Prior reports suggest using clinical and laboratory variables of a rapidly increasing white cell count (WCC) >30xl09/L, heart rate >170 bpm and respiratory rate >70 breaths/min., as well as the presence of pulmonary hypertension as a collective trigger for the institution of exchange transfusion.[13,14] Rowlands et al. recommend using a cut-off WCC value of 50 for leukoreduction therapy.[15] The therapeutic effects of exchange transfusion have been attributed to leukoreduction which reduces circulating pertussis toxin and, as a result, aids in overcoming the refractory hypoxaemia associated with severe pulmonary hypertension.[13] Other treatment modalities include extracorporeal membrane oxygenation (ECMO), which represents advanced respiratory support in the presence of severe pulmonary hypertension. ECMO can be offered to infants who do not improve with exchange transfusion but is associated with a high mortality rate of 70%.[16] Currently in our unit we offer exchange transfusions to critically ill infants, but not ECMO.
A seven-year retrospective review of pertussis admissions at a single centre in South Africa, reported a critical incidence of pertussis of 33%, with a mortality rate of 5.2%.[17] A PICU study from another LMIC, in India, showed a critical pertussis mortality rate of 22% over a three-year study period (2016 - 2018).[7] In 2013, a multicentre study across eight PICUs in the United States reported a 9% PICU mortality rate, and that 83% of infants admitted to PICU with critical pertussis were under two months of age.[11]
The present study aims to describe the characteristics and outcomes of children with critical pertussis admitted to RCWMCH PICU from January to December 2022 and to identify any risk factors for poor outcomes.
Method
This was a single-centre, retrospective analysis of all confirmed pertussis cases admitted to RCWMCH PICU from 1 January to 31 December 2022. RCWMCH PICU is a tertiary-level, 22-bed paediatric ICU situated in Cape Town, South Africa. It receives referrals of critically ill infants and children from paediatric and surgical disciplines throughout the Western Cape. Infants admitted to RCWMCH PICU with a clinical suspicion of Bordetella pertussis, as outlined by the World Health Organization (WHO), were screened.[18] Patients with B. pertussis positive nasopharyngeal aspirate (NPA), tracheal aspirate (TA) or bronchoalveolar lavage (BAL) were included in the study. Patients with B. holmesii/B. bronchiseptica were excluded from the study. Patients with B. pertussis not requiring ICU admission were also excluded. If a patient was readmitted with a complication of B. pertussis, this was counted as a single case. A positive test for B. pertussis was confirmed if two insertion sequences (IS481 and IS1002) were detected. Segments of insertion sequences as listed above are used by the National Health Laboratory Service (NHLS) to differentiate between different Bordetella species. IS1002 is used as a secondary test in order to improve specificity and to confirm a positive screen.[19]
Data collection
For all patients meeting inclusion criteria, de-identified data were extracted from the PICU electronic records system (IntelliSpace Critical Care and Anesthesia) and medical folders and captured onto a Microsoft Excel spreadsheet. The primary outcome of interest was hospital mortality. Data on clinical presentation, clinical course including development of complications (acute kidney injury (AKI), acute liver injury, pulmonary hypertension and hospital-acquired infections) and durations of mechanical ventilation and PICU stay were collected. Laboratory data collected included haematological, biochemical and micro-biological results. An in-depth folder review was conducted to review antimicrobial prescribing practices.
Definitions
Preterm infants were defined as infants born prior to 37 weeks' gestation. AKI was defined according to the Kidney Disease Improving Global Outcomes guidelines (KDIGO).[20] Acute liver injury/dysfunction was defined as liver dysfunction in the absence of known chronic liver disease, with onset of symptoms <8 weeks, combined with biochemical evidence of acute liver injury, and liver-based coagulopathy, with hepatic encephalopathy and an international normalised ratio between 1.5 and 2.0.[21] Septic shock was defined as severe infection leading to cardiovascular dysfunction (including hypotension, need for treatment with a vasoactive medication, or impaired perfusion). [22] Bloodstream infections (BSIs), confirmed on blood culture within 48 hours of admission, were assumed to be community acquired. Hospital-acquired infection was defined as clinically evident infections occurring 48 hours or more after admission and included central line infections, catheter-associated urinary tract infections and ventilator-associated pneumonias.[23]
Statistical analysis
Data were exported from the Excel spreadsheet to IBM SPSS Statistics (version 28.0.1.0) for statistical analysis. Data were initially analysed descriptively, after testing for normality. Central tendencies of continuous data are presented as appropriate for distribution (mean or median and standard deviation or interquartile range). Categorical variables are presented as n (%).
Association with the primary outcome of mortality was determined using t- tests for normally distributed data or Mann-Whitney U-tests for nonparametric data, and chi-square or Fisher's Exact tests for categorical variables.
Variables found to be significantly associated with the primary outcome of mortality on univariate analysis were entered into a best-fit multivariable binary logistic regression analysis, considering issues of collinearity and clinical relevance, to identify independently associated variables. A significance level of 0.05 was used.
Ethical considerations
Approval for the study was obtained from the University of Cape Town's Human Research Ethics Committee (HREC 082/2023). All recorded data were automatically de-identified and assigned a study number in order to ensure confidentiality. The need for parental consent was waived as this was a low-risk, retrospective study based on routinely collected clinical records.
Results
Admission characteristics
Over the study period there were 1 273 admissions to RCWMCH PICU. During this time, 101 patients were tested for B. pertussis, of whom 19 (18.8%) patients (median (IQR) age 45.0 (32.0 - 84.0) days; 45% male) tested positive for B. pertussis. Nine other patients tested positive for B. holmesii/B. bronchiseptica and were excluded from this study. The majority of patients were under 3 months of age (n=15;78.9%) and were admitted to PICU for the primary management of respiratory failure (n=17, 89.5%), apnoea (n=11, 57.9%), and shock (n=11, 57.9%). Eleven (57.9%) patients had existing comorbid conditions, most commonly HIV exposure (n=6, 31.5%) and a history of prematurity (n=8, 42.0%). None was HIV infected. In the study cohort, 13 (68.4%) patients were completely unimmunised. Of the 13 (68.4%) completely unimmunised patients, nine (69.2%) were too young to have received any vaccinations. Six (31.6%) patients had missed immunisations. Leuokocytosis was common, with eight (28.8%) patients having a WCC >50x109/L (Table 1).
Concomitant community-acquired infections
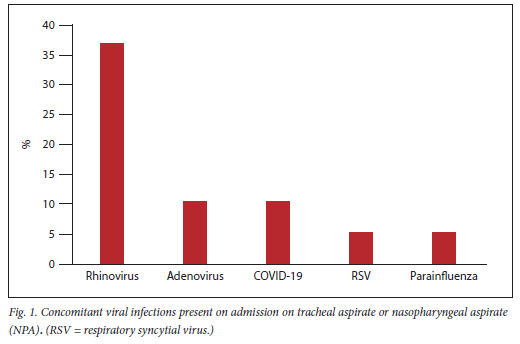
There were three possible concomitant BSIs identified <48 hours of PICU admission. One patient had confirmed Escherichia coli (E.coli) bacteraemia. There were two other positive blood cultures - one with a micrococcus and one with coagulase-negative staphylococcus, which were considered contaminants and were not treated.[24] Of the confirmed viral infections on tracheal or nasopharyngeal aspirates <48 hours of admission to PICU, human rhinovirus was most commonly identified (n=7, 36.8%) (Fig. 1). There was one case with a confirmed E. coli urine infection and one child had meningitis with Gram-positive and Gram-negative organisms on cerebrospinal fluid Gram stain, but no growth on culture.
PICU course
All patients who required invasive ventilation (n=18, 95.0%) were ventilated within 48 hours of admission to PICU. Only one patient was managed non-invasively, with continuous positive airway pressure (CPAP). Eight (42%) patients received conventional mechanical ventilation (CMV) only, while 10 (53%) required escalation to high frequency oscillatory ventilation (HFOV). The unit policy for escalation to HFOV includes failing to achieve adequate gas exchange with CMV when using peak inspiratory pressures >30 cmH2O, refractory hypoxaemia and severe respiratory acidosis.
There were 11/19 patients who were shocked on admission; one was fluid responsive not requiring inotropic support and two went on to develop shock later during their PICU stay (12/19 requiring inotropic support). Three patients 15.7% (3/19) received nitric oxide, in the setting of increased oxygen requirements without access to echocardiography, and all three survived. One patient received a partial exchange transfusion at George Hospital prior to transfer to RCWMCH PICU and survived. No other patients received any form of leukoreduction therapy, and no patients were offered ECMO as per the institution's protocol.
The most common complications during the PICU admission were seizures (n=9, 47.4%), acute kidney and liver injury (n=8, 42.1% each), and hospital-acquired infections (HAI) (n=5, 26.3%) (Table 2).
Patients who died had a significantly higher proportion of acute kidney injury than survivors (p=0.006), but there were no significant associations between other complications and mortality (Table 2). Of the HAI, there were three BSIs, two of which were confirmed central line-associated infections (CLABSI). One patient had an extended spectrum beta lactamase (ESBL) producing Klebsiella pneumoniae and one patient had Burkholderia cepacia complex. The other hospital-acquired BSI was Candida albicans. No significant association between BSI and mortality was seen (p=0.7). There were two HAIs identified from urine cultures - both grew C. albicans. There were also five new co-infections found on repeat tracheal aspirates >48 hours after admission: adenovirus (n=1), rhinovirus, (n=1) and parainfluenza virus (n=2), and one patient cultured both Staphylococcus aureus on tracheal aspirate and C. albicans on bronchoalveolar lavage.
Echocardiograms were performed on six patients (31%), none of which showed evidence of pulmonary hypertension.
Prescribing practices in PICU
Two cases had missing data and prescriptions could not be reviewed. Azithromycin was prescribed on day one of admission in 13 (68.4%) patients. There was a delay to initiation of treatment in three (15.7%) patients, who received treatment on days 4, 5 and 7 respectively. One patient died prior to initiation of azithromycin. Dosing was 10 mg/kg for all reviewed patients, according to institution protocol. Most patients received five days of treatment according to protocol while three patients had differing durations of treatment: two with four days' and one with 10 days' duration of azithromycin.
Outcomes
Median (IQR) duration of PICU admission was 6.5 (2.5 - 12.0) days, with patients receiving invasive or non-invasive mechanical ventilation for 7.0 (3.0 - 21.0) days (range 1.0 - 35.0 days). Seven (36.8%) patients with B. pertussis died.
Five of the eight patients with an admission WCC >50x109/L died, compared with two of the 11 with WCC <50 x109/L (p=0.08). Five of the seven (71.4%) patients who died received high-frequency oscillatory ventilation compared with two of the 12 who survived (p=0.06). The median (IQR) duration of ventilation in those who died compared with survivors was 3.0 (1.0 - 11.0) days v. 9 (5.3 - 25.0) days (p=0.14). Maximum median (IQR) oxygenation index during PICU admission was 142.3 (18.4 - 251.3) in those who died v.15.3 (4.7 - 28.1) in survivors (p=0.09).
HIV exposure, prematurity, increased WCC and the development of AKI were associated with mortality on univariate analysis (Tables 1 and 2). A binary logistic regression was conducted to determine the association between HIV exposure, prematurity, WCC, age and the development of AKI, with mortality. The final model was a good fit for the data (Nagelkerke R-square of 0.94), being able to correctly predict 95% of cases. None of the variables was independently associated with increased mortality (p>0.5 for all).
Discussion
Our study is the first to describe the clinical course and PICU outcomes of patients with critical pertussis admitted to a PICU in South Africa. The mortality rate amongst patients with B. pertussis infection of 36.8% was substantially higher than a previous report, which describes an overall mortality rate of 4.8% in 10 PICUs and 19 general ICUs in Australia/New Zealand.[25] Studies from two LMICs (India and Tunisia) reported mortality rates of 22% and 23% respectively.[7,26] The reason for our observed higher mortality rates is likely multifactorial, with potential contributing factors including discrepancies in healthcare services including admission criteria for PICU, socio-economic factors, differences in health-seeking behaviour, and patients presenting late in the disease process. As found in previous studies, there is an increased risk of mortality in infants <3 months of age.[10,11,16] Almost 80% of our patients were <3 months of age and 40% of those patients died. Almost 70% of patients in this cohort were completely unimmunised, with approximately 70% of those being too young to be vaccinated. A study by Muloiwa et al. found that the risk of pertussis declined with each dose of pertussis vaccine, although the reduction only reached significance after completion of the 3-dose primary vaccine schedule.[1] Our study only included two patients who had completed the primary schedule. This study's findings support the importance of public health efforts to improve vaccine coverage as well as protecting infants too young to be vaccinated through maternal immunisation against pertussis, which we identified as a highly vulnerable cohort. Vaccination against B. pertussis in the third trimester of pregnancy has been shown to be cost-effective in preventing pertussis disease in infants.[27] It is therefore hoped that the recent introduction (after the study period) of the Tetanus diphtheria and Acellular pertussis (Tdap) vaccine to the Expanded Programme on Immunisation in South Africa (EPI-SA) for pregnant mothers at 26 to 34 weeks' gestation, will lead to a reduction in both the prevalence of critical pertussis and the observed mortality of those infants too young to be vaccinated.
Hyperleukocytosis, HIV exposure, a history of prematurity and the development of AKI were significantly associated with mortality on univariate analysis; however, none of these variables was independently associated with mortality on multivariable analysis. Leucocytosis has previously been associated with pulmonary hypertension, refractory hypoxaemia, increased need for ventilation and death.[10,11] A previous study by Cherry et al. suggested monitoring WCC every 12 hours, with transfer to PICU if the WCC increased by >50% in 24 hours.[16] The rate of increase of WCC count was not evaluated in the present study but is a consideration for future research. Describing the markers of disease severity are important in our setting to identify children at risk of death or who may benefit from leukoreduction or other therapies. Only one of our patients received exchange transfusion therapy for hyperleukocytosis and this patient had a positive outcome. It has been recommended that exchange transfusion therapy should be instituted timeously before organ failure has occurred.[13,15] We recommend adopting clear unit protocols for commencing exchange transfusion in the face of hyperleukocytosis (WCC cutoff value >30) and refractory hypoxaemia.[13,14] Further audit of outcomes in such patients is imperative to guide the future care of these patients.
We could not identify any patients with pulmonary hypertension, but this complication was likely underreported, as only 31% of patients had an echocardiogram owing to resource constraints. Ideally, all patients with critical pertussis and refractory hypoxaemia should have echocardiography. Other common complications were seizures and both acute kidney and liver injury. Almost half this cohort with pertussis developed seizures. Neurological complications, including seizures and encephalopathy, are well-recognised complications of pertussis but these have previously been reported to be rare, occurring in under 10% of pertussis cases, albeit relatively more common in cases of critical pertussis.[28,29] Neuroimaging was not standardly performed in this cohort and the nature and aetiology of the neurological complications are not well understood. Similarly, AKI has been described previously as a rare complication of severe pertussis, but it is unclear why approximately 40% of our cohort developed this complication as well as liver injury.[30-32] Purposive selection of a critically ill cohort with severe pertussis may have skewed our results to suggest a greater burden of complications in our population, but our findings suggest that close monitoring of kidney and liver function is important for children admitted to PICU in our context.
Pertussis infection results in immune suppression which makes infants and children more susceptible to co-infection with secondary pathogens.[33] Rhinovirus was the most common co-respiratory viral isolate in our study, which was echoed in a prior study in Tunisia.[26] Bacterial superinfection and high procalcitonin (PCT) levels have been associated with mortality in prior studies.[34,35] We did not show an association between mortality and community- or hospital-acquired bacterial infections in children with critical pertussis, but this likely relates to the small sample size of the present study.
Only one of our patients did not receive azithromycin prior to death. Antibiotic therapy should not be delayed for diagnostic confirmation of ertussis.[36] Azithromycin has been shown to block the transmission of B. pertussis after 5 - 7 days from the beginning of treatment, but not to alter the clinical course of the disease.[36]
Up-to-date data on the burden of disease of B. pertussis is scant and is likely underreported in LMICs owing to the lack of availability of diagnostic testing for B. pertussis, especially in patients not meeting the WHO diagnostic criteria. Increased surveillance of pertussis outside of an outbreak is important to document the cyclical nature of pertussis outbreaks, to detect outbreaks early and institute preventative measures, and to offer treatment timeously. Prior studies have reported the contribution of B. holmesii/bronchisepta to the re-emergence of pertussis disease, and these require further monitoring and evaluation.[37] Furthermore, there is a need for future research on the long-term morbidity of children with critical pertussis, such as asthma and neurological impairment.[38,39]
Study limitations
This was a retrospective descriptive study, with inherent risk of sampling and reporting bias. This study was conducted at a single centre, with a small sample size and hence limited external validity and generalisability. Owing to the small sample size, a meaningful analysis of associations between pertussis disease, risk factors and complications was limited, and further studies with larger sample sizes and across multiple sites are recommended. Owing to the lack of standardisation of testing, the true prevalence of critical pertussis may have been underreported. Pulmonary hypertension was also likely underreported in our study owing to resource limitations with a lack of access to echocardiography.
Conclusion
Despite pertussis being a vaccine-preventable disease, it continues to have global relevance and carries a significant risk of mortality and morbidity, especially in infants who are not yet immunised. Describing the markers of disease severity are important in our setting to identify children potentially at higher risk of death and to ensure timeous transfer to a PICU setting. Further studies in a PICU setting, particularly from resource-constrained settings, are essential to develop evidence-based and standardised treatment protocols for the management of critical pertussis. We strongly advocate for the vaccination of mothers during pregnancy, to provide protection for those infants too young to be immunised against pertussis. We also strongly support advocacy measures to improve EPI-SA adherence among infants and children in South Africa.
Declaration. None.
Acknowledgements. The authors wish to dedicate this work to their patients. The authors wish to thank their colleagues for their invaluable contributions in drafting this manuscript.
Author contributions. HC, NN and CP contributed to the design of the study. HC and NN collected data over the study period. HC and NN drafted the manuscript which was critically revised by CP, BM and SS. BM contributed to data analysis and interpretation and critical review of the manuscript. All authors approved the final version of the manuscript.
Funding. None.
Conflict of interest. None.
References
1. Muloiwa R, Wolter N, Mupere E, et al. Pertussis in Africa: findings and recommendations of the Global Pertussis Initiative (GPI). Vaccine 2018;36(18):2385-2393. [ Links ]
2. NICD. Increase in pertussis cases in South Africa (21 Sept 2022). 2022; Available from https://www.nicd.ac.za/increase-in-pertussis-cases-in-south-africa-21-sept-2022/ [ Links ]
3. Mooi FR, Van Der Maas NA, De Melker HE. Pertussis resurgence: Waning immunity and pathogen adaptation-two sides of the same coin. Epidemiol Infect 2014;142(4):685-694. [ Links ]
4. NICD. Pertussis frequently asked questions. 2017; https://www.nicd.ac.za/assets/files/Pertussis%20FAQ_final_20170111_%20corrected%20date%20on%20bottom_30Jan2017.pdf [ Links ]
5. Bouguerra H, Hechaichi A, Atawa L, et al. Epidemiology of pertussis in Tunisia: Results of five years 2013-2017. Eur J Public Health 2019;29 (Supplement 4). [ Links ]
6. NICD. Update on increase in pertussis cases in South Africa, November 2018. https://www.nicd.ac.za/wp-content/uploads/2018/11/Update-on-increase-in-pertussis-cases-in-South-Africa.pdf [ Links ]
7. Kavitha T, Samprathi M, Jayashree M, Gautam V, Sangal L. Clinical profile of critical pertussis in children at a pediatric intensive care unit in Northern India. Indian Pediatrics 2020;57:228-231. [ Links ]
8. Birru F, Al-Hinai Z, Thani SA, Al-Mukhaini K, Al-Zakwani I, Al-Abdwan R. Critical pertussis: A multi-centric analysis of risk factors and outcomes in Oman. Int J Infect Dis 2021;107:53-58. [ Links ]
9. Winter K, Zipprich J, Harriman K, et al. Risk factors associated with infant deaths from pertussis: A case-control study. Clin Infect Dis 2015;61(7):1099-1106. [ Links ]
10. Shi T, Wang L, Du S, et al. Mortality risk factors among hospitalized children with severe pertussis. BMC Infect Dis 2021;21(1) 1057. [ Links ]
11. Berger JT, Carcillo J, Shanley TP, et al. Critical pertussis illness in children, a multicenter prospective cohort study. Pediatric Crit Care Med 2013;14(4):356-365. [ Links ]
12. Paddock CD, Sanden GN, Cherry JD, et al. Pathology and pathogenesis of fatal Bordetella pertussis infection in infants. Clin Infect Dis 2008;47(3):328-338. [ Links ]
13. Nieves D, Bradley JS, Gargas J, et al. Exchange blood transfusion in the management of severe pertussis in young infants. Pediatric Infect Dis J 2013;32(6):698-699. [ Links ]
14. Kuperman A, Hoffmann Y, Glikman D, Dabbah H, Zonis Z. Severe pertussis and hyperleukocytosis: Is it time to change for exchange? Transfusion 2014;54(6):1630-1633. [ Links ]
15. Rowlands HE, Goldman AP, Harrington K, et al. Impact of rapid leukodepletion on the outcome of severe clinical pertussis in young infants. Pediatrics 2010;126(4):816-827. [ Links ]
16. Cherry JD, Wendorf K, Bregman B, et al. An observational study of severe pertussis in 100 infants<120 days of age. Pediatric Infect Dis J 2018;37(3):202-205. [ Links ]
17. Hallbauer U, Joubert G, Goosen Y. Pertussis in children in Bloemfontein, South Africa: A 7-year retrospective review. S Afr Med J 2016;106(10):1042-1046. [ Links ]
18. World Health Organization. Surveillance standards for vaccine-preventable diseases, 2nd ed. 2018. https://www.who.int/publications/i/item/surveillance-sta1ndards-for-vaccine-preventable-diseases-2nd-edition [ Links ]
19. Martini H, Detemmerman L, Soetens O, Yusuf E, Piérard E. Improving specificity of Bordetella pertussis detection using a four-target real-time PCR. PLoS One 2017;12(4):e0175587. [ Links ]
20. Ostermann M, Bellomo R, Burdmann EA, et al. Controversies in acute kidney injury: Conclusions from a kidney disease: Improving Global Outcomes (KDIGO) Conference. Kidney Int 2020;98(2):294-309. [ Links ]
21. Squires J, McKiernan P, Squires R. Pediatric Organ Dysfunction Information Update Mandate (PODIUM) Collaborative. Acute liver dysfunction criteria in critically ill children: The PODIUM Consensus Conference. Pediatrics 2022;149:S59. [ Links ]
22. Weiss SL, Peters MJ, Alhazzani W, et al. Surviving sepsis campaign international guidelines for the management of septic shock and sepsis-associated organ dysfunction in children. Intensive Care Med 2020;46:10-67. [ Links ]
23. Horan TC, Andrus M, Dudeck MA. CDC/NHSN surveillance definition of health care-associated infection and criteria for specific types of infections in the acute care setting. Am J infect Control 2008;36(5):309-332. [ Links ]
24. Consolaro A, Giancane G, Schiappapietra B, Davi S, et al. Clinical outcome measures in juvenile idiopathic arthritis. Pediatric Rheumatol 2016;14:1-8. [ Links ]
25. Straney L, Schibler S, Ganeshalingham A, et al. Burden and outcomes of severe pertussis infection in critically ill infants. Pediatric Crit Care Med 2016;17(8):735-742. [ Links ]
26. Borgi A, Menif K, Belhadj S, et al. Predictors of mortality in mechanically ventilated critical pertussis in a low-income country. Mediterranean J Hematol Infect Dis 2014:6(1). [ Links ]
27. Munoz FM, Bond NH, Maccato M, et al. Safety and immunogenicity of tetanus diphtheria and acellular pertussis (Tdap) immunization during pregnancy in mothers and infants: A randomized clinical trial. JAMA 2014;311(17):1760-1769. [ Links ]
28. Abu-RayaB, Bettinger JA, Vanderkooi OG, et al. Burden of children hospitalized with pertussis in Canada in the acellular pertussis vaccine era, 1999-2015. J Pediatric Infect Dis Soc 2020;9(2):118-127. [ Links ]
29. Anh Tuan T, Xoay TD, Thuy PTB, Ouellette Y. Cerebral spinal fluid positive pertussis encephalopathy in infants. SAGE Open Medical Case Reports 2021;9:2050313X211052446. [ Links ]
30. Madden I, Roumenina LT, Langlois-Meurinne H, et al. Hemolytic uremic syndrome associated with Bordetella pertussis infection in a 2-month-old infant carrying a pathogenic variant in complement factor H. Pediatric Nephrol 2019;34:533-537. [ Links ]
31. Saida K, Ogura M, Kano Y, et al. Treatment of hemolytic uremic syndrome related to Bordetella pertussis infection-is plasma exchange or eculizumab use necessary? BMC Nephrol 2018;19:1-4. [ Links ]
32. Chaturvedi S, Licht C, Langlois V. Hemolytic uremic syndrome caused by Bordetella pertussis infection. Pediatric Nephrol 2010;25:1361-1364. [ Links ]
33. Bellettini CV, de Oliveira AW, Tusset C, et al. Clinical, laboratorial and radiographic predictors of Bordetella pertussis infection. Revista Paulista de Pediatria 2014;32:292-298. [ Links ]
34. Coquaz-Garoudet M, Ploin D, Pouyau R, et al. Malignant pertussis in infants: factors associated with mortality in a multicenter cohort study. Ann Intensive Care 2021;11(1):70. [ Links ]
35. Wu X-Y. The role of hyperleukocytosis in predicting the severity of pertussis. 2020. https://doi.org/10.21203/rs.3.rs-132275/v1 [ Links ]
36. Altunaiji SM, Kukuruzovic R, Curtis, N, Massie J. Cochrane Review: Antibiotics for whooping cough (pertussis). Evidence-based child health: A Cochrane Rev J 2012;7(3):93-956. [ Links ]
37. Mir-Cros A, Codina G, Martín-Gómez MT, et al. Emergence of Bordetella holmesii as a causative agent of whooping cough, Barcelona, Spain. Emerging Infect Dis 2017;23(11):1856. [ Links ]
38. de Greeff SC, van Buul LW, Westerhof A, et al. Pertussis in infancy and the association with respiratory and cognitive disorders at toddler age. Vaccine 2011;29(46):8275-8278. [ Links ]
39. Berger JT, Villalobos ME, Clark AE, et al. Cognitive development one year after infantile critical pertussis. Pediatr Crit Care Med 2018;19(2):89-97. [ Links ]
 Correspondence:
Correspondence:
C Procter
claire.procter@uct.ac.za
Received 24 March 2024
Accepted 25 February 2025
Contribution of the study
This study demonstrated high mortality rates in infants with critical pertussis under the age of 3 months, especially in infants who are unimmunized or partially immunized. We therefore recommend vaccination against Bordetella Pertussis in the third trimester of pregnancy and stress the importance of strengthening current immunization programmes. Our study highlights the importance of describing the markers of disease severity, such as hyperleukocytosis, especially in resource constrained settings, in order to ensure timeous transfer to PICU.













