Services on Demand
Journal
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Child Health
On-line version ISSN 1999-7671Print version ISSN 1994-3032
S. Afr. j. child health vol.18 n.3 Pretoria Sep. 2024
https://doi.org/10.7196/sajch.2024.v18i3.1767
RESEARCH
Similar mortality rates for early and late group B streptococcal disease among newborns at Groote Schuur Hospital in Cape Town
B BrowneI, II; A KhumaloIII; L TookeIV, V
IFCPaed(SA), MMed, DCH, DTM&H; Department of Paediatrics, Red Cross War Memorial Children's Hospital, Cape Town, South Africa
IIFCPaed(SA), MMed, DCH, DTM&H; Department of Paediatrics and Child Health, University of Cape Town, South Africa
IIIFCPath(SA) Micro; Department of Medical Microbiology, Groote Schuur Hospital, University of Cape Town, South Africa
IVFCPaed(SA), Cert neonatology(SA); Department of Paediatrics and Child Health, University of Cape Town, South Africa
VFCPaed(SA), Cert neonatology(SA); Department of Neonatology, Groote Schuur Hospital, Cape Town, South Africa
ABSTRACT
BACKGROUND: Invasive group B streptococcal disease (iGBS) is an important cause of morbidity and mortality in newborns globally and is more common in Sub-Saharan Africa and South Africa. Few studies describe newborn iGBS cases in the Western Cape, leaving limited data to guide local prevention and treatment strategies
OBJECTIVE: To describe all cases of newborn iGBS at Groote Schuur Hospital (GSH), Western Cape, between January 2015 and December 2020
METHODS: A retrospective review was conducted of all blood or cerebrospinal fluid-positive cases of newborn iGBS between January 2015 and December 2020
RESULTS: Thirty-five cases of iGBS were included in the study, 51% with early-onset disease (EoD) and 49% with late-onset disease (LoD). The median gestational age at birth was 27 and 31.5 weeks in the LoD and EoD groups, respectively. The median birth weight was 900 g and 1 812 g in the LoD and EoD groups, respectively. Meningitis was more common in the LoD group and pneumonia was more common in the EoD group. The overall mortality rate was 23%, with similar mortality rates in the EoD and LoD groups (22% and 24%, respectively). Invasive GBS disease accounted for a cumulative 274 intensive care unit days during the study period
CONCLUSION: Invasive GBS disease in newborns at GSH is associated with a high mortality rate, with this study showing similar mortality rates between the EoD and LoD groups
Streptococcus agalactiae, also known as Lancefield group B Streptococcus (GBS), is a leading cause of severe illness among infants.[1,2] In 2020, iGBS caused 46 000 stillbirths, 91 900 infant deaths and 518 000 preterm births globally.[3] In surviving children, GBS septicaemia and meningitis increase the risk for neurodevelopmental impairment (NDI).[4] Sub-Saharan Africa carries a disproportionately high burden of iGBS compared to the rest of the world.[5]
Invasive GBS disease in newborns is classified into early-onset disease (EoD) and late-onset disease (LoD), occurring between days 0 - 6 and 7 - 90 of life, respectively. EoD is acquired vertically and often presents as septicaemia or pneumonia, while LoD frequently causes septicaemia and meningitis.[2] Important risk factors in infants include maternal GBS colonisation of the gastrointestinal and genitourinary tract, preterm birth, very low birth weight, prolonged rupture of membranes (ROM) and maternal HIV infection.[2,5,6] While effective vaccines to prevent iGBS in newborns are anticipated, they are not yet available.[7] Routine antenatal testing for maternal GBS colonisation using vaginal swabs, combined with intrapartum antibiotic administration for those who test positive, is an effective but expensive strategy to prevent EoD in newborns.[8]
There are limited studies on the burden of iGBS in South African (SA) infants. Surveillance reports from the Gauteng Province indicate an incidence of 2.4 - 3 per 1 000 live births, higher than the estimated 1.1 in Sub-Saharan Africa and 0.53 globally.[9-11] Mortality rates in SA are also high, with one Gauteng study reporting a rate of 18%, almost double the global estimates.[11,12] A retrospective study in the Metro East district of Cape Town reported an iGBS crude incidence of 0.67 and a mortality rate of 6%; however, the authors noted that this was likely underestimated owing to incomplete records for deceased patients.[13] Studies from both SA and other countries report higher mortality rates in EoD compared with LoD. In addition, survivors in SA and other low-and middle-income countries (LMICs) have an increased risk of neurodevelopmental impairment (NDI).[14] Key factors contributing to GBS disease in SA include the high HIV prevalence of 30% among pregnant women,[15] a high GBS carriage rate and sub-optimal intrapartum antibiotic prophylaxis,[16,17]
Few publications describe newborn iGBS cases in the Western Cape, resulting in limited data for improving local preventive and treatment strategies. Therefore, this study aims to describe all cases of iGBS in newborns over six years at Groote Schuur Hospital (GSH).
Methods
Groote Schuur Hospital is a public hospital in Cape Town, Western Cape, with a specialised neonatal unit that provides tertiary care for a drainage area with ~40 000 deliveries per annum. The unit has 75 neonatal beds, including 20 for intensive care. The unit admits up to 2 000 babies a year, with ~500 infants having very low birth weight (VLBW). While high-risk deliveries are prioritised at GSH, approximately 15% of admissions are outborn and referred from other primary and secondary facilities.
Standard of care
Universal antenatal testing for maternal GBS colonisation is not performed in the public health service in SA. At GSH, the decision to perform a blood culture and commence antibiotic therapy is based on the newborn having two or more risk factors for early-onset sepsis (<72 hours of life).[18] Risk factors are categorised as 'antenatal, 'postnatal, or 'absolute. Standard neonatal septic risk factors and the antibiotic policy at GSH during the review period are detailed in Appendix A (https://www.samedical.org/file/2284). Blood cultures, are performed on all identified infants along with a C-reactive protein (CRP) between 36 and 48 hours after antibiotic initiation. Additionally, cerebrospinal fluid (CSF) analysis is conducted on all surviving infants with blood culture-confirmed iGBS or those with clinical suspicion of meningitis.
Study design
A retrospective review of medical records was conducted for all cases of newborn iGBS at GSH between January 2015 and December 2020. An iGBS case was defined as infants younger than 90 days with a positive blood or CSF culture for GBS. Data from National Health Laboratory Services (NHLS) laboratory information system was used to identify all iGBS cases during the study period.
The NHLS uses the BacT/Alert (Organon Teknika Corp, USA) instrument, an automated microbial detection system, to incubate and flag positive blood cultures. Positive cultures are then Gram-stained and sub-cultured onto appropriate media. Further organism identification and susceptibility testing are performed using Vitek 2 (BioMérieux, France). CSF specimens are Gram-stained and cultured onto appropriate media according to laboratory standard operating procedures. Microorganisms are further identified using Lancefield grouping and Vitek 2 (BioMérieux, France) for susceptibility testing.
Once cases were identified, data were collected on a case report form using Microsoft Excel. Data analysis was descriptive. Results are presented as medians with associated interquartile ranges (IQR). Where applicable, R version 4.05 was used to conduct the Mann-Whitney and Fisher's exact tests to assess differences in continuous and categorical variables, respectively.
Ethics clearance was obtained from the University of Cape Town Departmental Research Committee (ref. no. HREC 659/2020, Appendix B; https://www.samedical.org/file/2284).
Results
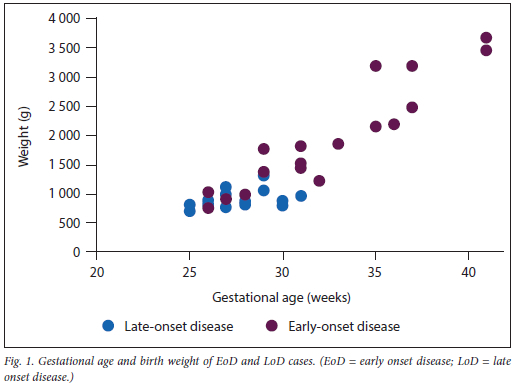
There were 36 cases of iGBS during the study period. One record was incomplete and excluded from the analysis. Of the remaining 35 cases, 18 (51%) were EoD and 17 (49%) were LoD. The average time to culture positivity was 9.2 hours in the EoD group and 6.8 hours in the LoD group. All GBS isolates were sensitive to penicillin. Eleven (31%) cases involved infants born at primary and secondary centres who were referred to GSH for tertiary care. The 24 inborn cases resulted in an incidence rate of 0.72 for deliveries at GSH. EoD cases occurred across all weights and gestational ages, while LoD cases were found only in VLBW preterm infants (Fig. 1). There was a difference between the EoD and LoD groups for both weight and gestational age (p<0.001). Table 1 presents comparisons of characteristics between the EoD and LoD groups.
Early-onset disease
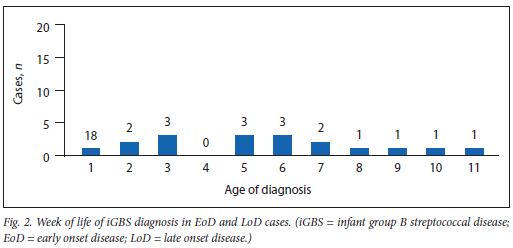
All 18 EoD cases were diagnosed in the first 24 hours of life, except for one diagnosed at 72 hours. The median birth weight and gestational age in the EoD group were 1 812 g and 31.5 weeks, respectively. Fourteen infants (78%) were born preterm (before 37 completed weeks gestation). Four mothers received no antenatal care, and 4 mothers had GBS cultured from urine specimens collected before delivery.
Among the EoD patients, 13 (72%) were vaginally delivered, while five (28%) were delivered via caesarean section (C/S). Of the five C/S, three were performed on mothers who had both spontaneous preterm labour (SPTL) and spontaneous rupture of membranes (ROM), while two were performed on mothers who were not in labour and had no ROM or other risk factors. Of the 13 vaginal deliveries, 10 had SPTL, and two had prolonged ROM. None of the infants in the EoD group received intrapartum ampicillin prophylaxis.
Late-onset disease
All cases in the LoD group were diagnosed between weeks 2 and 11 of life (Fig. 2). The median birth weight and gestational age were 900 g and 27 weeks, respectively. All infants were born very preterm (before 32 completed weeks gestation). Eleven (64%) of them were delivered vaginally, and six (36%) were delivered by C/S. In four cases (24%), no antenatal care was provided.
HIV exposure
Maternal HIV infection status and recent viral loads (within 12 weeks before delivery) were available for all cases. Seven (20%) infants were HIV-exposed, with three in the EoD group and four in the LoD group. Two of these infants were classified as 'high risk' owing to a recent maternal HIV viral load exceeding 1 000 copies/ mL. All HIV-exposed newborns received post-exposure prophylaxis with nevirapine (NVP), and the two high-risk infants received NVP and zidovudine. All cases had a negative HIV polymerase chain reaction (PCR) test at birth, and all survivors remained negative on repeat testing carried out at 6 - 10 weeks of life.
Primary site of infection and mortality
In the EoD group, the predominant presentations were septicaemia without a focus (11 cases, 61%), and bacteraemic pneumonia (seven cases, 39%). One of the newborns with pneumonia also had meningitis. In the LoD group, the predominant presentations were septicaemia without a focus (eight cases, 47%), followed by meningitis (five cases, 29%), bacteraemic pneumonia (three cases, 18%) and necrotizing enterocolitis (NEC) (one case, 6%). Of the meningitis cases in the LoD group, two cultured GBS from CSF, while the remaining three had suggestive CSF cell counts. The prevalence of meningitis was 5.3 times higher in the LoD group compared with the EoD group, although this difference was not statistically significant (95% CI: 0.69 - 40.8).
In the EoD group, four (22%) infants died as a result of iGBS. Among these, two had bacteraemic pneumonia and two had septicaemia without a focus. Three of these infants died within the first 24 hours of life, and one died on day 9. In the LoD group, four (24%) infants died because of iGBS: one had pneumonia, one had NEC and two had septicaemia. Of the eight deaths, two infants were HIV-exposed, one in the EoD and one in the LoD group.
The LoD newborn with NEC had a gestational age of 31 weeks and a birth weight of 985 g. The infant deteriorated on day 11 of life with clinical and radiological signs of NEC. The infant was managed medically but developed a coagulopathy and a large intracranial parenchymal haemorrhage 24 hours later, leading to death. Additionally, two other infants with EoD and one infant with LoD later developed NEC after recovering from iGBS on days 8, 17 and 67 of life, respectively. An infant with LoD diagnosed on day 37 of life recovered from iGBS but died 7 weeks later in the hospital with no clear cause, classified as sudden unexpected death in infancy.
Clinical presentation and care
The most common presenting clinical features were respiratory distress (88.6%), Shock (28.6%) and apnoea (22.9%). Seventeen (94%) infants in the EoD group required intensive care, accumulating 158 days. Fifteen (88%) infants in the LoD group required intensive care, accumulating 116 days. Invasive ventilation was needed for nine cases in both the EoD (50%) and LoD (53%) groups. Inotropic support was required in seven (39%) cases in the EoD group and two (12%) cases in the LoD group.
The cohort included one set of twins (monochorionic diamniotic) with LoD, born at 30 weeks gestation, with twin A and twin B weighing 795 g and 900 g, respectively. They presented on days 35 and 36 of life with pneumonia and septicaemia. Twin A required invasive ventilation for 5 days, and both survived to discharge.
Laboratory investigations
CRP measurements were taken from 36 - 48 hours of life. In the EoD group, 11 cases had a CRP measurement, with a median of 53 mg/dL, and four (36%) cases had CRP levels below 10 mg/dL. In the LoD group, 13 (76%) cases had CRP measurements, with a median of 83 mg/dL, and none below 10 mg/dL. The difference between the CRP levels in the EoD and LoD groups was not statistically significant (p=0.104).
Discussion
This study describes a high mortality rate of 23% in newborns due to iGBS, higher than in cohorts from Johannesburg (18%), Soweto (16.9%) and previous estimates from Metro East, Cape Town (6%).[6,12,13] Interestingly, the mortality rates in the EoD (22%) and LoD (24%) groups in our study were similar. This contrasts with other studies, which have shown higher mortality rates in EoD compared with LoD. A previous SA study reported EoD and LoD mortality rates of 22.7% and 12.5%, respectively,[12] while international findings show EoD and LoD mortality rates of 12.1% and 6.8%, respectively.[11] This difference in our findings may be partly explained by the significantly lower median gestational age and birthweight in the LoD group than in the EoD group (p<0.001). Our findings of an increased prevalence of meningitis in newborns with LoD compared with EoD align with previous studies,[12] although our sample size is too small to determine statistical significance.
The HIV exposure prevalence in our cohort (17%) is similar to the reported antenatal HIV prevalence in the Western Cape (17.9%).[15] However, we did not find an association between HIV exposure and LoD, which has been well-documented in other SA studies.[6] This lack of association is likely due to the small sample size in our study.
Recent studies conducted in LMICs indicate a high risk of NDI in newborns with iGBS, particularly in those with septicaemia without a focus or meningitis as the primary site of infection.[14] In our cohort, septicaemia without a focus and/or meningitis occurred in 71% of cases. Further studies are needed to assess the risk for NDI in iGBS survivors in our setting. Notably, 91% of iGBS cases required NICU admission, for a cumulative 274 days, which is important for a country with limited intensive care resources.
A correlation between iGBS and NEC has been previously described,[19] and in our study, one infant with LoD presented with NEC, while three others developed NEC 1 - 4 weeks after the iGBS diagnosis. Further research is needed to explore this association in our setting.
In this cohort, CRP levels were higher in LoD compared with EoD, although this difference was not statistically significant. CRP values were less than 10 mg/dL in 36% of EoD cases, but none of the LoD cases. This is in keeping with previous studies indicating that CRP has reduced sensitivity in newborns with early-onset sepsis,[20] which may have important clinical implications for the use of CRP to rule out EoD.
Limitations
The limitations of this study include the relatively small number of iGBS cases, which made it difficult to determine the statistical significance of various risk factors and outcomes. Furthermore, the study was limited to cases admitted to a single tertiary unit, excluding infants who developed iGBS after discharge from GSH and were treated at other facilities. Lastly, stillbirths caused by GBS were not included in the analysis.
Conclusion
Invasive GBS disease in newborns at GSH is associated with a high mortality rate, with a similar burden of both LoD and EoD. Further studies are needed to assess the burden of iGBS in the Western Cape and SA, and to better understand the risk factors contributing to mortality and poor neurodevelopmental outcomes.
Declaration. None.
Acknowledgements. None
Author contributions. BB contributed to the study design, protocol development, data collection and analysis as well as the main article write-up. LT contributed to the study design, protocol development, data analysis and article write-up. AK contributed to the protocol development, data collection and review of the article.
Funding. None.
Data availability statement. The datasets generated and analysed during the current study are available from the corresponding author upon reasonable request.
Conflicts of interest. None.
References
1. Glaser P, Rusniok C, Buchrieser C, et al. Genome sequence of Streptococcus agalactiae, a pathogen causing invasive neonatal disease. Mol Microbiol 2002;45(6):1499-1513. https://doi.org/10.1046/j.1365-2958.2002.03126.x [ Links ]
2. Seale AC, Bianchi-Jassir F, Russell NJ, et al. Estimates of the burden of group B streptococcal disease worldwide for pregnant women, stillbirths, and children. Clin Infect Dis 2017;65(Suppl 2):S200-219. https://doi.org/10.1093/cid/cix664 [ Links ]
3. Gonçalves BP, Procter SR, Paul P, et al. Group B Streptococcus infection during pregnancy and infancy: Estimates of regional and global burden. Lancet Glob Health 2022;10(6):e807-819. https://doi.org/10.1016/s2214-109x(22)00093-6 [ Links ]
4. Kohli-Lynch M, Russell NJ, Seale AC, et al. Neurodevelopmental impairment in children after group B streptococcal disease worldwide: Systematic review and meta-analyses. Clin Infect Dis 2017;65(Suppl 2):S190-199. https://doi.org/10.1093/cid/cix663 [ Links ]
5. Kwatra G, Cunnington MC, Merrall E, et al. Prevalence of maternal colonisation with group B Streptococcus: A systematic review and meta-analysis. Lancet Infect Dis 2016;16(9):1076-1084. https://doi.org/10.1016/s1473-3099(16)30055-x [ Links ]
6. Cutland CL, Schrag SJ, Thigpen MC, et al. Increased risk for group B Streptococcus sepsis in young infants exposed to HIV, Soweto, South Africa, 2004-20081. Emerg Infect Dis 2015;21(4):638-645. https://doi.org/10.3201/eid2104.141562 [ Links ]
7. Madhi SA, Anderson AS, Absalon J, et al. Potential for maternally administered vaccine for infant group B Streptococcus. N Engl J Med 2023;389(3):215-227. https://doi.org/10.1056/nejmoa2116045 [ Links ]
8. Fairlie T, Zell ER, Schrag S. Effectiveness of intrapartum antibiotic prophylaxis for prevention of early-onset group B streptococcal disease. Obs Gynecol 2013;121(3):570-577. https://doi.org/10.1097/aog.0b013e318280d4f6 [ Links ]
9. Quan V, Verani JR, Cohen C, Von Gottberg A, et al. Invasive group B streptococcal disease in South Africa: Importance of surveillance methodology. PLoS One 2016;11(4):1-10. https://doi.org/10.1371/journal.pone.0152524 [ Links ]
10. Sinha A, Russell LB, Tomczyk S, et al. Disease burden of group B Streptococcus among infants in sub-Saharan Africa: A systematic literature review and meta-analysis. Pediatr Infect Dis J 2016;35(9):933-942. https://doi.org/10.1097/inf.0000000000001233 [ Links ]
11. Edmond KM, Kortsalioudaki C, Scott S, et al. Group B streptococcal disease in infants aged younger than 3 months: Systematic review and meta-analysis. Lancet 2012;379(9815):547-556. https://doi.org/10.1016/s0140-6736(11)61651-6 [ Links ]
12. Danger Z, Lala SG, Cutland CL, et al. Burden of invasive group B Streptococcus disease and early neurological sequelae in South African infants. PLoS One 2015;10(4):1-13. https://doi.org/10.1371/journal.pone.0123014 [ Links ]
13. Frigati L, Merwe JL Van Der, Harvey J, et al. A retrospective review of group B streptococcal infection in the Metro East area of the Western Cape province: 2010 to 2011. S Afr J Infect Dis 2015;29(1):33-36. https://hdl.handle.net/10520/EJC155247 [ Links ]
14. Paul P, Chandna J, Procter SR, et al. Neurodevelopmental and growth outcomes after invasive Group B Streptococcus in early infancy: A multi-country matched cohort study in South Africa, Mozambique, India, Kenya, and Argentina. Lancet eClin Med 2022;47:101358. https://doi.org/10.1016/j.eclinm.2022.101358 [ Links ]
15. South African Department of Health. The 2019 National Antenatal HIV Sentinel Survey (ANCHSS) Key Findings. 2021. [ Links ]
16. National Department of Health (NDoH). Guidelines for Maternity Care in South Africa: A manual for clinics, community health centres and district hospitals. South African Government. 2015:174. Maternal-Care-Guidelines-2015_FINAL-21.7.15.pdf [ Links ]
17. Dangor Z, Lala SG, Madhi SA. Preventing invasive group B Streptococcus (GBS) disease in South African infants: Time for change. S Afr J Child Health 2015;9(3):69-70. http://dx.doi.org/10.7196/SAJCH.8575 [ Links ]
18. Joolay Y, Horn A. Neonatal Guidelines and Drug Doses. 5th ed. Cape Town: Department of Neonatal Medicine, Groote Schuur Hospital. 2020. [ Links ]
19. Stafford IA, Rodrigue E, Berra A, et al. The strong correlation between neonatal early-onset group B Streptococcal disease and necrotizing enterocolitis. Obs Gynecol Reprod Biol 2018;223:93-97. https://doi.org/10.1016/j.ejogrb.2018.02.024 [ Links ]
20. Hofer N, Müller W. An update on the use of c-reactive protein in early-onset neonatal sepsis : Neonatology 2012;102:25-36. https://doi.org/10.1159/000336629 [ Links ]
 Correspondence:
Correspondence:
B Browne
bradsbrowne@gmail.com
Submitted 29 November 2023
Accepted 21 June 2024













