Services on Demand
Journal
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
Journal of the Southern African Institute of Mining and Metallurgy
On-line version ISSN 2411-9717Print version ISSN 2225-6253
J. S. Afr. Inst. Min. Metall. vol.124 n.12 Johannesburg Dec. 2024
https://doi.org/10.17159/2411-9717/bm20/2024
BATTERY MATERIAL PAPER
Removal of iron from vanadium redox flow battery electrolyte using ion exchange
M. Makonese; K.C. Sole
Department of Materials Science and Metallurgical Engineering, University of Pretoria, South Africa. ORCiD: K.C. Sole: http://orcid.org/0000-0003-4707-1060; M. Makonese: http://orcid.org/0000-0002-9210-7352
ABSTRACT
Bushveld Belco is commissioning a plant for production of electrolyte used in vanadium redox flow batteries. Owing to the source material, the electrolyte is contaminated with Fe, at concentrations up to 140 mg/L, which negatively affects battery performance. This study examined the use of ion exchange to reduce the Fe concentration to the target value of < 100 mg/L from a feed solution containing 100 g/L V in 4 M H2SO4. Four resins with possible application were identified: Puromet MTS9570, Puromet MTX7010, Puromet MTS9500, and Puromet MTC1600H. Puromet MTS9570 outperformed the other three resins in all batch experiments with respect to loading capacity and selectivity over V, and was evaluated for Fe removal in fixed-bed column loading tests. Based on these data, preliminary sizing and design of a full-scale column was undertaken. Owing to the difficulty of eluting this resin, a philosophy of discarding the resin on loading to full capacity was considered. These data revealed that, despite the high selectivity of this resin for Fe over V, it would be too expensive and impractical to use this resin for full-scale operation under such conditions. It is recommended to remove iron earlier in the flowsheet prior to generation of the 4 M H2SO4 electrolyte.
Keywords: Vanadium redox flow battery, iron, ion exchange, Puromet MTS9570
Introduction
Vanadium redox flow batteries (VRFB) are currently the world's most promising technology for stationary energy storage. These batteries are widely used for solar energy applications, and can assist in providing a renewable energy solution that is crucial for reducing the global carbon footprint (Wang et al., 2023). As fossil fuels drive global warming, countries around the world, including South Africa, are advancing research in renewable energy. This will not only help combat climate change, but could assist in ensuring reliable electricity supply for the people of South Africa (Akinbami et al., 2021). South Africa is currently the third-largest vanadium producer in the world (Bushveld Minerals, 2024). Value addition by production of VRFB electrolyte from primary V products can contribute to a carbon-free future. It is therefore imperative that VRFB technology be given more attention in this country.
The most common VRFB system is based on the VO2+/VO2+ and V2+/V3+ systems (Chace et al., 2020). The catholyte and anolyte are fed through two separate half-cells within the battery. Each half-cell has an electrode and a bipolar plate. The two half-cells are separated by a membrane that serves two functions: firstly, it allows for selective exchange of ions between the two electrolytes, which completes the cell in the battery and allows for the passage of current; secondly, the membrane prevents cross-contamination, i.e., mixing of the two electrolytes, which prevents short-circuiting in the battery that would otherwise lead to current overload in the battery, with resulting overheating and malfunction (Prifti et al., 2012).
The following reduction and oxidation reactions occur at the cathode and anode, respectively:
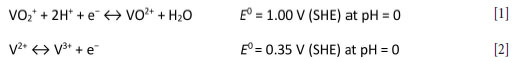
The overall electrochemical reaction is given by:
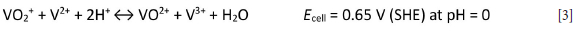
This reaction produces the potential difference in the battery that generates electricity that can be sent to a power grid or used to turn wind turbines for electricity production. Multiple VRFB stacks can be electrically connected to form a large array for applications that require more energy (Kim et al., 2015).
Bushveld Energy, under their subsidiary Bushveld Belco, is the only South African producer of electrolyte used in VRFB. The starting raw material for this electrolyte is magnetite ore, which contains 0.3% V (Bushveld Minerals, 2024). The main impurity in the electrolyte is Fe, which has certain detrimental effects on the function and performance of VRFB. The electrolyte comprised 100 g/L V, 140 mg/L Fe in 4 M H2SO4. The Bushveld specification prescribes a maximum of 100 mg/L Fe per 100 g/L V for optimal battery performance.
Ion exchange (IX) is extensively used in hydrometallurgy to purify aqueous solutions at low concentrations of impurities (< 500 mg/L) (Sole et al., 2017), so this technology was evaluated for removal of Fe from the Bushveld electrolyte.
Effects of iron in vanadium electrolyte
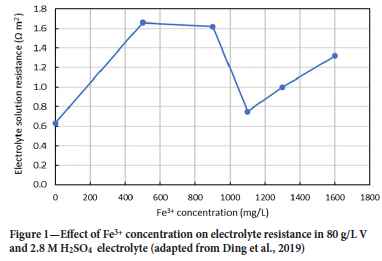
Effect of Fe on electrolyte resistance Ding et al. (2019) investigated the effect of Fe3+ concentration on the resistance of a VRFB catholyte (Figure 1). The presence of small amounts of Fe3+ (500 mg/L) resulted in a threefold increase in the electrical resistance of the electrolyte from 0.63 Ω cm2 to 1.66 Ω cm2. The resistance did, however, decrease between 500 mg/L Fe to 1100 mg/L Fe, reaching approximately 0.75 Ω m2 at 1100 mg/L Fe. Although this decrease in resistance was recorded, it should be noted that all resistances measured with Fe3+ in the electrolyte were higher than when no Fe3+ was present. A higher electrolyte resistance means that the V ions are restricted in their transport to the cathode to participate in the cathodic electrochemical reactions: a resistive force hinders the movement of the ions, which inhibits the electrochemical reaction and compromises battery performance and efficiency.
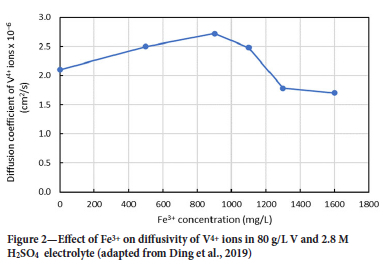
Effect of Fe3+ on diffusivity of V4+ ions Ding et al. (2019) also investigated the effect of Fe3+ concentration on the diffusivity of V4+ ions in the electrolyte (Figure 2). Concentrations below 900 mg/L Fe3+ had a positive effect on the diffusivity of V4+ ions. The diffusion coefficient of the V4+ ions increased from 2.06 cm2/s at 0 mg/L Fe3+ to 2.72 cm2/s at 900 mg/L Fe3+. This result indicates that a small addition of Fe3+ ions in the electrolyte allowed the V4+ ions to rapidly diffuse to the cathode to participate in the cathodic electrochemical reaction shown in Equation [1] (Ding et al., 2019). However, as the Fe3+ concentration increased above 900 mg/L, a decrease in diffusivity of the V4+ ions was observed: at 1300 mg/L Fe3+, the diffusion coefficient was 1.78 cm2/s, which is much lower than when there was no Fe in the electrolyte. This finding means that V4+ ions diffused slowly to the cathode to participate in the cathodic reaction, which would compromise efficiency of a battery. The Fe3+ concentration should therefore be kept low enough such that the presence of the Fe3+ ions is helpful to V ion diffusivity.
Iron side-reactions
The presence of Fe as Fe3+ in the electrolyte can also have a direct effect on the cathodic electrochemical reaction. Fe3+ ions can participate in side-reactions and be reduced to Fe2+ (Ding et al., 2019). There is competition for electrons between the Fe3/ Fe2+ reduction and the cathodic reaction (Equation [1]), thereby reducing current efficiency of the battery.
Experimental
Electrolyte generation
The electrolyte was generated as per Bushveld specifications by mixing 300 mL of 4 M H2SO4 (CP grade) and 50 g of Vametco-produced V2O3 in a 1 L beaker. The following reaction occurs:
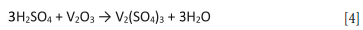
The mixture was agitated using an overhead stirrer and 50% H2O2 was added dropwise until all V2O3 had dissolved, according to the following reaction:
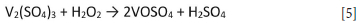
On average, a 1.6x stoichiometric excess of H2O2 was required to dissolve all V2O3. Addition of H2O2 caused a highly exothermic reaction, so the electrolyte was cooled before use. The electrolyte typically assayed 100 g/L V, 140 mg/L Fe, and 15 mg/L Cr, with the impurities originating from the V2O3.
Construction of electrolyte E-pH diagrams
To determine the speciation of Fe and V in the electrolyte, E-pH diagrams were constructed using The Geochemist's Workbench software (The Geochemist's Workbench, 2023).
Resin screening batch test methodology
Four resins were identified that are used for removal of Fe in various hydrometallurgical applications:
> Puromet MTS9570: A polystyrenic chelating resin containing phosphonic- and sulfonic-acid groups (R-SO2(OH)PO(OH)2; capacity 0.97 eq/L Fe);
> Puromet MTX7010: A polystyrenic macroporous resin solvent-impregnated with di(2-ethyl-hexyl)phosphonic acid functionality (P(OR)2OOH; capacity 0.40 eq/L Zn);
> Puromet MTC1600H: A polystyrenic macroporous strong-acid cationic resin (R-SO2(OH); capacity 2.3 eq/L);
> Puromet MTS9500: A polystyrenic macroporous aminomethylphosphonic-acid chelating resin (R-CH2-N-CH2-P(ONa)2; capacity 1.3 eq/L Ca).
Screening batch tests were carried out to evaluate the relative performance of these resins. The ideal resin would selectively extract most Fe from the electrolyte, leaving V in solution. The screening batch tests were carried out by adding 20 mL of the electrolyte and 1 mL of wet-tapped resin to a 100 mL conical flask. The flask was agitated for 24 h using a shaking table. The electrolyte was then filtered to separate it from the resin beads. The barren solution was assayed in duplicate, and the metal loadings calculated by difference. All aqueous analyses were carried out using inductively coupled plasma optical emission spectroscopy.
Isotherm loading test methodology
The two most promising resins identified from the screening tests, Puromet MTS9570 and Puromet MTX7010, were subjected to isotherm loading tests to determine their capacities for Fe under these aqueous conditions. The tests were performed at 25°C and 40°C, which is the maximum recommended operating temperature of these resins. Various volumes of electrolyte (10 mL to 600 mL) were added to eight conical flasks, each with 1 mL of resin. The solution:resin ratio (S/R) varied from 10:1 to 600:1. The flasks were shaken at 800 rpm for 24 h, after which the electrolyte was filtered to remove the resin beads and then assayed.
Column test methodology
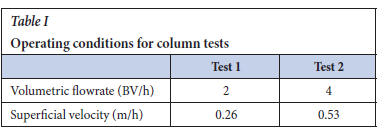
Based on the isotherm test results, Puromet MTS9570 was identified as the most suitable resin for this application. Fixed-bed column loading tests were conducted in a glass column with a height of 258 mm and inner diameter of 22.2 mm. A wet-tapped resin volume of 50 mL was employed, corresponding to a bed height of 140 mm and aspect ratio of 6.3. The electrolyte was fed downflow through the resin bed. The flowrate was manually controlled according to the output volume collected with time. Operating conditions are shown in Table I.
Results and discussion
Electrolyte E-pH diagrams
Figures 3 and 4, respectively, show the E-pH diagrams for V and Fe under the conditions of the electrolyte. The activities of Fe, V, and the sulfate anion were taken as approximately 0.0003, 0.2, and 0.04, respectively, considering the high ionic strength of 4 M H2SO4 electrolyte. These values are derived from their molar concentrations and activity coefficients estimated at 0.1 due to significant interaction of the ions.
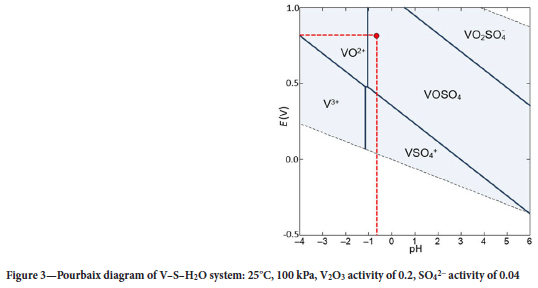
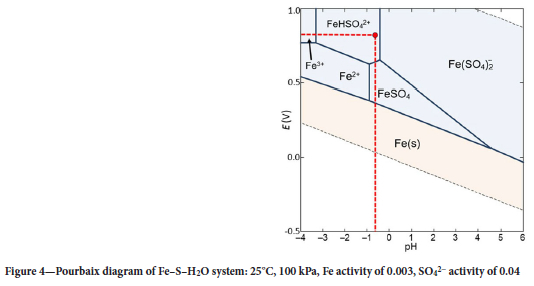
The average potential of the electrolyte was measured using an Ohaus ST20R ORP electrode as 0.8 V (SHE). The first proton of H2SO4 is fully dissociated (Ka1 = 1 x 103); the second is weakly dissociated (Ka2 = 1.2 x 10-2) (LibreTexts Chemistry, 2020). The proton concentration was hence calculated as [H+] = 1.012 mol/L. This means that 4 M H2SO4 has an approximate pH of -0.6. At these values of potential and pH (red dotted lines in Figures 3 and 4), V exists as the neutral VOSO4 species in the electrolyte. This accords with the expected reaction given in Equation [5]. Li et al. (2023) also concluded that the neutral VOSO4 species forms at high SO42-concentrations. Fe is expected to exist as the FeH(SO4)2+ cation under these high-acidity conditions. These speciation data suggested that use of a cation-exchange resin may offer the opportunity to load the cationic Fe species in preference to the neutral V species.
Resin loading results
Screening batch tests
Figure 5 shows the average extractions of Fe and V for the four resins. Puromet MTS9570 offered the highest Fe extraction of 65% under the experimental conditions employed; however, this resin, together with Puromet MTS9500, also gave the highest V extraction of 13%. Puromet MTX7010 exhibited only 38% Fe extraction, but gave the lowest V extraction of 7%. Puromet MTC1600H showed the lowest Fe extraction of 19% and coextracted 12% V. Puromet MTS9500 performed similarly to Puromet MTX7010, extracting 38% of the Fe in the electrolyte, but also gave high V extraction.
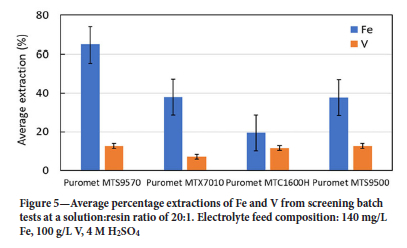
The two resins that showed most promise in the batch screening tests were therefore Puromet MTS9570 and Puromet MTX7010. Both resins reduced the Fe concentration to below the target of 100 mg/L (55 mg/L and 96 mg/L, respectively) using an S/R of 20:1. The co-extraction of V by these cation-exchangers suggested the presence of V in the cationic form as well as the neutral species. El Hage et al. (2019) studied the dissolution of VOSO4 in aqueous sulfuric acid solution, and concluded that this neutral species can dissociate into VO2+ and SO42-. It is likely that VO2+ competes with FeH(SO4)2+ for cation sites on these resins, but the iron species is more strongly complexed. This result suggested that it may be possible to displace V by Fe under capacity-constrained conditions.
Both resins were then subjected to isotherm batch tests to determine their loading characteristics and relative selectivity under maximum loading conditions.
Isotherm batch tests
Figure 6 shows the Fe loading isotherms for Puromet MTS9570 and Puromet MTC1600H at 25°C and 40°C. The maximum Fe loading on Puromet MTS9570 at 25°C was considerably higher (6 g/L Fe) than that of Puromet MTX7010 (1.2 g/L Fe), as expected from capacity data provided by the manufacturer. There was no significant difference on loading of Fe based on temperature. At 40°C, Puromet MTX7010 reached a maximum loading of 1.2 g/L Fe, which was the same value as at 25°C. Loading of Fe on Puromet MTS9570 reached a maximum of 6 g/L at 25°C and 6.3 g/L at 40°C, representing an improvement of only 5% at the higher temperature.
It should be noted that the shapes of these isotherms are distinctly unfavourable. A favourable shape would have a steep incline at low aqueous concentrations, indicating that the resin can reduce the target ion to low levels in the barren solution. Isotherms that have low loading at low solution concentrations, such as these, indicate sorption systems that only complex with the target metal ion at high aqueous concentrations and are more stable in the acid form (Naja and Volesky, 2006). In this case, strong extraction occurred at > 120 mg/L Fe in the aqueous phase. This system may therefore be appropriate for bulk reduction of Fe concentration in the electrolyte to meet the target specification, but will not achieve low Fe concentrations in the V-containing barren solution.
Separation factors
Results from the isotherm tests were used to calculate separation factors, KFe/V, to determine the relative selectivity for Fe loading over V. The selectivity factor, KMt/Mi, which denotes the selectivity of a target ion in solution, Mt, over any other ion in solution, Mi, is given by Equation [6] (Adhikari et al., 2011):

where DMt is the distribution ratio of the target ion in solution and DMi is the distribution ratio of any other ion in solution. The distribution ratio, D, for an ion x in solution in solution is calculated using Equation [7] (Adhikari et al., 2011):

where qe is the amount of metal ion exchanged at equilibrium (g/L) and Ce is the aqueous concentration of that ion at equilibrium (g/L).
Figure 7 shows separation factors KFe/V plotted against S/R for Puromet MTS9570 and Puromet MTX7010. The Puromet MTS9570 data exceeded those for Puromet MTX7010 for all separation factors, except at S/R of 300. This result shows that Puromet MTS9570 has a higher Fe/V selectivity for these experimental conditions. The separation factors for Puromet MTS9570 increased with S/R, reaching a value of 6.1 at S/R = 600. It was therefore concluded that the selectivity of Puromet MTS9570 for Fe over V was higher at higher S/R. This would be especially beneficial in an IX column: the results suggested that if the electrolyte was continuously fed into a column with Puromet MTS9570, Fe would displace V that co-loaded onto the resin, within the relative selectivity constraints of the system. In the case of Puromet MTX7010, it appears that selectivity reversed at higher S/R ratios, with V displacing Fe on the extractant.
From the batch test results, it was concluded that Puromet MTS9570 was the best resin of those considered for application to this highly acidic liquor. This resin extracted more Fe than the other resins in the screening batch tests, gave much higher Fe loading than Puromet MTX7010 in the isotherm tests at both 25°C and 40°C, and showed the highest selectivity for Fe over V. This resin functionality is unique in its ability to complex Fe under high-acidity conditions and has been commercially employed for removal of iron from copper electrolytes containing ~ 1.8 M H2SO4 (Izadi et al., 2017; Xue et al., 2001).
Column test results
Column tests using Puromet MTS9570 were conducted at room temperature (25°C), because there was no significant difference in loading at 25°C and 40°C. This is beneficial because it reduces the energy demands associated with heating of the electrolyte and column equipment. Figure 8 shows breakthrough curves obtained from fixed-bed downflow loading of this resin at 2 BV/h and 4 BV/h. These tests confirmed the selectivity of this resin for Fe over V: Fe loaded onto the resin and the V concentration remained at the feed concentration C0 throughout both experiments, i.e., no V loaded onto the resin. However, it should be noted that breakthrough of Fe occurred almost immediately, which accords with the unfavourable isotherm shape (Figure 6).
Table II shows the mass-transfer zone length (MTZL) calculations obtained from the breakthrough curves (Crittenden et al., 2012). This calculation assumed that breakthrough occurs when the exit concentration is equal to 10% of C0 and resin saturation occurs when the exit concentration is equal to 90% of C0. As an example, the red dotted lines in Figure 8 show the volumes of feed passed when the discharge Fe concentration was 0.1C0 (14 mg/L Fe; 2 BV) and 0.9C0 concentration (126 mg/L Fe; 33 BV) for 2 BV/h flowrate. Equation [8] was used to calculate the MTZL (McCabe et al., 2018):
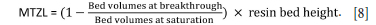
The MTZL occupied a significant portion of the resin bed height, indicating inefficient adsorption. The MTZL calculations suggest that it would be more beneficial to operate the column at a flowrate of 2 BV/h, owing to the slightly shorter MTZL. This means that less resin is required to achieve the same results for specific conditions. A shorter MTZL also suggests faster mass transfer. As shown in Table I, however, the linear flowrate (superficial velocity) through the resin bed was very low under these conditions, which may have caused a stagnant film diffusion layer around the resin beads that inhibited mass transfer. Furthermore, to reach 90% resin capacity (the assumed resin saturation in this case), a flowrate of 2 BV/h allowed treatment of 33 BV of electrolyte compared with 4 BV/h, which only allowed treatment of 23 BV (Figure 8). This means that a higher volume of electrolyte could be treated for Fe removal prior to reaching saturation if the column is operated at 2 BV/h.
Preliminary full-scale column sizing
Table III shows selected full-scale column sizing variables calculated using the design equations provided by Crittenden et al. (2012) to treat an annual electrolyte volume of 8000 m3 (Bushveld specification) at 2 BV/h.
The sizing calculations in Table III show that for a design flow rate Q of 1.01 m3/h, 500 L resin is required. This volume of resin would reach saturation after passing 43 BV electrolyte with an initial Fe concentration of 140 mg/L at 2 BV/h. However, it would not be possible to load all Fe from solution using a single column owing to the unfavourable isotherm shape.
To ensure that full resin loading is achieved without leakage of Fe to the V-containing barren solution exceeding the target specification, it would be necessary to employ two (or more) columns in series in a lead-lag(-lag) configuration. Figure 8 shows that the barren concentration exceeded 100 mg/L Fe when the resin capacity had only reached 70% (2 BV/h) at ~ 20 BV. Passing this eluate through a second (and possibly third) column in series would ensure that the discharge of the final column would remain below 100 mg/L Fe. Once maximum capacity is reached in the first column, the column can be taken off-line for unloading and loading of fresh resin. When the discharge from the second column exceeds 100 mg/L, this is directed into a third column, if necessary, or alternatively the first column, now loaded with fresh resin, is again brought online. The main advantage of such a system is the significant reduction in downtime. If a setup with only one column is used, the column would be offline at least once a day for unloading and loading of fresh resin (required every 21 h), during which time no processing would occur. If two (or three) columns were used, at least one column would continue to run whilst the saturated resin was unloaded and the column reloaded with fresh resin.
Because the electrolyte is highly acidic, it is very difficult to strip this resin with acid. Commercial applications of this resin to a copper electrolyte employ a reductive strip with Cu+ (Xue et al., 2001). An approach was therefore considered in which the resin would be discarded after reaching saturation. Table IV summarizes the cost analysis of this approach using a flowrate of 2 BV/h. Assuming quantitative Fe removal from the electrolyte, the cost for the resin alone exceeds ZAR 127 million/a.
Conclusions and recommendations
IX was evaluated as a technology to remove 140 mg/L Fe from an electrolyte containing 100 g/L V at 4 M H2SO4, for application to a VRFB electrolyte produced from Bushveld Complex magnetite. From the test work conducted, Puromet MTS9570 proved to be the best of the resins assessed, noting that most commercial IX resins are unable to load metal ions at the high acidity of the electrolyte. This resin gave the best performance in screening, isotherm, and selectivity batch tests, and was selected for column loading tests. Under these conditions, the resin selectively extracted Fe from the electrolyte, leaving V in solution. Maximum loading capacity of the resin was 6 g/L Fe3+ and the resin loaded 90% of the feed concentration after 33 BV at a flowrate of 2 BV/h.
A critical drawback of Puromet MTS9570 is the difficulty in stripping using conventional eluants. Design and sizing of a full-scale column therefore assumed discard of the resin (rather than elution) on reaching full loading capacity. Preliminary sizing of a system to treat 8000 m3/a electrolyte showed that the resin would require replacement every 21 h, with a resin replacement cost exceeding ZAR 127 million/a. This frequency of resin change-out can be extended by using a larger resin volume, but at the cost of higher capital cost. Furthermore, owing to the unfavourable isotherm of this resin, at least two columns in series would be required to ensure that the target specification of the barren solution is consistently attained. This is a significant shortcoming of this technology, because the product value cannot justify such a high processing cost.
To progress this concept, future work is required to develop an elution technique for Puromet MTS9570 that would not be detrimental to the V product if any cross-contamination from the elution step occurred. Large volumes of rinse waters between the loading and elution steps will likely be required. However, it would be preferable to consider a different process technology to achieve the target Fe specification of the electrolyte, in which Fe is separated from V earlier in the primary flowsheet (before generation of the 4 M H2SO4 electrolyte) and IX is merely used for scavenging trace residual Fe to meet the target specification.
Acknowledgements
Bushveld Vametco supplied V2O3 and background information for this testwork: Alicia Enslin is thanked for assistance and advice. All resins were kindly donated by Purolite: we thank Johanna van Deventer for assisting with training, advice, and experimental procedures. CM Solutions generously sponsored all chemical analyses for this work and provided laboratory equipment for some tests. Sincere appreciation is also extended to Johanna van Deventer (Purolite) and Olga Bazkho (Mintek), whose helpful comments considerably improved the quality of this manuscript.
CRediT author statement
MM: Investigation, Writing - Original draft preparation; KCS: Conceptualization, Supervision, Methodology, Funding, Writing- Reviewing and Editing.
References
Adhikari, C.R., Kumano, H., Tanaka, M. 2011. Selective removal of zinc from an electroless nickel plating bath by solvent impregnated resin using 2-ethylhexylphosphonic acid mono-2-ethylhexyl ester as the extractant. Solvent Extraction and Ion Exchange, vol. 29, no. 3, pp. 323-336. [ Links ]
Akinbami, O.M., Oke, S.R., Bodunrin, M.O. 2021. The state of renewable energy development in South Africa: An overview. Alexandria Engineering Journal, vol. 60, no. 6, pp. 5077-5093. [ Links ]
Bushveld Minerals. 2024. About vanadium. Retrieved from https://www.bushveldminerals.com/about-vanadium/ [accessed 14 Apr. 2024]. [ Links ]
Chace, W.S., Tiano, S.M., Arruda, T.M., Lawton, J.S. 2020. Effects of state of charge on the physical characteristics of V(IV)/V(V) electrolytes and membrane for the all vanadium flow battery. Batteries, vol. 6, no. 4, p. 49. [ Links ]
Crittenden, J.C., Trussell, R.R., Hand, D.W., Howe, K.J., Tchobanoglous, G. 2012. MWH's Water Treatment: Principles and Design. John Wiley & Sons. [ Links ]
Ding, M., Liu, T., Zhang, Y., Cai, Z., Yang, Y., Yuan, Y. 2019. Effect of Fe(III) on the positive electrolyte for vanadium redox flow battery. Royal Society Open Science, vol. 6, no. 1, p. 181309. [ Links ]
El Hage, R., Chauvet, F., Biscans, B., Cassayre, L., Maurice, L., Tzedakis, T. 2019. Kinetic study of the dissolution of vanadyl sulfate and vanadium pentoxide in sulfuric acid aqueous solution. Chemical Engineering Science, 199, 123-136. [ Links ]
Izadi, A., Mohebbi, A., Amiri, M., Izadi, N. 2017. Removal of iron ions from industrial copper raffinate and electrowinning electrolyte solutions by chemical precipitation and ion exchange. Minerals Engineering, vol. 113, pp. 23-35. [ Links ]
Kim, K.J., Park, M.S., Kim, Y.J., Kim, J.H., Dou, S.X., Skyllas-Kazacos, M. 2015. A technology review of electrodes and reaction mechanisms in vanadium redox flow batteries. Journal of Materials Chemistry A, vol. 3, no. 33, pp. 16913-16933. [ Links ]
LibreTexts Chemistry. 2020. Acid strength and the acid dissociation constant (Ka). Retrieved from https://chem.libretexts.org/Bookshelves/General_Chemistry/Map%3A_A_Molecular_Approach_(Tro)/16%3A_Acids_and_Bases/16.04%3A_Acid_Strength_and_the_Acid_Dissociation_Constant_(Ka) [ Links ]
McCabe, W.L., Smith, J.C., Harriott, P. 2018. Unit Operations of Chemical Engineering. McGraw-Hill. [ Links ]
Naja, G., Volesky, B. 2006. Behaviour of the mass transfer zone in a biosorption column. Environmental Science & Technology, vol. 40, no. 12, pp. 3996-4003. [ Links ]
Prifti, H., Parasuraman, A., Winardi, S., Lim, T.M., Skyllas-Kazacos, M. 2012. Membranes for redox flow battery applications. Membranes, vol. 2, no. 2, pp. 275-306. [ Links ]
Sole, K.C., Mooiman, M.B., Hardwick, E. 2018. Ion exchange in hydrometallurgical processing: an overview and selected applications. Separation & Purification Reviews, vol. 47, no. 2, pp. 159-178. [ Links ]
The Geochemist's Workbench. 2023. Retrieved from https://www.gwb.com/ [accessed 14 Apr. 2024]. [ Links ]
Wang, H., Pourmousavi, S.A., Soong, W.L., Zhang, X., Ertugrul, N. 2023. Battery and energy management system for vanadium redox flow battery: A critical review and recommendations. Journal of Energy Storage, vol. 58, p. 106384. [ Links ]
Xue, S.S., Gula, M.J., Harvey, J. T., Horwitz, E.P. 2001. Control of iron in copper electrolyte streams with a new monophosphonic/ sulphonic acid resin. Minerals and Metallurgical Processing, vol. 18, no. 3, pp. 133-137. [ Links ]
 Correspondence:
Correspondence:
K.C. Sole
Email: kathy.sole@up.ac.za
Received: 15 Feb. 2024
Published: December 2024













