Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Dental Journal
On-line version ISSN 0375-1562
Print version ISSN 0011-8516
S. Afr. dent. j. vol.71 n.2 Johannesburg Mar. 2016
CLINICAL REVIEW
Epithelial cell rests of Malassez: From quiescent remnants to front-runners in periodontal homeostasis and regeneration. A brief review
HD MiniggioI; EJ RaubenheimerII
IBDS, MSc. Unit 5 Oral Biology, Sefako Makgatho Health sciences University, Oral Health Centre, Setlogelo Drive, Ga-Rankuwa
IIPhD, DSc. unit 2 oral Pathology, Sefako Makgatho Health Sciences University, Oral Health Centre, Setlogelo drive, Ga-Rankuwa
ABSTRACT
In recent years, research has been directed towards elucidating the physiological role played by the epithelial cell rests of Malassez within the periodontal ligament and surrounding alveolar bone. These unique structures were, for many years, considered to be quiescent remnants of development, but are now emerging as key role-players in the maintenance of periodontal health and are also implicated in bone resorption through secretion of bone resorption mediators. Reviews on this topic are infrequent in the dental literature and the notable advances regarding the cytokine profile of the epithelial cell rests of Malassez and their autocrine and paracrine roles in the periodontal tissues make it opportune to review their physiological roles in maintaining periodontal health. Furthermore, experiments have established that epithelial cell rests of Malassez contain stem cells that can undergo epithelial-mesenchymal transitions, placing these cells at the centre of periodontal regeneration.
Key words: epithelial cell rests of Malassez, epidermal growth factor, periodontal homeostasis, periodontal regeneration, paracrine signaling, bone remodeling
METHODS
A literature search was performed on 17th February, 2015, using the term "epithelial cell rests of Malassez" which produced 159 results. From these results, articles were selected that were in English, dating from 1817, and were related to this review. A total of 23 articles were selected and included in the reference.
INTRODUCTION
A thorough understanding of the biological basis of maintenance of periodontal health is indispensable to many aspects of clinical dentistry in order that adequate dental treatment may be provided. Epithelial cell rests of Malassez (ERM) are integrated in the periodontal ligament and have been found to secrete several growth factors, cytokines, chemokines, adhesion molecules and associated proteins.1 We present here recent advances regarding the characterization of the cytokine profile of ERM which validates their important function in periodontal ligament homeostasis with an emphasis on their role in alveolar bone remodelling. We also highlight the capacity of ERM to undergo epithelial-mesenchymal transitions, which indicates their involvement in periodontal regeneration.
Epithelial cell rests of Malassez were first described by Augustin Serres in 1817.2 Later Louis-Charles Malassez provided a full description of their morphology and distribution in 1884 and noted that these epithelial cells persist to form part of the adult periodontal ligament.3 Epithelial cell rests of Malassez are characterised as bundles of epithelial cells with darkly stained nuclei that are arranged as an interconnected network throughout the periodontal ligament4 (Figures 1 and 2).
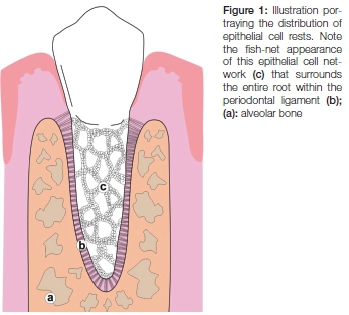
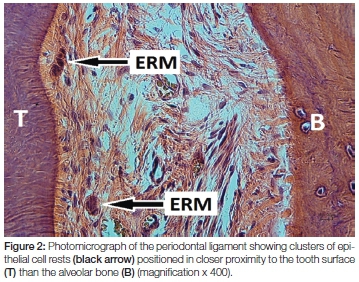
Epithelial cell rests of Malassez originate from the degeneration of Hertwig's epithelial root sheath to form quiescent cell rests that persist as the sole epithelial cells in the periodontium.5 For many years following their discovery they were considered inert remnants with little or no physiological role in the periodontal ligament.6 More recent experiments have elucidated their functional role in maintaining constant the width of the periodontal ligament and their additional role in the regeneration of periodontal tissues.1
Characterization of the cytokine profile of ERM have brought to light important bone resorbing factors, growth factors, chemokines and related proteins which are directly implicated in the process of bone remodelling.1,4 Furthermore, adhesion molecules such as osteopontin and bone sialoproteins which regulate mineralization, are secreted by ERM in the root regions of mature and developing teeth.5 These considerations have led to the postulate that one of the principal roles of ERM in the periodontal ligament involves paracrine signalling to the surrounding alveolar bone. This would explain their role in maintaining a constant periodontal ligament space and would also explain their proliferation and implication in induced tooth movements.1
Bone is a metabolically active specialized form of connective tissue that undergoes continuous remodelling through a tightly regulated interaction between two equivalent and opposing events, specifically, bone resorption of pre-existing bone by osteoclasts, followed by new bone formation by osteoblasts.7 Bone matrix contains primarily type I collagen and in lesser amounts type V collagen which together constitute approximately 90% of the total bone matrix proteins.7 Following completion of bone formation, a physiological process known as bone remodelling is responsible for maintaining bone mass, for the repair of fractures as well as sustaining calcium and phosphorous homeostasis.8-10 The successive bone-degradation and bone-formation sequence and the observation that under physiological conditions bone mass is kept constant, has led to the postulate that the two opposing processes regulate each other.8 Current understanding of the process of osteoclastogenesis stems from extensive studies of the interactions between several ligands and cell surface receptors.9-11
RANKL/RANK EXPRESSION IN OSTEOCLASTS AND OSTEOBLASTS
Macrophage-colony stimulating factor (M-CSF) and several transcription factors are required for the final transformation of haematopoietic stem cells to osteoclasts.9 Formation of osteoclasts is dependent on the receptor activator of nuclear factor kappa-B ligand (RANKL), the osteoclastogenic cytokine, which belongs to the tumour necrosis factor (TNF) superfamily and which is produced by various cell types including osteoblasts, osteocytes as well as lymphocytes.8,11,12 Receptor activator of nuclear factor kappa-B (RANK) is the cell bound receptor for RANKL found on the surface of preosteoclasts.9 Binding of RANKL to its receptor RANK initiates a series of signalling pathways that are mediated by TNF receptor-associated factor 6 (TRAF 6).9 Osteoblasts control osteoclastogenesis through the secretion of osteoprotegerin (OPG) protein which is also a member of the TNF receptor superfamily and is identical in structure to the extracellular domain found in RANK, thereby acting as a decoy receptor by binding directly to RANKL and inhibiting osteoclast activation and bone resorption9 (Figure 3).

ERM AND BONE REMODELLING
Epithelial cell rests of Malassez have the ability to maintain their structure within the periodontal ligament through the constant release of a polypeptide known as epidermal growth factor (EGF).13,17 Epidermal growth factor was identified by Stanley Cohen in 1962 and has been associated with odontogenesis and tooth eruption.14 Epidermal growth factor represents a mediator for cell differentiation, proliferation and cell growth.14 The receptor for this growth factor (EGFr) is made up of transmembrane proteins that activate tyrosine kinase intracellularly and trigger cellular events that lead to cell division that explains their role in regeneration of periodontal tissue.16,17 Epithelial cell rests of Malassez are able to proliferate and to sustain their structure through autocrine signalling by the constant release of EGF that is responsible for their growth and integrity.13
Epidermal growth factor released by the ERM is directly implicated in osteoclastogenesis and hence bone resorp-tion through the inhibition of osteoprotegerin (OPG), a well-established decoy receptor for RANKL.12,13,15 Con-comitantly EGF enhances the expression of monocyte chemoattractant protein-1 (MCP1) due to the proximity of ERM to surrounding alveolar bone and the continuous secretion of EGF which will have a resorptive effect on the adjacent alveolar bone with the net effect of alveolar bone remodelling in order to maintain the periodontal ligament space at a constant, that is approximately 0,25 mm.12,18
The connective tissue that separates ERM from adjacent bone cells is believed to provide the essential dilution site for EGF, preventing this polypeptide from reaching the EGF receptors on bone cells in high concentrations thereby avoiding excessive bone resorption.12 It is interesting to note that the junctional epithelium, the sulcular epithelium and the connective tissue attachment of the tooth are kept at a constant remove away from the alveolar bone crest, otherwise known as the biological distance.12 It is believed that this serves as the metabolism site for EGF as this polypeptide is only able to interact with its receptors on bone cells for a limited time and a minimum of one quarter of the receptors are required to be activated in order for EGF to have resorbing effects on the alveolar bone.12 This also clarifies why an operative or restorative procedure that impinges upon the biological distance will cause re-sorption of alveolar bone crest, due to the higher concentration of EGF that reaches the bone cell receptors.12 The junctional epithelium proliferates and migrates apically following the operative or restorative procedure and releases EGF in order to maintain its structure.12 This migration and proliferation of the junctional epithelium increases the concentration of EGF in the micro-environment of the alveolar bone crest thereby causing bone resorption.12
Epithelial cell rests of Malassez have also been shown to proliferate when stimulated by induced tooth movements.8,18-21 The proliferation of ERM increases the amount of secreted mediators, particularly EGF that induces bone resorption as well as the renewal and reorganization of the cells within the periodontal ligament including cementogenesis on the resorptive tooth surface.819,21 Experiments have demonstrated that Hertwig's epithelial root sheath, under the influence of transforming growth factor beta (TGF-β), is capable of undergoing epithelial-mesen-chymal transition.22 Furthermore, evidence suggests that ERM contain a subpopulation of stem cells which are capable of undergoing epithelial-mesenchymal transformation into mesenchymal stem-like cells with multi-lineage potential.22 These multi-potent epithelial stem cell populations have the potential to differentiate into, amongst others, osteoblasts, adipocytes and chondrocytes, and have been shown to be implicated in cementogenesis.22 These epithelial-mesenchymal conversions provide a supply of progenitor cells that have the capacity to migrate to sites of injury and thus play a pivotal role in the induction of periodontal regeneration.23,24
In the absence of EGF release, ERM would lose their ability to self-stimulate and maintain their structure within the periodontal ligament. In addition, paracrine signalling through the secretion of mediators of bone resorption would not take place, impacting on the process of bone remodelling around the tooth and leading to the tooth assuming the traits of ankylosis. Therefore, the persistence of ERM in the periodontal ligament is essential to the maintenance of periodontal homeostasis.
CONCLUSION
Epithelial cell rests of Malassez are functional components of the periodontium that fulfil key physiological and regenerative functions to ensure periodontal integrity. These include:
- Maintenance of a constant periodontal ligament space by indirect stimulation of osteoclast differentiation though decreasing the decoy receptor OPG and increasing MCP1;
- Prevention of the formation of alveolodental ankylosis by constantly secreting bone resorptive mediators thereby preventing mineralisation of the periodontal ligament;
- Participation in the process of periodontal regeneration by providing a stem cell supply which can migrate to sites of resorption and injury and stimulate cementogenesis;
- Participation in tooth movement by increasing EGF production in periodontal tissues and helping to repair root resorption areas while stimulating cementogenesis.
Due to the numerous physiological and regenerative functions that ERM perform within the periodontal ligament, the authors suggest that the current nomenclature of epithelial cell "rests" should be replaced by the term epithelial "network" in order to emphasize the central role that these cells play in the homeostasis of the tooth supporting apparatus.
Acknowledgement
The authors would like to acknowledge Nicolette Jordaan from Medical Illustration & Audio-Visual Services, SMU, for her assistance with the illustrations in this article.
References
1. Ohshima M, Yamaguchi Y, Micke P, Abiko Y, Otsuka K. In vitro characterization of the cytokine profile of the epithelial cell rests of Malassez. J Periodontol 2008; 79 (5): 912-9. [ Links ]
2. Serres A. Essai sur Lanatomie et la physiologie des dents ou nouvelle théorie de la dentition. Mequignon-Marvis, Paris. 1817: 28. [ Links ]
3. Malassez L. Sur l'existence damas epitheliaux autour de la racine des dents chez l'homme adulte et a l'etat normal (debris epitheliaux paradentaires). Arch Physiol 1885; 5: 129-48. [ Links ]
4. Rincon JC, Young WG, Barltold PM. The epithelial cell rests of Malassez: a role in periodontal regeneration? J Period Res 2006; 41 (4) 245-52. [ Links ]
5. Wang Y, Linlin Lv, Yu X, et al. The characteristics of epithelial cell rests of Malassez during tooth eruption of development mice. J Mol Hist 2014; 45: 1-10. [ Links ]
6. Nanci A. Ten Cate's Oral Histology: Development, Structure and Function, 8th edition Edition: Elsevier, 2013. [ Links ]
7. Ross MH, Pawlina W. Histology, A Text and Atlas. 2006. Lip-pincott Williams & Wilkins, 5th Ed. [ Links ]
8. Corral DA, Amling M, Priemel M, et al. Dissociation between bone resorption and bone formation in osteopenic transgenic mice. Proc Natl Acad Sci USA 1998; 95(23): 12835-13840. [ Links ]
9. Boyce BF. Advances in the regulation of osteoclasts and os-teoclast functions. J Dent Res 2013; 92(10): 860-7. [ Links ]
10. Soysa NS, Alles N, Aoki K, Ohya K. Osteoclast formation and differentiation: An overview. J Med Dent Sci 2012; 59(3): 65-74. [ Links ]
11. Weitzmann, MN. The role of inflammatory cytokines, the RANKL/OPG axis, and the immunoskeletal interface in physiological bone turnover and osteoporosis. Scient 2013; doi:10.1155/2013/125705. [ Links ]
12. Consolaro A, Consolaro MF. ERM functions, EGF and orthodontic movement. Dental Press J Orthodont 2010; 15 (2)-24-32. [ Links ]
13. Raisz LG, Simmons HA, Sandburg AL, Canalis E. Direct stimulation of bone resorption by epidermal growth factor. Endocrinol 1980; 107(1): 270-3. [ Links ]
14. Cohen S. 1962. Isolation of a mouse submaxillary gland protein accelerating incisor eruption and eyelid opening in the new-born animal. J Biol Chem 1982; 237: 1555-62. [ Links ]
15. Zhang X, Tamasi J, Lu X, et al. Epidermal growth factor receptor plays an anabolic role in bone metabolism in vivo. J Bone and Min Res 2011; 26(5): 1022-34. [ Links ]
16. Carpenter G. Receptors for epidermal growth factor and other polypeptide mitogens. Ann Rev Biochem 1987; 56: 881-914. [ Links ]
17. Carpenter G. Epidermal growth factor: biology and receptor metabolism. J Cell Sci Suppl 1985; 3:1-9. [ Links ]
18. Schneider MR, Sibilia M, Erben RG. The EGFR network in bone biology and pathology. Trends in Endocrinol and Met 20(10): 517-524. [ Links ]
19. Walker GC, Dangaria S, Ito Y, Luan X, Diekwisch TGH. Oste-opontin is required for unloading-induced osteoclast recruitment and modulation of RANKL expression during tooth drift associated bone remodelling but not for super-eruption. Bone 2010; 47: 1020-9. [ Links ]
20. Saddi KR, Alves GD, Paulino TP, Ciancaglini P, Alves GB. Epidermal growth factor in liposomes may enhance osteoclast recruitment during tooth movement in rats. Angle Orthodont 2008; 78(4): 604-9. [ Links ]
21. Alves JB, Ferreira CL, Martins AF, et al. Local delivery of EGF-liposome mediated bone modelling in orthodontic tooth movement by increasing RANKL expression. Life Sci 2009; 85(19-20):693-9. [ Links ]
22. Xiong J, Mrozik K, Gronthos S, Barltold PM. Epithelial cell rests of Malassez contain unique stem cell populations capable of undergoing epithelial-mesenchymal transition. Stem Cell and Dev 2012; 21(11): 2012-25. [ Links ]
23. Seo BM, Miura M, Gronthos S, et al. Investigation of multipo-tent postnatal stem cells from human periodontal ligament. Lancet 2004; 364: 149-55. [ Links ]
24. Battula VL, Evans KW, Hollier BG, et al. Epithelial-mesenchy-mal transition-derived cells exhibit multi-lineage differentiation potential similar to mesenchymal stem cells. Stem Cells 2010; 28: 1435-45. [ Links ]
 Correspondence:
Correspondence:
Hilde Doris Miniggio
P.o. Box 0204, medunsa, Pretoria
Tel: 012 521 4882, Fax: 012 521 4274, Cell: 072 869 4045
E-mail: hilde.hendrik@smu.ac.za
SA dentists receive certification by World Clinical Laser Institute
At the 2-day International Laser Dentistry workshop held recently at Melrose Arch Johannesburg, 38 leading South African dentists received international Certification from the The World Clinical Laser Institute and Germany's Aachen University Dental Laser Centre. Hosted by SciVi-sion, importer of Biolase dental laser technology, and guest speaker - internationally acclaimed author and President of the World Laser Dentistry Federation, Professor Nor-bert Gutknecht, the workshop delivered superlative international pedigree to the local dentals. Skills gained at the workshop are now poised to bring advanced, pain reduced dental treatment to South African consumers.
Prof. Gutknecht comments, "With more than 25 years' laser research experience, I am excited to share my dental laser knowledge with leading South African dentists, and in turn, your public. I highly appreciate the fact that SciVi-sion is so strongly focussed on education it's dentists on the latest trending technology.
The most important aspect of laser dentistry is having an adequately qualified dentist to use a laser on a patient, in a successful way. For the South African market, laser dentistry offers reduced pain during treatment, stimulated treatment of children who are normally afraid of dentists, while giving practitioners the ability to treat all patients with less pain.
In infectious cases, lasers have a high disinfecting potential - enabling dentists to treat a wider variety of patients which they would not have been able to treat with conventional methods. Our intention is to use lasers as an integrative dental tool so as to improve the overall outcome of general dentistry, paediatric dentistry, orthodontics, periodontistry and surgery. South Africa is the ideal market to focus on the use of dental lasers as it has dedicated, highly skilled and progressive dentists in good facilities. I'm optimistic that South Africa will soon be the strongest in the world in terms of using lasers in dentistry." (Interview with Prof. Gut-kneckt - https://www.facebook.com/scivision/videos)
Rhoodie Garrana - WITS University: Department of Oral Medicine & Periodontology says, "It was a great privilege being able to spend two days with Professor Gutnekcht from Aachen University where he shared the advantages of incorporating laser Technology into the field of Dentistry. I believe the Waterlase and Epic laser have the potential to become valuable adjuncts in various dental fields where they provide management options as well as disinfection and healing promotive properties. The technology available has opened up a door to a new realm waiting to be explored. The literature has so far been promising and the course highlighted the potential of incorporating laser therapy into day to day therapy. This technology is rapidly progressing and I have no doubt that it will become an invaluable tool in the future of dentistry. Thank you Scivision for a great opportunity."
According to Peter Doubell, CEO of Scivision, laser technology is an essential tool for innovative dental practitioners wishing to treat patients in a modern and pain reduced manner. Doubell infers that lasers enable new dental treatments and operating methods, as well as complementing and supporting existing therapeutic dental methods. "With patients increasingly seeking alternative therapies, our Biolase dentists can meet these expectations by providing patients with less invasive solutions with added benefits when compared to more traditional methods," he says.
Dr Preshaylin Govender adds, "I had the privilege of attending the prestigious Scivision workshop which was a great learning experience which I have already applied to my practice with great success. The lectures given by Prof. Gutknecht were of high calibre - his professionalism and 25 years of laser research knowledge reminded us that practicing with lasers drives practitioners to achieve dental results on a higher level."
Dr Herman Botha adds, "As new dental laser user I was thrilled at the opportunity to attend the course with Professor Gutknecht. His depth of knowledge on biophysics and laser treatments was simply outstanding. I am inspired and have more confidence and understanding in treating my patients using laser."
For more information on SciVision and Biolase technology, visit http://www.scivision.co.za or visit www.biolas-edentist.co.za for a list of Biolase dentists countrywide.













