Serviços Personalizados
Artigo
Indicadores
Links relacionados
-
 Citado por Google
Citado por Google -
 Similares em Google
Similares em Google
Compartilhar
South African Dental Journal
versão On-line ISSN 0375-1562
versão impressa ISSN 0011-8516
S. Afr. dent. j. vol.71 no.10 Johannesburg Nov. 2016
COMMUNICATION
Biological significance of palatine tonsillar epithelium: microstructure and disease
HD Miniggio
BDS, MScMed(Bioethics and Health Law). Unit 5: Oral Biology, Faculty of Health Sciences- Sefako Makgatho Health Sciences University, Pretoria, South Africa
ABSTRACT
The epithelial microstructural configuration of the paired palatine tonsils is adapted to optimize lympho-epithelial symbiosis at the first point of contact with an antigen introduced to the pharynx through breathing or swallowing. The loosely arranged reticulated nature of the epithelium of the tonsillar crypt and vesicular transcytosis of live bacteria and viruses across receptor cells contribute to the early pathogenesis of specific neoplastic- and infective diseases which are discussed in the manuscript.
Key words: tonsillar-epithelium, tonsillar-crypt, immunity, M-cells, oncogenesis, epithelial-lymphoid symbiosis, tonsillar-disease.
INTRODUCTION
The paired palatine tonsils are situated between the palatoglossal- and palatopharyngeal arches and form part of a group of oropharyngeal lymphoid tissue collectively known as Waldeyer's ring. Other components of this lymphoid ring are located on the posterior surface of the tongue (lingual tonsils), roof of the pharynx (pharyngeal tonsils or adenoids) and tubal tonsils which are found posterior to the openings of the Eustachian tubes on the lateral pharyngeal wall. Waldeyer's ring of lymphoid tissue is also functionally referred to as "mucosa-associated lymphoid tissue" or MALT1 and is the first line of adaptive defence against inhaled or ingested antigens. The epithelial surfaces of the tonsils are modified to provide a large contact area for the capturing and presentation of antigens to the associated B- and T lymphoid stem cell populations. Induction of a sub population of B- memory cells with a capacity to produce antigen-specific secretory antibodies on demand and the establishment of a directed lineage of functional T cells2, form important components of the adaptive immune protection of the respiratory system and digestive tract.
MALT in Waldeyer's ring is well-organized and is in close contact with a specialized epithelial surface. The palatal- and lingual tonsils are covered by stratified squamous epithelium and the tubal- and pharyngeal tonsils by pseudo stratified columnar ciliated epithelium with goblet cells. Although the palatine- and tubal tonsils have several crypts, the lingual tonsil has only one and the columnar epithelium covering the pharyngeal tonsils is folded with no true crypts. Variations in the micro- structural arrangement of the epithelium play a significant role in the early pathogenesis of specific disease processes originating in the tonsillar epithelium.
This manuscript provides a functional description of the epithelium and highlights micro-anatomical factors which impact on the early pathogenesis of selected disease states originating in the palatine tonsils.
MATERIALS AND METHODS
Haematoxylin and eosin stained sections of tonsils previously removed during routine tonsillectomies were retrieved from the files of Oral Pathology at Sefako Makgatho Health Sciences University. The sections were screened by an Oral Pathologist and 15 cases with reactive lymphoid hyperplasia were selected. Sections were prepared from the archived blocks and stained with the immunoperoxidase technique for S100 protein, low- and high molecular weight cytokeratin, Ki 67 and IgA. The sections were viewed under a light microscope with a fluorescence attachment and digital camera.
RESULTS AND DISCUSSION
Surface epithelium
The palatine tonsil is covered by 15-20 layers of either flattened non-keratinised- or parakeratinised squamous epithelium resting on a basement membrane. A layer of fibrous connective tissue separates the surface epithelium from the lymphoid component. Selected surface epithelial cells express both cell surface glycoprotein CD44 and nerve growth factor receptor NGRF (or CD271), fulfilling the requirement that stem cells should have a potential for prolonged growth.3 Dendritic cells migrate in the intercellular spaces and play a key role in antigen recognition. CD8+ T-cells are increased in the surface epithelium in recurrent tonsillitis when compared with idiopathic tonsillar hypertrophy.4 The thickness of the covering epithelium and the dense junctions provide a barrier protecting basally located stem cells against external irritants such as tobacco and infection by human papilloma virus (HPV).5 There must be alterations to the basally located stem cell population to allow for any advancement of the pathogeneses associated with these and other irritants. Small ulcers resulting from surface abrasions may however breach this protection (Figure. 1).

Examination of serial sections indicated that the development of tonsillar crypts is dynamic and initiated in the surface epithelium. Crypt formation of the surface epithelium followed upon a localized immune reaction with increased mitotic activity (Figure 2), invagination of epithelium and formation of a crypt. The crypt epithelium ultimately reticulates and induction of MALT close to the crypt results in the establishment of a functional crypt. Through the formation of a crypt the efficiency of antigen presentation to the underlying lymphoid component is facilitated.

Crypt epithelium
The main function of a tonsillar crypt is to initiate the first step of the adaptive immune reaction. This is achieved by capturing and then transferring an antigen from the tonsillar crypt to the MALT. In order to facilitate this function, the crypt is lined by reticulated epithelium intermingled with lymphoid cells and alternated by patches of squamous epithelium resembling surface epithelium (Figure 3).

The number of crypts in a palatine tonsil correlates with the extent and reactivity of the lymphoid component. Enlargement and branching of crypts increase significantly the contact area between the epithelial and lymphoid components. The degree of reticulation increases upon antigenic stimulation. When extensively challenged the junction between the intermingled epithelial- and lymphoid component becomes difficult to visualize on sections stained with haematoxylin and eosin (Figure 3). Stains for cytokeratin are helpful in distinguishing the epithelial cells from the lymphoid component (Figure 4).

The epithelium rests on a porous basement membrane6 which consists of a rich fibre network composed of specific collagen types which maintain intimate contact between the stationary epithelial cells and the migratory lymphoid cell population. Macromolecules in the basement membrane contribute to the immunological events which follow upon contact with an antigen.7 Unlike surface epithelium, close contact is established between the stationary epithelial cells lining a crypt, mobile components of MALT and endothelial lined capillaries. Capillary endothelium is frequently in direct contact with the reticulated epithelial cells and high endothelial lined venules extend into the reticulated epithelium, particularly in extensively reticulated areas where the venules often reach the surface of the crypt.8
Microfold cells (M-cells) are specialized epithelial cells within the crypt lining which act as the gateway for antigens to the immune system. They are also located in other sites such as in Peyer's patches in gut associated lymphoid tissue (GALT). M-cells are adapted structurally and functionally for the transepithelial passage and presentation of foreign antigens to lymphoid cells in close proximity to the crypt. They are difficult to demonstrate microscopically in the tonsils on routine stained sections but may be visualized on scanning electron micrographs as microvillous cells in small pore-microcrypts between the epithelial cells lining the crypts.9 The microvilli are oriented towards the crypt lumen and are responsible for endocytosis of antigens which bind to specific receptors on the M-cell surface. A recent study proposed class II β-tubulin as a novel marker for tonsillar M-cells.10 Following endocytosis, vesicular transcytosis occurs through the M-cell cytoplasm and the antigen is presented to dendritic cells, macrophages, and T-and B lymphocytes closely associated with the invaginated surfaces on the M-cells which are exposed to the lymphoid component (Figure 5).

A direct targeted immune response is then induced. This step completes the trans-epithelial cycle of MALT immune activation.2 The final outcomes are activation of B- and T cells in the lymphoid component of the tonsil through presentation of the antigen by follicular dendritic cells to the lymphoid stem cell populations in and around the lymphoid follicles (Figure 6).

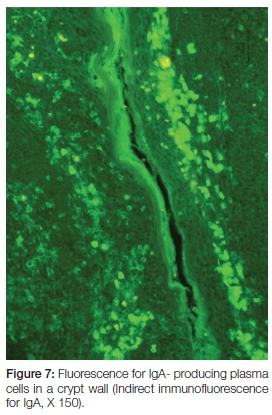
Production of secretory immunoglobulins facilitate mucosal immunity and the seeding of lymphoid cells (via their close association with the high venules) to systemic sites. Here they exert their specific and directed immunological actions against the antigen which was originally presented via the trans-epithelial route of the tonsillar crypt. The sleeve of plasma cells around capillaries form finger-like projections between the crypt epithelium of a reactive tonsil.8 Not only do they shed immunoglobulins into the crypt for local protection, but also into circulation for systemic distribution (Figure 7).
Micro anatomical determinants of selected diseases
Antigens and some microorganisms are transported intact and alive across the M-cells.11 These cells are therefore important vectors for the presentation of active bacteria (such as Mycobacterium tuberculosis) and viruses (such as Epstein Barr virus) which primarily target cells of the lymphoid cell lineages. Multiplication in the tonsillar lymphoid cells and seeding to distant sites can subsequently occur. Human immunodeficiency virus infection (HIV) reduces the number of follicular dendritic cells and impacts on the efficiency of antigen transfer between the M-cell and the MALT stem cell populations.12 Highly active anti-retroviral therapy (HAART) restores the lymphoid dendritic cell population within months.12 It has recently been demonstrated that the tonsillar crypt epithelium is an important site for Enterovirus 71 replication, the causative agent of hand foot and mouth disease and a rare form of fatal encephalomyelitis in humans.13 In acute tonsillitis, which is caused by a wide range of viruses (adenovirus, influenza virus, respiratory syncytial virus and others) or bacteria (beta-haemolytic streptococci, staphylococcus and others), the protective immune response is overwhelmed by the infection and although most cases there is uneventful healing, serious complications such as a cellulitis or pharyngeal abscess could follow. The necrotic debris in the crypts of the palatine- and lingual-tonsils may undergo dystrophic calcification with the formation of a tonsillolith. With chronic irritation the antigen presentation role of an intact crypt is neutralized due to the replacement of the specialized crypt lining by squamous epithelial cells (Figure 8).

Epithelial basement membrane proteins serve as primary receptors for HPV,5 the most prevalent sexually transmitted infection.14 Due to the loosely-arranged syncytial nature of the epithelium, which by virtue of its functional adaptation exposes parts of the basement membrane to the external environment, the tonsillar crypt is the preferred site in the oropharynx for HPV infection. It is estimated that 8.5% of normal tonsils contain HPV DNA, either types 6 and 11 or the high risk type 16.15 Local immune recognition of HPV is supressed by the virus.16 HPV 16 viral oncoprotein E7 reduces e-cadherin dependant dendritic cell adhesion to epithelial cells,16 the virus supresses dendritic cell migration to epithelium17 and suspends the release of pro-inflammatory cytokines by HPV infected epithelial cells.5 Viral integration in the crypt stem cell population secures a constant shedding of viral particles through release of HPV into the crypt as infected cells lyse. Carcinomas induced by high risk HPV type 16 are more likely to develop through infection and transformation of the CD44 and nerve growth factor receptor (NGFR) positive stem cells within a crypt than in the other epithelial surfaces of the pharynx where the stem cell population is protected by the multilayered nature of the epithelium. A recent (2010) summary of malignant histopathological transformation is available.18 HPV associated tonsillar cancer develops after more than a decade of exposure to high risk HPV's which are biologically characterized by HPV's E6 and E7 proteins that bind to- and inactivate tumour suppressor protein p53 and retinoblastoma protein (pRB), with a resultant upregulation of p16, leading to malignant transformation of HPV infected cells.18
CONCLUSION
Characterization of the molecular nature of the receptors on the apical surface of M-cells and an understanding of the lympho-epithelial symbiosis in the tonsillar crypt are important fields of research for the pharmaceutical manipulation of the trans-mucosal route of infectious agents. Furthermore, probing of the receptor classes on the microvillous surface of M-cells could potentially improve the efficiency of parenterally administered vaccines.
Acknowledgement: The author wishes to thank Prof EJ Raubenheimer for his guidance in preparing this manuscript.
Note: Some of the concepts reviewed in this manuscript were presented at the HPV Symposium and AML-Riatol Chair Inauguration, May 2016, University of Antwerp, Belgium.
Conflict of interest: None declared.
ACRONYMS
MALT: mucosa-associated lymphoid tissue
HPV: human papilloma virus
References
1. Brandzaeg P, Kiyono H, Pabst R, Russel MW. Terminology: nomenclature of mucosa-associated lymphoid tissue. Immunol 2008; 1:31-7. [ Links ]
2. Sada-Ovale I, Talayero A, Chavez-Galan L, et al. Functionality of CD4+ and CD8+ T cells from tonsillar tissue. Clin Exp Immunol 2012; 168: 200-6. [ Links ]
3. Kang SY, Kannan N, Zhang L, et al. Characterization of epithelial progenitors in normal human palatine tonsils and their HPV16 E6/E7- induced perturbation. Stem Cell Rep 2015; 5: 1210-25. [ Links ]
4. Olofsson K, Hellstrőm S, Hammerstrőm M-L. The surface epithelium of recurrent infected palatine tonsils rich in yỡ T cells. Clin Exp Immunol 1998; 111:36-47. [ Links ]
5. Egawa N, Egawa K, Griffin H, Doorbar J. Human papillomaviruses; epithelial tropisms, and the development of neoplasias. Viruses 2015; 16:3863-90. [ Links ]
6. Choi G, Suh YL, Lee HM, Jung KY, Hwang SJ. Prenatal and postnatal changes of the human tonsillar crypt epithelium. Acta Otolaryngol Suppl 1996; 523:28-33. [ Links ]
7. Määtta M, Liakka A, Salo S, et al. Differential expression of basement membrane components in lymphatic tissues. J Histochem Cytochem 2004;52:1073-81. [ Links ]
8. Perry M. The specialised structure of crypt epithelium in the human palatine tonsil and its functional significance. J Anat 1994;185:111-27. [ Links ]
9. Jovic M, Avramovic V, Vlahovic P, et al. Ultrastructure of the human palatine tonsil and its functional significance. Rom J Morphol Embryol 2015; 56:371-7. [ Links ]
10. Lee J-H, Kong S-K, Wu Z-S, et al. Class II β-tubulin is a novel marker for human tonsillar M-cells and follicular dendritic cells. Oral Pathol Med 2010; 39:533-9. [ Links ]
11. Neutra MR, Frey A, Kraehenbuhl J-P. Epithelial M-cells: gateways for mucosal infection and immunization. Cell 1996;86:345-48. [ Links ]
12. Zhang Z-Q, Schuler T, Cavert W, et al. Reversibility of the pathological changes in the follicular dendritic cell network with treatment of HIV-1 infection. Procl Natl Acad Sci USU 1999;96:5169-72. [ Links ]
13. He Y, Ong KC, Gao Z, et al. Tonsillar crypt epithelium is an important extra-central nervous system site for viral replication in EV71 encephalomyelitis. Am J Pathol 2014;184:714-20. [ Links ]
14. Haddad RI. Human papillomavirus associated head and neck cancers. Official reprint from UpToDate® 2016; www.uptodate.com. [ Links ]
15. Syrjanan S. HPV infections and tonsillar carcinoma. J Clin Pathol 2004;57:449-55. [ Links ]
16. Caberg JHD, Hubert PM, Begon DY, et al. Silencing of E 7 oncogene restores functional E-cadherin expression in human papillomavirus 16-transformed keratinocytes. Carcinogenesis 2008; 29:1441-7. [ Links ]
17. Herman L, Hubert P, Herfs M, et al. The L1 major capsid protein of HPV16 differentially modulates APC trafficking according to the vaccination or natural infection context. Eur Immunol 2010;40:3075-84. [ Links ]
18. Lajer CB, Von Buchwald C. The role of human papillomavirus in head and neck cancer. APMIS 2010;118:510-9. [ Links ]
 Correspondence:
Correspondence:
HD Miniggio
Tel: 012 521 4882
Fax: 012 521 4274
Cell: 072 869 4045
E-mail: hilde.hendrik@smu.ac.za













