Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
South African Journal of Chemistry
On-line version ISSN 1996-840X
Print version ISSN 0379-4350
S.Afr.j.chem. (Online) vol.72 Durban 2019
http://dx.doi.org/10.17159/0379-4350/2019/v72a13
RESEARCH ARTICLE
Mononuclear Schiff Base Complexes Derived from 5-Azophenylsalicylaldehyde with Co(II), Ni(II) Ions: Synthesis, Characterization, Electrochemical Study and Antibacterial Properties
Leila Kafi-AhmadiI, *; Ahmad Poursattar MarjaniII
IDepartment of Inorganic Chemistry, Faculty of Chemistry, Urmia University, Urmia, Iran
IIDepartment of Organic Chemistry, Faculty of Chemistry, Urmia University, Urmia, Iran
ABSTRACT
In the present research, 1,2-di[N-2-phenylthio-5-azophenylsalicylidene]ethane (H2L1, 1,3-di[N-2-phenylthio-5-azophenylsali-cylidene]propane(H2L2) and 1,4-di[N-2-phenylthio-5-azophenylsalicylidene]butane (H2L3) were synthesized. ML1,ML2 and ML3 complexes were prepared through the reaction of H2L1,H2L2 and H2L3 with Ni(II) and Co(II) acetate salts. The prepared ligands and their complexes were characterized with FT-IR, 1H-NMR, elemental analysis, conductivity and magnetic measurements, ESI-mass, UV-Vis spectroscopy and cyclic voltammetry. The complexes had monomeric structure according to the mass spectral data. All of the complexes had a six-coordinated octahedral geometry. The electrochemical behavior of the ligands, Ni(II) and Co(II) complexes was investigated by CV in DMF under argon atmosphere. The results show that the coordination of Ni(II) causes the ligands to be deactivated. The voltammogram of Co(II) complexes shows that the reduction of CoL1 is harder than CoL3 and CoL2 is inactive. Their antibacterial activity was investigated and the complexes exhibited better antibacterial activity than the free ligands against selective bacteria.
Keywords: Schiff base complexes, 5-azophenylsalicylaldehyde, electrochemical study, antibacterial properties.
1. Introduction
Schiff bases are synthesized through the reaction of primary amines and aldehydes or ketones. Schiff bases are extensively used as ligands in as much as their high stability and appropriate solubility in common solvents. Frequently, a geometrical constriction is imposed by the π-system in a Schiff base which it affects the electronic structure as well.1 The characteristics of metal complexes including Schiff base ligands and their wide applications have aroused significant attention. Schiff base complexes is a fast-growing field due to the numerous possible structures for the ligands varying with thealdehyde and amine used.2 Schiff base complexes have some properties like antibac-terial,3 antifungal,4 anticancer,5 antioxidant,6 anti-inflammatory,7 antimalarial,8 antiviral activity,9 and are used in oxygen storage devices,10 molecular architectures,11 lasers,12 OLED applica-tions,13 transistors14 and fluorescent sensors as well.15
Interest in the chemistry of nickel compounds have been grown in a great manner due to the finding of sulfur donor ligands to nickel in various metalloenzymes.16-19 The development of coordination chemistry and bio-mimetic chemistry are somehow dependent on molecules containing O, N, S donor sites. Specially, N2O4 and N2O2S2 binding cores containing non-cyclic hexadentate ligands are greatly suitable for this purpose.20 The reaction of a Co(II) salt with 1,2-di[N-2-phenyl-thio-5-azophenylsalicylidene]ethane led to the oxidative cleavage of the ligand, instead of forming a simple complex. C-S bond cleavage is common, especially when elements of cobalt group is used in the presence of cyclic thioether ligands and some non-cyclic ones as well. The purpose of the current research is to prepare and investigate the structure of H2L1,H2L2 and H2L3 Schiff base ligands with Co(II) and Ni(II) ions (Scheme 1).
In addition, the electrochemical properties of the prepared ligands and complexes were studied by cyclic voltammetry. Furthermore, the antibacterial effect of prepared ligands and complexes were investigated on some clinically significant bacteria like Staphylococcus aureus, Erichia coli and Bacillus subtilis.
Our group interest in the synthesis of Schiff base is motivated by the fact that Schiff bases are one of the important classes of organic compounds with a broad range of biological properties. They are often used as chelating ligands in the field of coordination chemistry and when complexed with different metals, they exhibit a broad range of biological activities, including anti-malarial, antibacterial, antifungal, etc. Furthermore, bacterium and fungi have developed resistance to the current line of treatment recently, which has to be of concern. Accordingly, novel Schiff bases with new properties and better performance is our main aim.
2. Experimental
2.1. Materials
All reagents were purchased from Merck and were used without further purification. 5-Azophenylsalicylaldehyde (1), 1,2-di(o-aminophenylthio)ethane (2), 1,3-di(o-minophenylthio)pro-pane (3) and 1,4-di(o-aminophenylthio)butane (4) were synthesized according to published procedure.20
2.2. Physical Measurements
CHNS analyses were conducted through an Elemental Vario EL III elemental analyzer. Electronic spectra were recorded in DMF solvent with a Shimadzu, UV-1650 PC spectrophotometer. FT-IR spectra were obtained by using KBr pellets on a Bruker Tensor instrument. Conductance measurements were conducted in DMF solvent with a Metrohm 712 conductometer. 1H-NMR spectrum were obtained with a Bruker Avance, 400 MHz, ultrashield in CDCl3 as a solvent and Me4Si as internal standard. Mass spectra of all compounds were recorded using LCMS-2010A of make Shimadzu. A vibrating sample magnetometer (Model PAR 155) was used to measure the magnetic susceptibility that was done at room temperature. Cyclic voltammetry (CV) measurements were carried out with an Autolab potentiostat PGSTAT 302 Eco Chemie. Melting points of the ligands and complexes were determined by using the finely powdered sample in a fused capillary using Elico melting point apparatus. The antimicrobial studies of the Schiff bases and the complexes were carried out by disc diffusion method.
2.3. Synthesis
2.3.1. Ligands Preparation
10 mmol (2.76 g, 2.90 g and 3.04 g for 2,3 and 4, respectively) of diamine solution in 15 mL of absolute ethanol was added to the solution of compound 1 (20 mmol, 4.52 g) in ethanol/dichloro-methane (5:1) (12 mL). In addition, 1 drop of glacial acetic acid was added to the aforementioned solution. The resultant mixture was kept under stirring and reaction under reflux condition for 1 h. The yielded precipitate (orange colour) was filtered out followed by washing using ethanol and ether and recrystallization from CH2Cl2 for H2L1,H2L3 and CH2Cl2/EtOH for H2L2, respectively.
2.3.2. Cobalt(ll) Complexes Preparation (CoL1, CoL2, CoL3)
1 mmol (0.249 g) of Cobalt(II) acetate tetrahydrate was added to 30 mL of ethanol, after which the obtained solution was added to a heated suspension of H2L1,H2L2 or H2L3 (1 mmol) in 10 mL of dichloromethane. Then, the prepared mixture was heated under vigorous stirring to accomplish the reaction for 4 h. In the next step, a rotary evaporator was used to evaporate the solvent in order to reduce its volume to 10 mL. Finally, collecting of the products were carried out by filtration and recrystallization from CH3CN/CH2Cl2 (1:1) for CoL1 and CoL3 and from petroleum ether/CH2Cl2 (1:1) for CoL2.
2.3.3. Nickel(II) Complexes Preparation (NiL1, NiL2, NiL3)
For this purpose, 1 mmol (0.248 g) of Ni(CH3COO)2.4H2O was added to 30 mL of ethanol, then the solution was added to a hot suspension of H2L1,H2L2 or H2L3 (1 mmol) in dichloromethane (10 mL). The obtained mixture was kept under stirring and heating under reflux for about 4 h. The solvent was evaporated using a rotary up to 10 mL. In the end, collection of the products was carried out by filtration and recrystallization CH3CN/CH2Cl2 (1:1) for NiL1, NiL2 and NiL3. The physical properties of ligands and complexes are tabulated in Table 1. Nevertheless, suitable crystals could not be prepared for single crystal diffraction studies.
2.3.4. Investigation of Antibacterial Activity by Disc Diffusion Method
The ligands and synthesized metal complexes were screened for antibacterial activity against pathogenic bacterial species Erichia coli, Staphylococcus aureus, Bacillus subtilis and comparisons were made. The paper disk diffusion method was adopted for the determination of antibacterial activity.21 This method is simple and is routinely used in hospital laboratories. The ligand/complex (30 μg) in DMF (0.01 mL) was placed on a paper disk (Whatman-model), with 5 mm in diameter with the help of a micropipette. The disks were left in an incubator for 48 h at 37 °C and then were applied to the bacteria grown on agar plates. For the preparation of agar plates for bacterial species, Mueller-Hinton agar with2%of glucose (50 g), obtained from Merck, was suspended in freshly distilled water (1 L). It was allowed to soak for 20 min and then was boiled in a water bath until the agar was completely dissolved. The mixture was autoclaved for 15 min at 120 °C, poured into sterilized Petri dishes, and then stored at 40 °C for inoculation. Inoculation was performed with a platinum wire loop that was made red hot in a flame, cooled, and then used for the application of bacterial strains. Each paper disk was placed on an already inoculated agar plate which was then incubated at 37 °C for 18 h. Activity was determined by measuring the diameter of zones showing complete inhibition (mm).
3. Results and Discussions
3.1. FT-IR Spectra of Ligands and Complexes
Although the single X-ray crystallography is a powerful technique, infrared spectra is an alternative as a suitable method to obtain sufficient information for clarifying the nature of bonding. Obtained IR bands are shown in Table 2. In the spectra associated to H2L1,H2L2 and H2L3 strong absorption bands can be seen at 1611-1618 cm-1 that could be associated to the C=N bond,22 confirming the successful synthes of the Schiff base. Moreover, the absence of C=O and N-H peaks, indicates Schiff bases condensation. In addition, a broad band centred at around 3440-3446 cm-1 is due to v(OH) of the phenolic hydroxyl group.23 The reaction of Ni(II) and Co(II) acetate salts with the Schiff bases (H2L1,H2L2,H2L3) yield NiL1, NiL2, NiL3 and CoL1, CoL2, CoL3 complexes. The C-O stretching vibration appeared as a weak band within the 1279-1286 cm-1 for the free ligands and in the 1330-1388 cm-1 region for the complexes. In the spectra related to complexes, the C-O peak was shifted to higher frequency that indicates the formation of oxygen bond between phenolic group and the metal ions.24 Furthermore, vC=N peak was shifted to lower frequencies in compare to free Schiff bases, showing a decrease in its bond order. This phenomenon is because of the coordination bond of the metal with the imine nitrogen lone pair.22 According to the results, it was possible to come to a conclusion that the binding of the OH group was the reason for disappearance of the corresponding vO-H vibration existing in the precursor ligands spectra.25 The CoL1 complex showed a strong vC=C vibration related to the -S-HC=CH2 moiety formed as a result of the C-S bond cleavage at 1525 cm-1 that was not seen in the other complexes. All of the obtained data suggested that the metals were bonded to the Schiff bases with the phenolic oxygen and the imine nitrogen. At lower frequency the complexes exhibited bands around 359-387 cm-1 which are assigned to vM-S vibration mode.
3.2. Conductance Measurements
The molar conductance of the complexes was measured for 10-3 M solutions in DMF at 26 °C. The results indicated that all complexes (except CoL1 which is an electrolyte) are non-electrolytes in DMF.26,27 According to the results (Table 1), H2L1, H2L2 and H2L3 Schiff bases were coordinated to the Ni(II) and Co(II) ions as doubly negatively charged anions. Hence, the deprotonation of two phenolic OH has generated oxygen anions that were bonded to the metal ions.
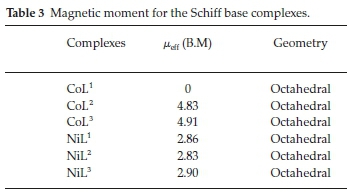
3.3. Magnetic Moments
The magnetic moments for the Ni(II) complexes were in the range of 2.83-2.90 B.M which were consistent with the octahedral stereochemistry and the magnetic moments of Co(II) complexes (CoL2, CoL3) were found to be 4.83 and 4.91 B.M, respectively. The values confirmed that the Co(II) complexes had octahedral geometry (Table 3).28 Magnetic susceptibility measurement exhibited that the CoL1 is diamagnetic, supporting the +3 oxidation state of the cobalt.
3.4. Mass Spectra of the Compounds
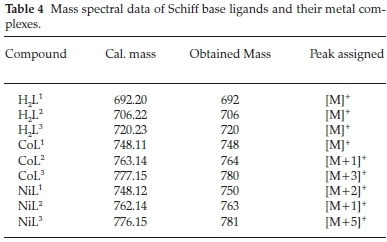
The ESI-mass spectra were measured to confirm the composition and the purity of the complexes and ligands under investigation (Table 4). The obtained mass spectra showed molecular ion peaks, which were matching with the expected values.
3.5. Electronic Spectra
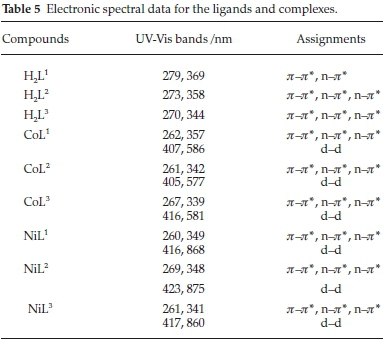
All spectra were recorded in DMF (10-3 M) at room temperature. Obtained results are shown in Table 5. The ultraviolet absorption spectrum of the Schiff base ligands demonstrated two bands positioned at 269-273 and 344-369 nm assigned to 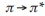 (phenyl ring) and n
(phenyl ring) and n 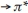 (-C=N)group.29A shift was seen to blue regions in all complexes that confirms the coordination of the ligand to the metal. Moreover, d
(-C=N)group.29A shift was seen to blue regions in all complexes that confirms the coordination of the ligand to the metal. Moreover, d  d transitions led to some new bands for the complexes in the visible region. Co(III) is a d6 system and complexes can be high spin or low spin depending upon the type of the ligand. For d6 high spin (four unpaired electron) case, the ground state term is 5D which is split into 5T2g and 5Eg states so the transition can be assigned to 5T2g
d transitions led to some new bands for the complexes in the visible region. Co(III) is a d6 system and complexes can be high spin or low spin depending upon the type of the ligand. For d6 high spin (four unpaired electron) case, the ground state term is 5D which is split into 5T2g and 5Eg states so the transition can be assigned to 5T2g 5Eg while for d6 low spin case, the ground state is 1A1g and there are two singlet excited states, 1T1g and 1T2g and the two observed spectral transitions are assigned as 1A1g
5Eg while for d6 low spin case, the ground state is 1A1g and there are two singlet excited states, 1T1g and 1T2g and the two observed spectral transitions are assigned as 1A1g 1T1g and 1A1g® 1T2g. The electronic spectrum for synthesized Co(III) complex (CoL1) showed two bands which are assigned to 1A1g
1T1g and 1A1g® 1T2g. The electronic spectrum for synthesized Co(III) complex (CoL1) showed two bands which are assigned to 1A1g 1T1g and 1A1g
1T1g and 1A1g 1T2g transition with octahedral geometry around central Co(III). The absorption spectrum of Co(II) complexes exhibited two bands at lmax= 405-116 nm and lmax= 577-586 nm. These bands correspond to the 4T1g(F)
1T2g transition with octahedral geometry around central Co(III). The absorption spectrum of Co(II) complexes exhibited two bands at lmax= 405-116 nm and lmax= 577-586 nm. These bands correspond to the 4T1g(F)  4T1gm and 4T1g(F)
4T1gm and 4T1g(F)  4A2g(F) transitions, respectively, that confirm the presence of an octahedral structure around Co(II) ion.30,31 Ni(II) complexes exhibit three bands which are assigned to 3A2g(F)
4A2g(F) transitions, respectively, that confirm the presence of an octahedral structure around Co(II) ion.30,31 Ni(II) complexes exhibit three bands which are assigned to 3A2g(F)  3T2g(F), 3A2g(F)
3T2g(F), 3A2g(F)  3T1g(F) and 3A2g(F)
3T1g(F) and 3A2g(F)  3T1g(p) transitions, respectively. Since the transition 3A2g
3T1g(p) transitions, respectively. Since the transition 3A2g 3T1g(P) generally occurs in the region of 330-400 nm in which the bands due to organic fraction of the complexes arise as well, hence this region is not useful from the point of view of drawing any conclusion about the stereochemistry around the Ni(II) centre. However, the first two low-energy bands observed in the region of 400-900 nm in all of the complexes are characteristic of nickel(II) in octahedral environment.
3T1g(P) generally occurs in the region of 330-400 nm in which the bands due to organic fraction of the complexes arise as well, hence this region is not useful from the point of view of drawing any conclusion about the stereochemistry around the Ni(II) centre. However, the first two low-energy bands observed in the region of 400-900 nm in all of the complexes are characteristic of nickel(II) in octahedral environment.
3.6. 1H NMR Spectral Studies
In the all 1H-NMR studies, chloroform was used to record the spectra. In the obtained spectra of the ligands, the singlet resonances in the δ = 13.77-13.80 ppm and multiplet peaks at δ = 7.88-8.21 ppm regions could be related to the protons of the phenolic OH groups,20 and aromatic protons32, respectively. Additionally, azomethine group protons exhibited singlet signals in the range of δ = 8.70-8.73 ppm in all of the spectra of the ligands.33 Also, aliphatic protons showed some multiple signals in the range of δ = 1.25-3.16 ppm.23 No signals were observed in the spectrum related to the free amino and aldehyde groups that confirms the formation of the Schiff base ligands. The 1H-NMR spectrum of the complexes showed signals located at δ = 7.90-8.15 ppm and δ = 7.30-8.08 ppm for Ni(II) and Co(II) complexes, respectively. Aforementioned signals were attributed to the aromatic protons chemical shifts.34 In the complexes and during the chelation process, the azomethine proton signal of ligands is shifted to low field that affirms the bonding to the metal ions through azomethine nitrogen. Furthermore, the signal of the phenolic proton in the free ligands was not seen that confirmed the deprotonation of the phenolic group on complexes formation.20 In 1H-NMR spectra of Co(II) complexes (CoL2 and CoL3), the paramagnetic metal ion effect on chemical shifts is observed. On the contrary, no paramagnetic shifts was seen for CoL1 that affirms the presence of a Co(III) centre as a result of Co(II) oxidation. The peak observed at δ = 3.17 ppm was because of the bridging -S-CH2-CH2-S- moiety in H2L1 disappears in the spectrum of CoL1. In addition, two new multiplets appeared in the region of δ = 5.57-6.04 ppm were related to the protons of vinyl (-S-CH=CH2).20These observations in 1H-NMR spectrum of CoL1 show the cleavage of C-S bond accompanied by Co+2 oxidation to Co+3. However, no cleavage of C-S bond and metal oxidation occurs in CoL2 and CoL3 complexes.
3.7. Electrochemical Studies
The pathway of the chemical reactions can be demonstrated by electron transfer as one of the most effective methods. The electrochemical behavior of the ligands, Ni(II) and Co(II) complexes have been investigated by cyclic voltammetry in DMF under argon atmosphere in the potential range from +2.0 to -2.0 V The CV of ligands show at the range of scan rate of 0.05-0.4 V s-1. It can be observed that there are one cathodic peak and one anodic peak. The Ep and Ip values of the various peaks are listed in Table 6.
The cyclic voltammogram of Ni(II) complexes show at a scan rate of 0.3 V s-1. On comparing the voltammogram of ligands and their Ni(II) complexes, it is evident that the oxidation and reduction peaks appeared in the voltammogram of Schiff bases were totally disappeared on complexation. Thus, the coordination of Ni(II) causes the ligands to be deactivated. Also, in voltammo-gram no peak related to Ni(I) and Ni(III) was found.
The cyclic voltammetric curves for the electrochemical reduction of the CoL1 and CoL3 exhibit a redox couple, which can be attributed to the reduction of the metal centre. The comparison of the voltammogram of Co(II) complexes shows that the reduction of CoL1 is harder than CoL3. For CoL1 and CoL3, the reduction couple (Com/Con) was at -0.475 V and 0.0095 V, respectively, but CoL2 was inactive (Table 7).
3.8. Antibacterial Activity of Ligands and Complexes in Agar Medium
The antibacterial activity of the prepared ligands and complexes was investigated using Ampicillin as standard. They affected bacterial species' growth in different manner. The results and the diameter of inhibition zones (mm) are presented in Table 8. Furthermore, the biological activities of the Schiff bases were less than that of the standard, Ampicillin. In addition, metal complexes were more effective than the ligands. Nevertheless, this phenomenon is probably because of the more lipophilic feature of the complexes. Additionally, the enhanced metal chelates activity can be clarified based on Overtone's concept and Tweedy's chelation theory.35 According to Overtone's concept of cell permeability, only the lipid soluble materials are let to pass the lipid membrane surrounding the cell. In this respect, liposolubility is a substantial factor determining the anti-microbial activity. When chelation occurs, an overlap occurs between the orbital of the ligand and metal ion's positive charge partial sharing with donor groups, as a result, the polarity of the metal ion will be reduced as a consequence.36 Moreover, the π electrons over the chelate ring will be further delocalized leading to higher lipophilicity of the complex. As a consequence, the complexes will penetrate into lipid membrane more, so the microorganism's enzymes binding sites will be blocked. In addition, respiration process of the cells can be disrupted by metal complexes, as a consequent, the synthesis of proteins will be blocked and growth of the organism will be limited.37 The effectiveness of complexes against organisms relies on the impermeability of the microbe's cells or difference in ribosomes of microbial cells. Furthermore, the data showed that Erichia coli and Staphylococcus aureus were been inhibited in a greater degree by the Co(II) and Ni(II) complexes respectively; however, none of the compounds were active against Bacillus subtilis.
4. Conclusions
In this research, Schiff base ligands and prepared complexes with Ni(II), Co(II) ions were successfully synthesized and characterized. The analytical results showed 1:1 metal:ligand stoichiometry. According to the elemental CHNS analysis, FT-IR, 1H-NMR spectral studies, Schiff base ligands acted as N2O2S2 ligands. The molar conductance values in complexes revealed the presence of non-electrolytic compounds (except CoL1 that is electrolyte). The magnetic moments and electronic spectra studies suggested an octahedral structure. Reaction of H2L1 with Co(II) salt resulted in oxidative cleavage of the C-S bond to produce a Co(III) product. In addition, the effects of prepared ligands and complexes were investigated on some bacteria such as Erichia coli, Staphylococcus aureus and Bacillus subtilis. Synthesized complexes exhibited appropriate antibacterial effect.
Acknowledgement
The authors are grateful to Urmia University for supporting this work.
ORCID iDs
L. Kafi-Ahmadi: orcid.org/0000-0001-8947-2706
A. Poursattar Marjani: © orcid.org/0000-0002-5899-4285
References
1 R.K. Al-Shemary, A.M.A. Al-Khazraji and A.N. Niseaf, Preparation, spectroscopic study of Schiff base ligand complexes with some metal ions and evaluation of antibacterial activity, Pharm. Innov. J., 2016, 5, 81-86. [ Links ]
2 G. Kumar, D. Kumar, C.P. Singh, A. Kumar and V.B. Rana, Synthesis, physical characterization and antimicrobial activity of trivalent metal Schiff base complexes, J. Serb. Chem. Soc., 2010, 75, 629-637. [ Links ]
3 L. Kafi-Ahmadi, A. Poursattar Marjani and M. Pakdaman-Azari, Synthesis, characterization and antibacterial properties of N,N-bis(4-dimethylaminobenzylidene)benzene-1,3-diamine as new Schiff base ligand and its binuclear Zn(II), Cd(II) complexes, S. Afr. J. Chem., 2018, 71, 155-159. [ Links ]
4 A.P. Sangamesh, T.P. Chetan, M.H. Bhimashankar, S.T. Shivakumar and S.B. Prema, DNA cleavage, antibacterial, antifungal and anthelmintic studies of Co(II), Ni(II) and Cu(II) complexes of coumarin Schiff bases: synthesis and spectral approach. Spectrochim Acta A., 2015,137, 641-651. [ Links ]
5 M.A. Mokhles, A.L. Ammar, A.M. Hanan, A.M. Samia, M.A. Mamdouh and A.E. Ahmed, Synthesis, anticancer activity and molecular docking study of Schiff base complexes containing thiazole moiety, Beni-Seuf Univ. J. Appl. Sci., 2016, 5, 85-96. [ Links ]
6 M. Kumar, T. Padmini and K. Ponnuvel, Synthesis, characterization and antioxidant activities of Schiff bases are of cholesterol, J. Saudi. Chem. Soc, 2017, 21, S322-S328. [ Links ]
7 L. Jia, J. Xu, X. Zhao, Sh. Shen, T. Zhou, Z. Xu, T. Zhu, R. Chen, T. Ma, J. Xie, K. Dong and J. Huang, Synthesis, characterization, and antitumor activity of three ternary dinuclear copper(II) complexes with a reduced Schiff base ligand and diimine coligands in vitro and in vivo, J. Inorg. Biochem, 2016,159, 107-119. [ Links ]
8 J.F. Adediji, E.T. Olayinka, M.A. Adebayo and O. Babatunde, Anti-malarial mixed ligand metal complexes: synthesis, physicochemical and biological activities, Int. J. Phys. Sci., 2009, 4, 529-534. [ Links ]
9 E.L. Chang, C. Simmers and D.A. Knight, Cobalt complexes as antiviral and antibacterial agents, Pharmaceuticals, 2010, 3, 1711-1728. [ Links ]
10 N. Chantarasiri, T. Tuntulani, P. Tongraung, R. Seangprasertkit-Magee and W. Wannarong, New metal-containing epoxy polymers from diglycidyl ether of bisphenol A and tetradentate Schiff base metal complexes, Eur. Poly. J, 2000, 36, 695-702. [ Links ]
11 I.P. Oliveri, S. Failla, G. Malandrino and S. Di Bella, New molecular architectures by aggregation of tailored zinc(II) Schiff-base complexes, New J. Chem., 2011, 35, 2826-2831. [ Links ]
12 S.Gh. Musharraf, A. Bibi, N. Shahid, M. Najam-ul-Haq, M. Khan, M. Taha, U.R. Mughal and Kh.M. Khan, Acylhydrazide and isatin Schiff bases as alternate UV-laser desorption ionization (LDI) matrices for low molecular weight (LMW) peptides analysis, Am. J. Analyt. Chem., 2012, 3, 779-789. [ Links ]
13 P. Kathirgamanathan, S. Surendrakumar, J. Antipan-Lara, S. Ravic-handran, Y.F. Chan, V. Arkley, S. Ganeshamurugan, M. Kumaraverl, G. Paramswara, A. Partheepan, V.R. Reddy, D. Bailey and A.J. Blake, Novel lithium Schiff-base cluster complexes as electron injectors: synthesis, crystal structure, thin film characterisation and their performance in OLEDs, J. Mater. Chem., 2012, 22, 6104-6116. [ Links ]
14 A. Wachi, Y. Kudo, A. Kanesaka, H. Nishikawa, T. Shiga, H. Oshio, M. Chikamatsu and R. Azumi, Organic field-effect transistor based on paramagnetic Cu(II) neutral complexes coordinated by Schiff base-type TTF ligands, Polyhedron., 2017,136, 70-73. [ Links ]
15 V. Nishal, D. Singh, R.K. Saini, V. Tanwar, S. Kadyan, R. Srivastava and P.S. Kadyan, Characterization and luminescent properties of zinc-Schiff base complexes for organic white light emitting devices, Cogent Chem., 2015,1, 1079291. [ Links ]
16 S.G. Rosenfield, W.H. Armstrong and P.K. Mascharak, Convenient synthesis and properties of (R4N)2[Ni(SAr)4](Ar = C6H5, p-C6H4Cl, p-C6H4CH3, and m-C6H4Cl) and the structure of tetra ethyl ammonium tetra kis(p-chlorobenzenethiolato)nickelate(II), Inorg. Chem., 1986, 25, 3014-3018. [ Links ]
17 W. Rosen and D.H. Busch, Octahedral nickel(II) complexes of some cyclic polyfunctional thioethers, Inorg. Chem., 1970, 9, 262-265. [ Links ]
18 S. Chandrasekhar and A. McAuley, Syntheses and reactivity of nickel complexes of 8-aza-1,5-dithiacyclodecane and the macrobicyclic ligand 1,2-bis(8-aza-1,5-dithia-8-cyclodecanyl)ethane, Inorg. Chem., 1992, 31, 2234-2240. [ Links ]
19 C.P. Kulatilleke, S.N. Goldie, M.J. Hegg L.A. Ochrymowycz and D.B. Rorabacher, Formation and dissociation kinetics and crystal structures of nickel(II)-macrocyclic tetrathiaether complexes in aceto-nitrile. Comparison to nickel(II)-macrocyclic tetramines, Inorg. Chem., 2000, 39, 1444-1453. [ Links ]
20 G. Rajsekhar, C.P. Rao, P. Saarenketo, K. Nattinen and K. Rissanen, Complexation behaviour of hexadentate ligands possessing N2O4 and N2O2S2 cores: differential reactivity towards Co(II), Ni(II) and Zn(II) salts and structures of the products, New. J. Chem., 2004, 28, 75-84. [ Links ]
21 A. Reiss, A. Samide, G. Ciobanu and I. Dabuleanu, Synthesis, spectral characterization and thermal behaviour of new metal(II) complexes with Schiff base derived from amoxicillin, J. Chil. Chem. Soc., 2015,60, 3074-3079. [ Links ]
22 M.K.B. Break, M.I.M. Tahir, K.A. Crouse and T.J. Khoo, Synthesis, characterization, and bioactivity of Schiff bases and their Cd2+,Zn2+, Cu2+ and Ni2+ complexes derived from chloroacetophenone isomers with S-benzyldithiocarbazate and the X-Ray crystal structure of S-benzyl-B-N-(4-chlorophenyl)methylenedithiocarbazat, Bioinorg. Chem. Appl., 2013, 2013, 1-13. DOI: 10.1155/2013/362513 [ Links ]
23 R. Arab Ahmadi and S. Amani, Synthesis, spectroscopy, thermal analysis, magnetic properties and biological activity studies of Cu(II) and Co(II) complexes with Schiff base dye ligands, Molecules, 2012, 17, 6434-6448. [ Links ]
24 N.P. Singh, P. Saini and A. Kumar, Synthesis and spectroscopic characterization of some novel Schiff base compounds of transition metal and their biological studies, RJC, 2013, 6, 190-195. [ Links ]
25 H.A. Bayoumi, Characteristic studies of hexamethylene diamine complexes, Inter. J. Inorg. Chem., 2013, 2013, 1-12. DOI: 10.1155/2013/458018 [ Links ]
26 R.J. Angelici, Synthesis and Technique in Inorganic Chemistry, 2nd edn.,Saunders Company, 1977, p 221. [ Links ]
27 M.A. Ali, A.H. Mirza and R.J. Butcher, Synthesis and characterization of Copper(II) complexes of the methyl pyruvate Schiff base of S-methyldithiocarbazate (HMPSME) and the X-crystal structures of HMPSME and [Cu(MPSME)Cl], Polyhedron, 2001, 20,1037-1043. [ Links ]
28 S. Jyothi, K. Sreedhar, D. Nagaraju and S.J. Swamy, Synthesis and spectral investigation of Co(II), Ni(II), Cu(II) and Zn(II) complexes with novel N4 ligands, Can. Chem. Tran, 2015, 3, 368-380. [ Links ]
29 N.K. Chaudhary and P. Mishra, Metal complexes of a novel Schiff base based on penicillin: characterization, molecular modeling, and antibacterial activity study, Bioinorg. Chem. & Appl., 2017,2017,1-13. DOI: 10.1155/2017/6927675 [ Links ]
30 C.R. Hammond, The Elements, In Handbook of Chemistry and Physics, 1st edn., New York. 2000. [ Links ]
31 A.S. Abu-Khadra, R.S. Farag and A.El.M. Abdel-Hady, Synthesis, characterization and antimicrobial activity of Schiff base (E)-N-(4-(2-hydroxybenzylideneamino)phenylsulfonyl) acetamide metal complexes, AJAC, 2016, 7, 233-245. [ Links ]
32 N.H. Al-Saadawy, F.F. Alyassin and H.R. Faraj, Preparation and characterization of some new complexes of Schiff bases derived from benzoin and glycine, GJPACR, 2016,4, 13-20. [ Links ]
33 M. Shibata and H. Kandori, FTIR studies of internal water molecules in the Schiff base region of bacteriorhodopsin, Biochemistry, 2005, 44, 7406-7413. [ Links ]
34 J.G. Gilbert, A.W.W. Addison and R.J. Butcher, Some nickel(II) complexes with pentadentate and tridentate heterocyclic N- and S-donor ligands, Inorg. Chim. Acta., 2000, 308, 22-30. [ Links ]
35 B.G. Tweedy, Plant extracts with metal ions as potential antimicrobial agents, Phytopathology, 1964, 55, 910-914. [ Links ]
36 J. Parekh, P. Inamdhar, R. Nair, S. Baluja and S. Chanda, Synthesis and antibacterial activity of some Schiff bases derived from 4-amino- benzoic acid, J. Serb. Chem. Soc., 2005, 70, 1155-1161. [ Links ]
37 Y. Vaghasia, R. Nair, M. Soni, S. Baluja and S. Chanda, Synthesis, structural determination and antibacterial activity of compounds derived from vanillin and 4-aminoantipyrine, J. Serb. Chem. Soc.,2004, 69, 991-998. [ Links ]
Received 8 January 2019
Revised 23 May 2019
Accepted 23 May 2019
* To whom correspondence should be addressed. E-mail: l.kafiahmadi@urmia.ac.ir














