Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
Journal of the South African Veterinary Association
On-line version ISSN 2224-9435
Print version ISSN 1019-9128
J. S. Afr. Vet. Assoc. vol.81 n.4 Pretoria Dec. 2010
ARTICLE ARTIKEL
Risk factors for smallholder dairy cattle mortality in Tanzania
E S SwaiI,*; E D KarimuriboII; D M KambarageII
IVeterinary Investigation Centre, PO Box 1068, Arusha, Tanzania
IIDepartment of Veterinary Medicine and Public Health, Sokoine University of Agriculture, Box 1320, Morogoro, Tanzania
ABSTRACT
A retrospective cross-sectional study of mortality was conducted on smallholder dairy farms in 2 separate regions (Iringa and Tanga) of Tanzania during the period of January to April 1999. A total of 1789 cattle from 400 randomly sampled smallholder dairy farms (200 each from Iringa and Tanga regions) were included in the study. These animals contributed a total risk period of 690.4 and 653.95 years for Tanga and Iringa, respectively. The overall mortality rates were estimated to be 8.5 and 14.2 per 100 cattle years risk for Tanga and Iringa regions, respectively; 57.7 % of the reported deaths were of young stock less than 12 months old; 45 % of reported young stock deaths (<12 months old) were due to tickborne diseases, mainly East Coast Fever (ECF) and anaplasmosis. Disease events including ECF were reported to occur in all months of the year. Survival analysis using Cox proportional hazard models indicated that, in both regions, death rate and risk was higher in young stock less than 12 months than in older animals (relative risk RR = 4.92, P <0.001 for Iringa; RR = 5.03 P = 0.005 for Tanga). In the Tanga region reported mortality rates were significantly higher for male animals (RR = 3.66, P = 0.001) and F2 compared with F1 animals (RR = 3.04, P = 0.003). In the Iringa region, reported mortality rates were lower for cattle on farms where the owner had attended a dairy development project training course (RR = 0.47, P = 0.012). Farms located in Iringa urban district and Pangani were associated with higher risk (mortality risk 21 % for Iringa urban and 34 % for Pangani). Our findings suggest that timely health and management interventions on these factors are necessary to alleviate losses from disease and emphasise that understanding variation in mortality risk within a population can enhance early response to potential outbreaks, reducing losses.
Keywords: dairy cattle, mortality, risk factors, smallholder, Tanzania.
INTRODUCTION
The dairy industry, dominated by smallholder farmers, has been recognised as one of the most important industries in Tanzania in the quest to attain human food security and good welfare13, 21, 25. The smallholder farmers often keep fewer than 10 cattle with milk yields of less than 10 litres per cow per day and 89 % are zero-grazed25, 29. The farmers are increasing the use of exotic dairy cattle and their crosses in order to increase their milk yields 18. However, these exotic cattle are less tolerant to local diseases which may result in high mortality24.
Currently, country demand for milk exceeds production and there is a projected growth of the sector19. In anticipation of this growth, potential production constraints, among them animal health, need to be identified. Efficient production and limited losses are important for the farmers to realise maximum benefits from their enterprises. In order to minimise these losses, the causes of morbidity and mortality and the associated risk factors need to be identified and appropriate control measures implemented1, 5, 13, 22. However, little is known about the causes of morbidity and mortality and their risk factors on smallholder dairy farms in Tanzania.
A fuller understanding of the causes of dairy stock deaths and mortality patterns will help in: 1) identifying major management problems in the herds and hence areas for improvement; 2) guiding research efforts; and 3) guiding extension personnel, veterinarians and policy makers to the important management and disease control problems on dairy farms.
In this study we first aimed to identify the major causes of mortality of smallholder dairy cattle in 2 regions of Tanzania, and secondly to identify and quantify potential animal and management risk factors for these causes. The purpose of this study was to generate baseline epidemiological data that could facilitate the development of effective interventions to control mortality on smallholder dairy cattle.
MATERIALS AND METHODS
Study sites and population
This study was conducted on smallholder dairy farms in 2 separate regions (Iringa and Tanga) of Tanzania.Tanga region is situated on the northeastern corner of Tanzania (longitude 36ºE and 38ºE and latitude 4º and 6ºS) and Iringa region is 1 of 3 in the southern highland zone of Tanzania and lies between latitude 07º39'and 08º06'S and longitude 35º30' and 36º04'E. Detailed information on study areas are described elsewhere24. The type of animals kept in smallholder units includes taurus breeds (Friesian, Ayrshire, Jersey, Simmental) and crosses of these breeds with indicus breeds (Tanzania shorthorn zebu, Boran and Sahiwal). The level of genetic make-up from taurus breeds varies from 50-85 %. Animals with taurus breed genetic makeup of 50 %, 62.5 % and above were classified as F1, F2 and F3, respectively. Studies were carried out in 4 administrative districts of Tanga region and 2 administrative districts in Iringa Region.
Study design and sample size estimation
This retrospective cross-sectional study was conducted between January and April 1999. The sample size of 200 farms, randomly selected from each region, was estimated using Epi-Info version 6.04b6 (CDC, Atlanta, USA) in order to provide 80 % power to detect a relative risk of 2.0 with 95 % confidence and 'design effect' of 2.08. Exposure to disease was estimated to occur in 40 % of the cattle population in which 5 % of unexposed died. Farms in each study region were randomly selected from a sampling frame of 3001 in Tanga and 500 in Iringa, using the databases of the Tanga and Iringa Dairy Development Projects. The average herd size was estimated to be 3 cattle. A smallholder dairy farm was defined as one with 10 cattle or fewer (of all ages and sexes).
Data collection
Information was gathered through a pre-tested, structured questionnaire, which was administered on a single day visit, on all 200 farms in each region. Of necessity, data were collected from farms by 2 separate teams, 1 in each region. Two of the authors, ESS and EDK, personally administered questionnaire in Tanga and Iringa region, respectively.
The information collected concerned the farm management events that occurred during 1998. Farmers were asked to give details of cattle that were alive at any stage during 1998. Information collection involved detailed tracing of all animals on the farm and examination of written records such as date of birth, deaths, movement on and off farm. Information collection procedures continued until all the ages of the cattle, dates of birth, dates of deaths and movements on to and off the farm agreed chronologically. Detailed information on variables investigated are described elsewhere24, 27. Identified animal and farm-level management variables were explored individually or together in multivariate regression models.
Data analysis
Mortality rate was estimated using the following equation: Mortality rate(λ)= 
Animal days at risk are the total number of days the study animals were present during the year under study. An animal's number of days present during the study was calculated as the difference between its date of exit (or end of December 1998) and its date of entry (or start of 1998).
Mortality rate was converted into mortality risk from the following equation:
Mortality risk (η) = 1 - e -λ ,
where e = natural base logarithm and -λ = exponentiated mortality rate.
Mortality risk or risk rate (expressed as a percentage) is defined as the probability of an animal not surviving 1 year, assuming mortality events are exponentially distributed16.
Mortality risks were estimated for various animal-and farm-level factors. For mortality estimates, the farm was the primary sampling unit. The study population was all dairy stock that was alive at any time during 1998. The outcome variable was the time to death or censoring (whether the animal left the farm or reached the end of the study period). Individual animal-and farm-level risk factors examined for both study sites, and their categories are detailed in Table 1. Data were analysed using Epi-Info version 6.04d6 (CDC, Atlanta, USA), S-plus 2000 (Math soft version Inc.) and EGRET for Windows version 2.04

Survival analysis and statistical modelling
The non-parametric method of Kaplan- Meier11 plot and semi-parametric2 proportional hazard models were used to explore the determinants of time to death as a result of all causes. Farm IDs as a random effect (frailty term)28 were added to the model in order to allow for possible variation attributed by farms. Multivariate models were used to explore the relationship between mortality and animal-and farm-level variables. The final models were constructed by a forward stepwise procedure and the criteria for inclusion and exclusion was a change of deviance significant at the 5 % level according to the maximum likelihood ratio test-chi-square distribution. Kaplan-Meier survival curves were plotted to show relationships between survival and key animal- and farm-level variables.
RESULTS
Descriptive statistics
All 200 farms from Tanga and Iringa were visited and interviewed during the period of January 1999 to April 1999 (100 % response rate). Between January and December 1998, 376 animals left the study areas from both sites due to various reasons, including 78 (21 %) sold for slaughter, 116 (31 %) sold for breeding or paying back credit, 148 (39 %) animals died and 34 (9 %) left for other reasons including gifts. During the same period, 532 animals entered the study area as a result of birth (405; 76 %) or purchases for breeding (127; 24 %). At the end of the study, data were available for 894 and 895 animals for Tanga and Iringa regions, respectively. These animals were alive at some stage during 1998 and contributed to the period risk of 653.95 and 690.4 years for Iringa and Tanga, respectively. The distribution and annual mortality risks for each farm-level factor are summarised in Table 1.
Reported causes of death and temporal pattern
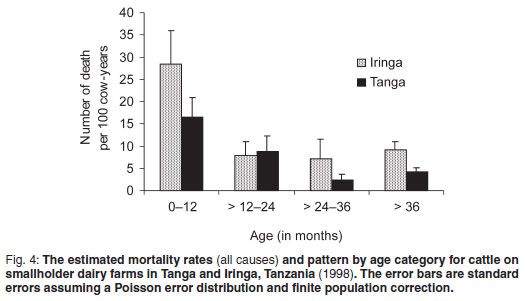
The major reported causes of death and monthly pattern are shown in Figs 1 and 2. Tick-borne diseases (TBDs), specifically ECF, were reported to be the major cause of death. Deaths (all causes) were reported to occur in all months of 1998. Deaths due to ECF were reported to occur in all months of the year except January (Fig. 3). Although not statistically significant, ECF mortality rates were highest from August to December.



Mortality rate estimates
Of the 1789 dairy cattle from both regions that were reported to have been alive at some stage in 1998, 148 were reported to have died between January and December 1998 including 4 stillbirths. Of these, 58 (39 %) were males and 90 (61 %) were females. Of the recorded deaths, 93 (62 %) were reported from Iringa and 55 (36 %) from Tanga, respectively. The overall estimated mortality rates by administrative region were 8.5 and 14.2 per 100 cattle years in Tanga and Iringa, respectively.
Factors associated with variation in mortality
Effect of age
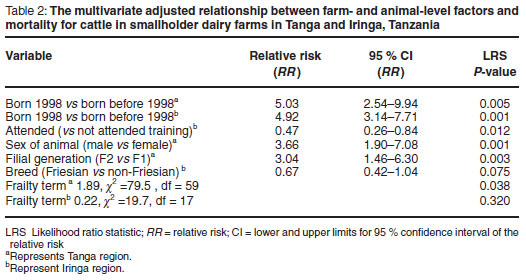
Eighty-five (57.5 %) of 148 reported deaths were of young age stock <12 months old. Mortality in the 1-2 years age category represented 15.5 % of total mortality whereas mortality in animals over 2 years old represented 27 % of all mortality. Nearly half (45 %) of the recorded mortalies in young stock less than 12 months old were related to TBDs, mainly ECF and anaplasmosis (Fig. 4). Mortality risk was well described after stratification of age of study animals as being born in 1998 (young stock) and before 1998 (older-age stock) (Fig. 5). After allowing for confounding effect of sex and breed, higher relative risk was observed for young stock than older animals in both study regions (RR = 5.03, P = 0.005 and RR = 4.92, P = 0.001, Table 2 for Tanga and Iringa regions, respectively).

Effect of animal sex
The mortality risk by sex category is shown in Table 1 and Fig. 6. The results of multivariate analyses are summarised in Table 2. After allowing for confounding by age and breed, male animals in Tanga were 3 times more likely to die than females (RR = 3.66, P = 0.001). However, in Iringa there was no significant difference in the mortality rates of males and females (P = 0.690).
Effect of the level of taurus genetic make-up
In both regions, F1 animals were associated with increased mortality risk compared with F2 and F3 (Table 1). This variable was strongly confounded by the age and the source of animals. After allowing for confounding with age, F2 animals in Tanga were 3 times more likely to die than F1 animals from multivariate survival analysis model estimate (RR = 3.04, P = 0.003). Mortality amongst F3 was not significantly different when compared with F1 (P = 0.170). In Iringa, mortality rates between F1 and F2 and F3 were not significantly different (P = 0.910).
Effect of ECF immunisation
In a univariable (Cox-proportional hazard model) analysis, immunised animals in Tanga were associated with lower death rates compared with non-immunised animals (RR = 0.52, P = 0.001, Table 1). The relationship in Iringa was not significant (P = 0.970). This variable was strongly confounded by age, sex and level of taurus genetic make-up. However, in both regions, the sample size of the vaccinated animals was too small to make any meaningful statistical conclusion.
Attended training course
In both regions, mortality risk for cattle belonging to farmers trained in basic animal husbandry was less compared with that in cattle belonging to untrained farmers in the univariate survival regression models (RR = 0.52, P = 0.053 and RR = 0.30, P = 0.001 for Tanga and Iringa respectively, Fig. 7). Training remained as an explanatory variable for mortality risk in Iringa in the multivariate model. Training demonstrated a protective effect from the multivariate regression model analysis (RR = 0.47, P = 0.012). In Tanga, training was not significantly associated with mortality (P = 0.061).
Effect of grazing
On univariable analyses, grazed animals were significantly associated with decreased mortality in Iringa but not in Tanga (RR=0.64, P=0.050 for Iringa and RR = 1.73, P = 0.230 for Tanga) (Table 1). After allowing for confounding with age, sex and farm, grazing was not significantly associated with mortality in the final multivariate models.
Effect of geographical location
The mortality risk for each district category is reported in Table 1. Animals from Pangani (mortality risk 34 %) in Tanga and Iringa Urban district (mortality risk 21 %) in Iringa had a significantly higher death rate compared with animals in other districts. Farm classification (urban, peri-urban and rural) and agro-ecological zone (AEZ) confounded this variable. However, in both regions and in both models, geographical location of animals was not significantly associated with mortality (P = 0.333 and P = 0.960 for Pangani and Iringa Urban, respectively). No other variables examined were significantly associated with mortality from the 2 studied regions.
DISCUSSION
In both regions, the 'young stock' age group was at a higher risk than older stock. This finding is consistent with other studies in East and Central Africa7, 9, 15. In our study young stock mortality was a significant problem; nearly half of the young stock died within the 1st year of life. Age was considered as static variable and non-time dependent covariate. Including it as a time dependent variable made no difference to the conclusion (data not shown here). Some studies in the USA have shown that in well-managed dairy herds, young stock mortality does not exceed 5 % from birth to 30 days of age23. If young stock mortality rate is taken as an indicator of quality of husbandry practice, then it would appear husbandry practices on most smallholder farms was generally poor because of the high mortality of 16-25 per 100 animal year-risk observed. This observation is well supported by the higher rate of unknown and non-infectious causes of deaths reported in this study, suggesting the need to improve husbandry practices.
Despite the fact that studied animals were at risk of a variety of causes of mortality, mortality owing to TBDs (ECF and anaplasmosis) was reported to account for over of all deaths, consistent with other studies in some parts of Tanzania12, 17, 22 . This observation confirms our findings that current intensive tick control methods used on the smallholder farms20 are ineffective in preventing animals from being exposed to infective ticks26. Similar observations have been reported in smallholder dairy farms of coastal regions of Tanzania and Kenya15, 24. Our observations suggest the need for a re-assessment of recommendations for tick control on smallholder dairy farms in these regions of Tanzania.
of all deaths, consistent with other studies in some parts of Tanzania12, 17, 22 . This observation confirms our findings that current intensive tick control methods used on the smallholder farms20 are ineffective in preventing animals from being exposed to infective ticks26. Similar observations have been reported in smallholder dairy farms of coastal regions of Tanzania and Kenya15, 24. Our observations suggest the need for a re-assessment of recommendations for tick control on smallholder dairy farms in these regions of Tanzania.
Mortality rates for male animals in Tanga were higher than for those in female cattle, perhaps reflecting the relative value attached to female stock, either as future replacement stock or as animals for sale. This may also reflect the high economic cost of feeding male calves with milk in the specialist dairy farming system7, 9. The lower observed mortality rates for male animals than in females in Iringa may warrant prospective studies aiming at looking for detailed sex-specific variables associated with mortality in this region.
The attendance of a farmer at a training course appeared to be protective against mortality of his or her animals. This suggests the extension messages of existing (or past) training courses have been effective in reducing mortality. The effect of training on mortality may have been due to early recognition and treatment of clinical cases. In Tanga, training was not significantly associated with mortality. Lack of association could be linked to the nature of the diseases, i.e. ECF, which is highly prevalent and difficult to treat once established and the lack of efficient animal health delivery services in most of the rural setting.
Animals with high levels of taurus genetic make-up (F2 and F3) had higher mortality risk than F1 after stratifying by age. Some studies in East Africa and elsewhere in the tropics have shown that F1 cattle are better able to acclimatise to a tropical environment than are taurus cattle or their crosses (above F1)10. Their ability to withstand disease, harsh nutritional and environmental condition, most likely due to heterosis3, may place F1 crossbred at an advantage over those with a higher level of taurus blood despite their generally lower milk yield.
CONCLUSION
In this study, reported mortality rates were very high, particularly for animals aged less than 12 months, suggesting that improvements in management are needed. There was further evidence that disease events were spread throughout the year, with no discernible monthly pattern, suggesting strict and closer following of disease control packages (if any exist). Tick-borne diseases were identified as a major cause of mortality, but detailed studies of cause-specific mortality are required to confirm how reliable farmer reporting is at identifying these causes. If the reports are correct, tick and tick-borne disease control strategies on these farms should be re-addressed. Male calves were more likely to die than females (but in Tanga only), possibly because of lack of care due to their low economic value. Although our study suggests that farmer training programmes used to date seem to have been effective in reducing young stock mortality, the need to further improve husbandry practices was highlighted.
ACKNOWLEDGEMENTS
We thank the Government of UK through DFID/NRRD Animal Health Research programme for financing this work. Generous cooperation of smallholder 's dairy farmers, and extension staff is also acknowledged. Thanks are extended to the Director of Veterinary Service for permission to publish this work.
REFERENCES
1. Ahmad R, Amin A, Kazmi S E 1986 Studies on the bacterial causes of calf mortality. Pakistan Veterinary Journal 6: 116-118 [ Links ]
2. Cox D R 1972 Regression models and life table (with discussion). Journal of the Royal Statistical Society, Series B, 34: 187-220 [ Links ]
3. Cunningham E P, Syrstad O 1987 Cross breeding Bos indicus and Bos taurus for milk production in the tropics. FAO Animal Production and Health Paper No. 68, FAO, Rome [ Links ]
4. Cytel Software Corporation 1999 Statistics and Epidemiology Research Corporation, version 2.0. Seattle, USA. [ Links ]
5. Debnath N C, Taimu M J FA, Sana A K, Ersaduzaman M, Helaluddin M, Rahman M L, Roy D K, Islam M N 1995 A retrospective study of calf losses on the central dairy cattle breeding station in Bangladesh. Preventive Veterinary Medicine 24: 43-53 [ Links ]
6. Epi-info 1996 Centres for Disease Control, version 6.04d, Atlanta, USA, and Geneva, Switzerland [ Links ]
7. French N P, Tyrer J, Hirst W M 2001 Smallholder dairy farming in the Chikwakwa communal land, Zimbabwe: birth, death and demographic trends. Preventive Veterinary Medicine 48: 101-112 [ Links ]
8. French N P, Tyrer J 1997 Birth and death of cattle on small scale dairy farms in Zimbabwe. Proceedings of the Society of Veterinary Epidemiology and Preventive Medicine, Chester, UK, 1997: 40-45 [ Links ]
9. Gitau G K, McDermott J J, Waltner-Toews D, Lissemore K D, Osumo J M Muriuki D 1994 Factors influencing calf morbidity and mortality in smallholder dairy farms in Kiambu District, Kenya. Preventive Veterinary Medicine 21: 167-177 [ Links ]
10. Kahi A K, Thorpe W, Nitter G V, Gall C F 2000 Cross breeding for dairy production in the lowland tropics of Kenya. Prediction of performance of alternative crossbreeding strategies. Livestock Production Science 63: 55-63 [ Links ]
11. Kaplan E L, Meier P 1958 Non-parametric estimation from incomplete observation. Journal of the American Statistical Association 53: 457-481 [ Links ]
12. Kifaro G C, Temba E A 1990 Calf mortality and culling rates in two dairy farms in Iringa Region, Tanzania. Proceedings of the 17th Scientific Conference of the Tanzania Society of Animal Production (TSAP), Arusha, 25-27 September 1990: 23-28 [ Links ]
13. Lema B E, Banda G 1991 Infectious causes of calf diarrhoea in selected regions of Tanzania. Proceedings of the 9th Tanzania Veterinary Association Scientific Conference, Arusha, 2-4 December: 216-221 [ Links ]
14. Leslie J, Swai E S, Karimuribo E, Bell C 1999 Tanga and Southern Highlands Dairy Development Programme: social economic aspects and farmer perception of animal disease. (DFID/NRRD) Animal Health Research Programme: 23-45 [ Links ]
15. Maloo S H, Thorpe W, Kioo G, Ngumi P, Rowland G J, Perry B D 2001 A longitudinal study of disease incidence and case fatality risks on small-holder dairy farms in Coastal Kenya. Preventive Veterinary Medicine 52: 17-29 [ Links ]
16. Martin S W, Meek A H, Willeberg, P 1987 Veterinary Epidemiology, principles and methods. Iowa State University Press, Ames, Iowa, USA: 121 [ Links ]
17. Mhaiki J J K 1979 Studies on some genetic and environment effects of traits of economic importance in dairy cattle. MSc thesis, University of Dar-es-Salaam, Tanzania. [ Links ]
18. Ministry of Water and Livestock Development 2005 Livestock Sector Development in Tanzania. Online at: www.mifugo.go.tz/sectorBrief.htm (accessed April 2010) [ Links ]
19. MOAC/SUA/ILRI 1998 The Tanzania dairy sub-sector. A rapid rural appraisal, vol. 3, Main report. Dar-es-Salaam, Tanzania: 6-20 [ Links ]
20. Ogden N H, Swai E S, Beauchamp G, Karimuribo E, Fitzpatrick J L, Bryant M J, Kambarage D, French N P 2005 Risk factors for tick attachment to smallholder dairy cattle in Tanzania. Preventive Veterinary Medicine 67: 157-170 [ Links ]
21. Payne W J A, Wilson R T 1999 An introduction to animal husbandry in the tropics. Blackwell Science, Malden, MA, USA, Paris, London: 12-45 [ Links ]
22. Shoo M K, Semvua R H, Kazwala R R, Msolla P 1992 A study on the causes of specific mortality rates of dairy calves on farms in the eastern zone of Tanzania. Preventive Veterinary Medicine 13: 59-63 [ Links ]
23. Speicer J A, Hepp R E 1973 Factors associated with calf mortality in Michigan Dairy herds. Journal of American Veterinary and Medical Association 162: 463-466 [ Links ]
24. Swai E S 2002. Epidemiological studies of tick borne diseases in small scale dairy farming system in Tanzania. PhD thesis, University of Reading, Reading, UK [ Links ]
25. Swai E S, Bryant M J, Karimuribo E D, French N P, Ogden N H, Fitzpatrick J L, Kambarage D M 2005 A cross-sectional study of reproductive performance of smallholder dairy cows in coastal Tanzania. Tropical Animal Health and Production 37: 513-525 [ Links ]
26. Swai E S, French N P, Beauchamp G, Fitzpatrick J L, Bryant M J, Kambarage D, Ogden N H 2005 A longitudinal study of sero-conversion to tick-borne pathogens in smallholder dairy youngstock in Tanzania. Veterinary Parasitology 131: 129-137 [ Links ]
27. Swai E S, Karimuribo E D, Moshy W, Mbise A N 2007 A comparison of seroprevalence and risk factors for Theileria parva and T. mutans in smallholder dairy cattle in the Tanga and Iringa regions of Tanzania. Veterinary Journal 174: 390-396 [ Links ]
28. Therneau T M, Grambsch P M 2001 Modeling survival data: extending the Cox model. Springer, New York: 23-45 [ Links ]
29. Urassa J K, Raphael E 2002 The contribution of small scale dairy farming to community welfare: a case study of Morogoro Municipality. Online at: http://www.fiuc.org/esap/MOROG/MOROG5/General/dairyfarming.pdf (accessed 2 March 2010) [ Links ]
Received: August 2010.
Accepted: November 2010.
* Author for correspondence. E-mail: esswai@gmail.com














