Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
SA Orthopaedic Journal
On-line version ISSN 2309-8309
Print version ISSN 1681-150X
SA orthop. j. vol.9 n.4 Centurion Jan. 2010
CLINICAL ARTICLE
A deadly bed partner: m'Fesi (Mozambique spitting cobra)
SS VermaakI; A VisserII; TLB le RouxIII
IMBChB(Pret), MSc(Sports Medicine)(Pret), Senior Registrar, Department Orthopaedic Surgery, University of Pretoria
IIMBChB(Pret), PG(Dip)TM(UFS), DTM&H(Wits), Senior Registrar, Department Clinical Pathology, University of Pretoria National Health Laboratory Services
IIIMBChB(Pret), FCS(Ortho)(SA), MMed(Ortho)(Pret), National Chief of Orthopaedics Military Hospitals Professor of Orthopaedic Surgery, University of Pretoria
ABSTRACT
Venomous snake bites are estimated to occur in 2.5 million people annually worldwide, with 125 000 resulting in death. The incidence of snake bites in Africa has been estimated at 100-400 bites per 100 000 people. Currently literature is largely restricted to case reports. In this case series, we describe three patients who sustained Mozambique spitting cobra bites on their hands, with their subsequent management.
As was reported in the previous publications, all of the patients sustained snake bites while at home sleeping and two of the patients sustained multiple bites. All patients were bitten on their hands and developed significant soft tissue necrosis requiring surgical intervention. As reported in international publications, an initial delay in debridement led to improved outcome. Initial soft tissue necrosis has been shown to correlate poorly with ultimate wound demarcation. In accordance with publications, compartment syndrome did not occur in any of the patients, and swelling could be easily managed with simple elevation.
In conclusion, m'Fesi bites are medical emergencies, but they warrant delayed wound debridement. Orthopaedic surgeons are often consulted for management of limb injuries; therefore, a thorough knowledge regarding management of cytotoxic snake bites should be a part of the general orthopaedic surgeon's training.
Introduction
Venomous snake bites are estimated to occur in 2.5 million people annually worldwide, with 125 000 resulting in death.1 The incidence of snake bites in Africa has been estimated at between 100-400 bites per 100 000 people.2 Currently literature is restricted to predominantly case reports, which has been suggested by some authors to be useful in contributing to the establishment of formal protocols on management.2In this case series, we describe three patients who sustained Mozambique spitting cobra bites on their hands, with their subsequent management.
Patient 1
A 13-year-old male patient sustained multiple snake bites on both hands and neck while sleeping. The patient was taken to the local emergency department within 45 minutes of the injury, where he received supportive therapy. The patient also showed signs of respiratory distress.
For this reason, a total of 8 ampoules of polyvalent antiserum were administered intravenously. These symptoms improved within 5 hours, and intubation was never indicated.
The patient was subsequently transferred to a tertiary hospital where he was admitted to the intensive care unit (ICU) for observation by a general surgeon. While in ICU the patient was never intubated, and did not developed features suggestive of compartment syndrome. On day 6, he was referred to an orthopaedic surgeon for management of necrotic hand tissue. On evaluation of the wounds intra-operatively, the right index finger was necrotic and septic to the web space between the second and third fingers. A ray amputation into normal tissue was performed and closed over a drain. The adjacent middle finger had a volar area of necrosis and sepsis over the middle phalanx, which extended onto the flexor tendon sheath, with necrosis of the overlying pulleys, while the neurovascular structures remained intact. A debridement and cross-finger flap was performed from the adjacent fourth finger's dorsal side. The donor site was covered with a split thickness skin graft. The left hand's pinkie, which was necrotic and septic up to the metacarpal phalangeal joint (MPJ), was also addressed at the same sitting. A standard ray amputation was performed. Tissue specimens taken in theatre were sent for microbiological analysis, rendering no growth. Following discharge, the patient recovered full hand function (Figure 1).

Patient 2
A 41-year-old male sustained multiple snake bites on his left hand while asleep. The patient reached the local emergency department within 15 minutes, complaining of respiratory distress. For this reason, 5 ampoules of polyvalent antiserum were administered intravenously, which alleviated the respiratory distress but not the pain symptoms.
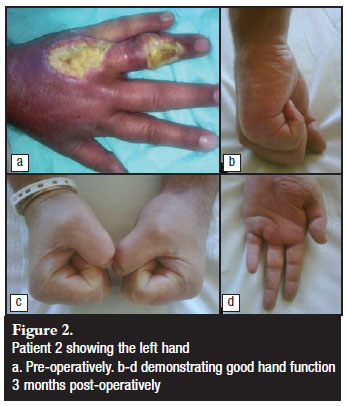
Following two days of observation, he was referred to a specialist unit for possible compartment syndrome of the forearm (although patient was bitten on the hand only). Although markedly swollen, compartment syndrome of the forearm was clinically excluded by showing absence of paraesthesia, pain on passive stretch and disproportionate pain. Compartmental pressures of the forearm were not measured. The patient sustained two bites to his left hand, one to the dorsum of the ring finger, and one the dorsum of the hand. Surgery was delayed until full demarcation, which occurred 10 days after admission (12 days post injury). The ring finger was still viable but was necrotic and septic over the dorsal aspect, with partial destruction of the extensor tendons. This area showed a 5 by 5 cm undermined wound, with extensor tendon loss to the ring finger, as well as loss of the extensor digiti minimi quinti and partial sloughing of the third finger's extensor tendon (<50%). The wounds were dressed with Intrasite gel and the patient was empirically started on amoxicillin-clavulanate. Tissue samples were submitted for microbiological analysis, showing Morganella morganii and Proteus vulgaris, both sensitive to ciprofloxacin and trimethoprim-sulphamethoxazole. Considering the fact that the patient was formally debrided and showed improvement prior to receiving the culture results, the initial antibiotic course was completed. The patient opted for a ray amputation. He currently has good hand function and is participating in a rehabilitation programme (Figure 2).
Patient 3
A 44-year-old female sustained a single snake bite on her right hand while asleep. She was rushed to the local emergency department where she developed respiratory distress. Polyvalent antiserum was administered intravenously. Following 48 hours of observation in ICU, she was discharged. The wound became septic, extending into the interphalangeal (IP) joint space. Despite this, there was a delay in referral and she only reached tertiary care 26 days after the initial insult. A formal debridement was performed on admission. The thumb was deemed viable, with loss of soft tissue cover over the interphalangeal joint. Microbiological cultures revealed no growth, probably because the patient had received antibiotic therapy prior to sampling.
She received a second wound debridement a few days later after hand baths in the ward. Microbiological specimens cultured a generally sensitive Pseudomonas fluorenscens, which was treated with ciprofloxacin and rifampicin. A local pedicle island flap was taken from the ulnar aspect of the middle finger to cover the defect. The IP joint was also formally arthrodesed with a headless compression screw. The donor area was covered with a full thickness skin graft. On follow up examination, there were no complications, and she was referred for rehabilitation (Figure 3).

Discussion
The snake
The Mozambique spitting cobra (m'Fesi) scientifically known as Naja mossambica is part of the elapid family of snakes (Figure 4).3-6 This snake can reach a length of 1.5 m when fully grown6 and is found over a large triangular area from Tanzania in the north, west to southern Angola and northern Namibia, and south into KwaZulu-Natal;4 it is most commonly found in low-lying areas, and commonly around permanent water.6

It is described as a slate grey to olive brown snake with black edged scales. The under surface of the snake is a salmon-pink or yellowish colour, with irregular black bars across the throat.5 They are considered nocturnal, but juveniles can be active during the day.5 They mainly consume rodents, and are therefore often found in urban areas.6 The highest incidence of bites occur during the spring and summer months.7-8 A study performed by Tilbury highlighted the fact that 94% of the snake bites occurred inside human dwellings, and 81% of these while the victim was asleep.8 Similar observations were made by other authors.9 Blaylock has shown that multiple bites are more likely in sleeping patients.10 In the current study, all patients were bitten while sleeping at home and two sustained multiple bites.
This snake also spits very accurately, as a defensive behaviour,4-5 up to a distance of 2 metres,11 aiming for the eyes of the victim. This results in severe eye pain on contact; eyed should be rinsed well to prevent the rare complication of permanent blindness.3,9
The Mozambique spitting cobra's venom has a predominantly cytotoxic effect, but also has additional neurotoxic effects.4,6,8 The enzyme responsible for most of the cytotoxic damage is phospholipase A2 (PLA2).12 It binds to fatty acids and is allosterically activated by long chain fatty acids13 causing electron transfer inhibition, dissolving mitochondrial bound enzymes, hydrolysis of phospholipids in nerve axons, breakdown of acetylcholine vesicles at neuromuscular junctions, myonecrosis, and inducing lysis of red cell membranes.1 m'Fesi's neurotoxic component is attributed to an alpha-neurotoxin that binds with high affinity to nicotinic acetylcholine receptors.14 It has a very large venom reserve, and can repeatedly expel large quantities of venom.4-5
Management
Clinical presentation
Patients sustaining snake bites are often anxious, contributing to the development of symptoms. The cytotoxic component of the venom causes immediate severe local pain, with rapidly spreading swelling.9 Blistering at the bite site with superficial necrosis can present within a few hours of the bite, but it does not reflect the final demarcation, as was demonstrated in this case series. The demarcation line will only be evident several days after the insult8 with occasional skip lesions in the skin. Although rare in adults, massive swelling and oedema may lead to hypovolaemic shock.8-9 The neurological symptoms usually ascribed to the elapid group are restlessness, vomiting, dizziness, increased salivation, sweating, difficulty in swallowing and speech, ptosis (one of the first signs), impaired vision and eye movements, and respiratory distress,4,11 Convulsions have also been described in the Naja species.11 Neurotoxic effects are completely reversible and usually minimal.9
First aid
Early patient resuscitation is an important part of management, thus immediate transport to an emergency department is essential. This process may be facilitated by keeping the patient calm,3,9 and immobilising the affected limb through splinting, to alleviate pain and reduce venom spread.2-3,9 Excision of the wound, incision and suction of the wound, the use of a tourniquet, application of ice packs and the application of local chemicals to the wounds have not shown any benefit, and may actually be harmful.2-3,9,15 The patient should be discouraged from eating, as it may worsen vomiting,2 and may delay emergency surgery, if indicated.9 Fluid intake may be necessary if a delay in transport is expected. Whenever possible the snake should be positively identified.2,4,9 All rings and tight-fitting jewellery should be removed immediately from the affected limb.
Emergency room management
General measures include basic resuscitation as neurotoxic effects may cause respiratory compromise. Although snake bites do not carry a high risk of tetanus exposure, patients should ideally receive toxoid prophylaxis.1,4 Base-line laboratory examination should include a full blood count, renal function, random glucose, liver functions, myoglobin, coagulation profile and an arterial blood gas.9
Specific treatment options include anti-venom16 (antisnakebite serum), analgesia and antimicrobials. No specific monovalent anti-venom exists for m'Fesi, but a polyvalent formulation is produced and distributed by the South African Institute for Medical Research (SAIMR).17 Child and adult dosing is similar.9,18 In total 100 ml of the antivenom should be diluted in 500 ml of Ringer's solution or saline and given at an initial rate of 5 ml/hr, with an increase in rate if no reaction occurs.2 It should not be administered at or around the bite site.1
The anti-venom should be crystal clear and without sediment before use.19 Indications for use are predominantly to combat neurotoxic effects. It may also improve cytotoxic effects, provided adequate amounts are administered timeously (<3 hrs after bite),8-9 but this remains controversial.20 Despite early administration of anti-venom, two of the three patients in this case series required ray amputations due to tissue necrosis, but neither of them received the full dose. Reactions associated with anti-venom administration can occur in 40% to 76%18 of cases and are classified as either anaphylactoid (no previous exposure to anti-venom) or anaphylactic (where after exposure, reaction is caused by the interaction of immunoglobulins with horse proteins in anti-venom). These are typically life-threatening, and should be managed accordingly.3-4 Late reactions (serum sickness) can occur as late as 12 days post administration.3-4 Although widely used, the value of giving steroids, antihistamine or small doses of adrenalin prior to anti-venom to prevent the above mentioned reactions has not been proven.4 They may be considered if hypersensitivity is suspected.2,4 These drugs are better suited to treat reactions once they occur.18 Sensitivity testing of the anti-venom (i.e. skin or conjunctival test with dilute anti-venom) is also not recommended due to the poor negative predictive value (negative tests do not exclude anaphylaxis),2,10 and may delay initiation of treatment.2 Ultimately, the benefit should outweigh the risk of administration.18
Adequate analgesia is essential during the acute management of the patient. Although morphine is a good emergency analgesic drug, it should be used with caution in the acute period after the bite, due to its respiratory depressant properties.9
In general prophylactic antibiotics are not recommended for most snake bites, especially if good wound care is followed.1,9,21 Snake mouths have been shown to have low bacterial numbers, and venom of the Naja spp has been shown to have antibacterial properties to both aerobic and anaerobic bacteria.10,22 Tetanus and gas gangrene have not been described in Southern African snake bites to date.10 Empiric antibiotics, typically ampicillin-clavulanate, can be considered if there is a delay in wound care, or if wounds were inflicted as part of the initial management.
Wound care and surgical intervention
Daily cleaning with soap and water, followed by sterile dressings is essential.1 In the cases described Betadine hand baths were used, with Intrasite-Jelonet dressings pre-operatively. Surgery may be indicated in the acute setting if compartment syndrome23 is suspected. Compartment syndrome can be confirmed using intracompartment pressure measurements, as described by Whiteside.24-25 These techniques have also been applied with some success in cases of suspected upper limb compartment syndrome.26-27 Although more common in cytotoxic snake bites, it remains a rare complication.8
Surgical exploration of the bite wound is not recommended.1 Progression to full demarcation does not occur soon after the insult. Therefore, early debridement has been discouraged by various authorities, and is typically advocated after day 6.8 This issue differs from fasciotomies, which are indicated in cases of typical compartment syndrome. Surgery in the first three days following the snake bite may be significantly complicated due to the underlying coagulopathy. This contributes to the current reasoning to delay early wound debridement.1
Conclusion
In conclusion, m'Fesi bites are medical emergencies, but they warrant delayed wound debridement. Orthopaedic surgeons are often consulted for management of limb injuries; therefore a thorough knowledge regarding management of cytotoxic snake bites should be a part of the general orthopaedic surgeon's training.
References
1. Otten E. Venomous animal injuries. In: Marx J, Hockberger R, Walls R, editors. Rosen's Emergency Medicine. Philedelphia: Mosby Elsevier; 2006. p. 894-903. [ Links ]
2. Mebs D. Venomous and poisonous animals. 1st ed. Stuttgart: Medpharm Scientific Publishers; 2002. [ Links ]
3. Backshall S. Venomous animals of the world. 1st ed. Baltimore: The Johns Hopkins University Press; 2007. [ Links ]
4. Broadley D. Fitzsimons' Snakes of Southern Africa. 1st ed. Johannesburg: Delta Books; 1962. [ Links ]
5. Marais J. A complete guide to the snakes of Southern Africa. 1st ed: Creda Press; 1992. [ Links ]
6. Stuart C, Stuart M. Dangerous creatures of Africa. 1st ed. Cape Town: Struik Nature; 2009. [ Links ]
7. Leisewitz A, Blaylock R, Kettner F, Goodhead A, Schoeman J. The diagnosis and management of snakebite in dogs - a southern African perspective. S Afr Vet Ass. 2004;75(1):7-13. [ Links ]
8. Tilbury C. Oberservations on the bite of the Mozambique spitting cobra (Naja mossambica mossambica). SAfrmedj. 1982;61:308-13. [ Links ]
9. Meier J, White J. Handbook of clinical toxicology of animal venoms and poisons. 1st ed. New York: CRC Press; 1995. [ Links ]
10. Blaylock R. Epidemiology of snakebite in Eshowe, KwaZulu-Natal, South Africa. Toxicon. 2004;43:159-66. [ Links ]
11. Russel F. Snake venom poisoning. 1st ed. New York: Scholium International; 1983. [ Links ]
12. Wacklin H. Interfacial mechanism of phospholipase A2: pH-dependent inhibition and ME-B-cyclodextrin activation. Biochemistry. 2009;48:5874-81. [ Links ]
13. Ahmed T, Kelly S, Price N, Lawrence A. Activation of the acidic isoform of phospholipase A2 from Naja mossambica mossambica venom by oleoyl imidazolide requires the cooperative action of two ionizing groups. IUBMB Life. 1999;48:287-91. [ Links ]
14. Ackermann E, Taylor P. Nonidentity of the a-neurotoxin binding sites on the nicotinic acetylcholine receptor revealed by modification in a-neurotoxin and receptor structures. Biochemistry. 1997;1997(36):12836-44. [ Links ]
15. Cawrse N, Inglefield C, Hayes C, Palmer J. A snake in the clinical grass: late compartment syndrome in a child bitten by an adder. British Journal of Plastic surgery. 2002;55:434-5. [ Links ]
16. Hadley G, Mars M. Snakebites in children in Africa: a practical approach to management. http://www.ptolemy.ca/members/archives/2006/Snake_Bites.htm. 2006 [cited 2010 1 June]; Available from: [ Links ]
17. //home.intercom.com/pharm/SAIMR/snake.html. [cited 10 April 2010]; Available from: [ Links ]
18. Moran N, William J, Newman R, Theakston D, Warrell D, Wilkinson D. High incidence of early anaphylactoid reaction to SAIMR polyvalent snake antivenom. Transactions of the Royal Society of Tropical Medicine and Hygiene. 1998;92:69-70. [ Links ]
19. Muguti G. Management of snake bites. Cent Afr J Med. 2003;49:142-4. [ Links ]
20. Fulton J, Hoffman R. Fasciotomy after envenomation: Measure twice and cut once. Annals of Emergency Medicine. 2005;45(3):338-9. [ Links ]
21. Campbell B, Corsi J, Boneti C, Jackson R, Smith S, Kokoska E. Pediatric snakebites: lessons learned from 114 cases. Journal of Pediatric Surgery. 2008;43:1338-41. [ Links ]
22. Blaylock R. Antibacterial properties of KwaZulu-Natal snake venom. Toxicon. 2000;38:1529-34. [ Links ]
23. Mars M, Hadley G. Raised intracompartmental pressure and compartment syndromes. Injury. 1998;29(6):403-11. [ Links ]
24. Whiteside T, Heckman M. Acute compartment syndrome: Update on diagnosis and treatment. J Am Aca Orth Surg. 1996;4(4):209-18. [ Links ]
25. Whiteside T, Morimoto H. Compartment syndrome and the role of fasciotomy, its parameters and techniques. Instr Course Lect. 1977;26:179-96. [ Links ]
26. Antebi A, Hescovici D. Acute compartment syndrome of the upper arm: a report of cases. Am J Orthop. 2005;34(10):498-500. [ Links ]
27. Drouet A, Jaquin O, Guilloton L, Dumas P, Volckmann P, Ribot C. Anterior compartment syndrome of the forearm caused by exercise: unusual cause of recurrent episodes of acute effort rhabdomyolysis. Rev Med Interne. 2001;22(4):394-7. [ Links ]
 Reprint requests:
Reprint requests:
Dr Slade Solomon Vermaak
Cell: +2783 412 7071
Email: slvermaak@telkomsa.co.za
The authors state that no benefit has been derived from any commercial party related directly or indirectly to the subject of this article. Dr Visser's work is supported by the Discovery Foundation.














