Services on Demand
Article
Indicators
Related links
-
 Cited by Google
Cited by Google -
 Similars in Google
Similars in Google
Share
SA Orthopaedic Journal
On-line version ISSN 2309-8309
Print version ISSN 1681-150X
SA orthop. j. vol.13 n.2 Centurion Apr./Aug. 2014
SPINE
The differential diagnosis of neurogenic and referred leg pain
JA Louw
MBChB(Pret), MMed(Orth), PhD, MD; Full-time private practice, Pretoria, South Africa
ABSTRACT
BACKGROUND: Neurogenic and referred leg pain are some of the most challenging clinical problems in spinal surgery due to the diversity of conditions that can act as aetiological factors.
METHODS: The literature was reviewed and combined with case studies to demonstrate different aetiological factors.
RESULTS: At least 300 conditions were identified that might cause neurogenic and referred leg pain. An aetiological classification of neurogenic and referred leg pain is presented. The classification includes systemic conditions, conditions from the brain, spinal cord, cervical and thoracic spinal canal, lumbar spinal canal, lumbar nerve root canal, lumbar extraforaminal area, the pelvis and the lower extremity. Each one of these conditions can mimic a lumbar disc herniation accurately.
CONCLUSION: The aetiological classification can be used as a checklist when evaluating neurogenic and referred leg pain. Each condition deserves careful consideration and when overlooked might result in a missed diagnosis.
Key words: neurogenic, leg pain, nerve root
Introduction
Leg pain is a common presenting symptom of lumbar disc herniation due to neurological compression. If conservative management fails, a large number of these patients might undergo lumbar spine surgery. Magnetic resonance imaging (MRI) is considered to be the cornerstone special investigation to confirm the diagnosis of a lumbar disc herniation. However, between 38% and 52% of asymptomatic individuals demonstrated significant lumbar disc bulging on MRI.1,2 Given the high prevalence of these findings and of back pain, the discovery by MRI of disc bulges may frequently be coincidental and the leg pain caused by an unrelated condition. It is therefore essential to consider all possible aetiological factors when evaluating leg pain.
Literature reviews combined with the author's personal experience in the examination of more than 15 000 patients with back pain were used to propose an aetiological classification of conditions to consider when evaluating leg pain.
Conditions to consider when evaluating leg pain
1. Systemic conditions
Metabolic neuropathy
Diabetes mellitus is the most common metabolic neuropathy. Its most common presentation is the distal symmetrical polyneuropathy with bilateral symmetrical pain in the lower extremities starting distally and moving proximally.3,4 Other subtypes include proximal diabetes, truncal, cranial, median and ulnar neuropathies. Diabetic autonomic neuropathy affects each tissue, organ and system in the whole body and is strongly involved in the development of foot ulceration.5
A less common diabetes presentation is diabetic amyotrophy6 which has most probably a vasculitis aetiology with ischaemia followed by axonal degeneration and demyelination. The main features are unilateral weakness, wasting and pain most commonly in the quadriceps. It spreads later to the contralateral side in an asymmetrical manner.
Other metabolic neuropathies to consider are alcoholic and uraemic neuropathies.7,8
Vasculitic neuropathy
The primary pathology of vasculitic neuropathies is a vasculitis of the small and medium-sized vessels in the peripheral nervous system9 with an area of infarction in the nerve.10,11 The presenting symptoms are severe pain localised to the region of the infarction, motor deficit, numbness and paraesthesias. Vasculitic neuropathies are classified into primary and secondary vasculitides.10 Primary vasculitic neuropathy includes Churg-Strauss syndrome, microscopic polyangiitis, classic polyarteritis nodosa and Wegener granulomatosis. Secondary vasculitis occurs as a complication of connective tissue disease (systemic lupus erythematosus, rheumatoid arthritis and Sjogren syndrome), infection (hepatitis B and C, human immunodeficiency virus, Lyme disease, cytomegalovirus, Herpes zoster virus and various bacterial infections), medication (sulphonamides, other antibiotics and anti-viral agents) and malignancies representing a paraneoplastic vasculitis.11-15
Paraneoplastic peripheral neuropathy
In addition to the paraneoplastic vasculitis and the local effect of the tumour, malignancies can also cause a parane-oplastic sensorimotor neuropathy which might even be more debilitating than the cancer itself.16 The detection of anti-neuronal antibodies and EMG changes help to identify the neuropathy as paraneoplastic. A paraneo-plastic myopathy might also develop which can cause leg pain.16,17
Case 1
This 53-year-old female patient presented with severe acute onset unilateral pain in the L5 nerve root distribution. The MRI of the lumbar spine failed to demonstrate any neurological compression. The coronal images of the MRI thoracic spine demonstrated a lesion in the lung which was biopsied and turned out to be a bronchus carcinoma (Figure 1). The antineuronal antibodies and EMG confirmed a paraneoplastic neuropathy.

Case 2
This 64-year-old male patient presented with severe unilateral pain in the L5 and S1 nerve root distribution 25 months after an uncomplicated L4 to S1 decompression and fusion procedure. The plain radiographs demonstrated an uncomplicated fusion and the MRI a wide decompression without neurological compression. The anti-neuronal antibodies demonstrated a positive anti-PNMA(Ma/Ta) which is associated with small cell bronchus carcinoma. This prompted a chest CT scan which demonstrated a small lesion in the apex of the right lung. A lung biopsy demonstrated a bronchus carcinoma.
Vascular occlusion
Vascular occlusion can mimic neurogenic claudication. The peripheral pulses should therefore be palpated in every back pain examination.
2. Brain
Brain-related causes of leg pain include multiple sclerosis,18,19 Parkinson's disease,20 motor neurone disease,21 post-stroke pain in lenticulo-capsular haemorrhages22 and space-occupying lesions.
Case 3
This 57-year-old male patient presented with spontaneous onset pain in the right lower extremity 12 years after an L4 to S1 fusion, neurological decompression and instrumentation. The plain radiographs failed to demonstrate any complications and no neurological compression could be demonstrated on the MRI lumbar spine. The history of the systemic diseases revealed resection of a melanoma behind the right scapula three years before. A CT scan of the brain demonstrated multiple melanoma metastases (Figure 2).

3. Spinal cord
Spinal cord conditions which should be excluded in the evaluation of leg pain are multiple sclerosis,18,19 motor neurone disease,21 transverse myelitis,22,23 syringomyelia,24,25 and any posterior (compression of substantia gelatinosa at the tip of the posterior column) or anterolateral (compression of lateral spino-thalamic tract)26 situated space-occupying lesion, for example intervertebral disc herniation,27,28 tumour, haematoma or abscess. An atypical Brown-Sequard lesion can also be produced, with loss of pain and thermal sensation on the contralateral side and leg pain on the ipsilateral side.27,29
Case 4
This 73-year-old female patient presented with excruciating pain in the L5 distribution of the right lower extremity and was booked for an emergency laminectomy.
An MRI thoracic spine demonstrated a posterior situated meningioma at T7
When the patient presented for a second opinion, the MRI of the lumbar spine demonstrated a small disc bulging on the asymptomatic side. An MRI thoracic spine was requested which demonstrated a posterior situated meningioma at T7 (Figure 3) which was removed and the L5 nerve root symptoms resolved completely.

4. Cervical and thoracic spinal canal
Any posterior or anterolateral situated space-occupying lesion in the cervical and thoracic spinal canal may cause compression on the lateral spino-thalamic tract and substantia gelatinosa at the tip of the posterior column of the spinal cord with resulting leg pain, for example haematoma, abscess, soft tissue or bone tumours, etc.26-29
5. Conus medullaris
At the conus medullaris the nerve roots are positioned lateral to the spinal cord (Figure 4). A parasagittal disc herniation or any other space-occupying lesion (for example intradural or extradural tumours, haematoma, abscess, primary bone tumours, metastases, etc.) can therefore cause compression of the nerve roots with leg pain without much compression on the spinal cord.

Case 5
This 58-year-old male patient presented with severe pain in the right lower extremity in the L4 nerve distribution seven years after a T12 to S1 fusion and pedicle instrumentation. The CT myelogram demonstrated intervertebral disc and gas sequestration in the right parasagittal position with severe compression on the nerve roots but only slight displacement of the conus medullaris (Figure 5). Removal of the gas and disc sequestration alleviated the L4 symptoms completely.

6. Lumbar spinal canal
As far back as 1933, Baastrup30 reported on the interspinous bursa (kissing spines, Baastrup's disease) as a cause of leg pain31,32(Figure 6). The bursa can communicate with an epidural cyst with severe neurological compression.33

In facet joint syndrome, osteoarthrosis of the facet joint with a normal spinal canal and nerve root canal can give buttock and leg pain34,35(Figure 7). Hypertrophy of the facet joints gives spinal stenosis and leg pain (Figure 8).

A facet joint cyst can compress the nerve root with leg pain. With both superior or inferior rotational instability, the facet joints may rotate into the spinal canal with neurological compression and leg pain (Figure 9).

In degenerative spondylolisthesis, the vertebral body and inferior facet joints displace anteriorly. The inferior facet joints move forwards and compress the dural sac and nerve roots against the posterior aspect of the cranial vertebral body. If the flexion radiographs demonstrate superimposed instability with further anterior translation of the inferior facet joints in the flexed position, the compression on the dural sac is aggravated significantly. This dynamic compression on the dural sac is not demonstrated on the MRI and can only be appreciated on the stress radiographs (Figures 10 and 11).


Any space-occupying lesion in the lumbar spinal canal can give leg pain, for example intervertebral disc herniation or sequestration, soft tissue or bone tumours, haematoma, etc.
7. Lumbar nerve root canal
Inside the nerve root canal the facet joint might give leg pain with a combination of loss of disc height and posterior subluxation when the superior articular process moves upwards and forwards and might impinge the nerve root against the pedicle or the posterior aspect of the vertebral body, especially the inferior endplate (Figure 12).

In spondylosis osteophytes from the superior facet joint or posterior inferior vertebral endplate (Figure 13) may compress the nerve root.

Anterior subluxation of the vertebra in degenerative spondylolisthesis might be associated with hypertrophy of the superior facet joint with extension of the superior facet joint into the nerve root canal with compression of the nerve root and resulting leg pain.
Any nerve root lesion, for example a nerve root cyst or nerve root tumour, should be considered. The most common benign nerve root tumours are the schwannomas, neurofibromas36 and less common haemangioblastomas37,38 and non-Hodgkin lymphomas.39 Nerve sheath tumours can originate from the intradural or extradural position.40
Malignant peripheral nerve sheath tumours (MPNSTs) are grouped together by the World Health Organisation as MPNST and include previous terminology such as malignant neurilemmomas, neurogenic sarcoma and neurofibrosarcoma.41
In isthmic spondylolisthesis the nerve root canal is narrowed by the combination of disc bulging and pars interarticularis hypertrophy.
The pedicle moves down and might compress the nerve root in complete loss of disc height in combination with spondylolisthesis (Figure 14) and degenerative scoliosis.

Intervertebral disc herniation into the nerve root canal is the most common cause of nerve root canal narrowing and neurological compression.
A nerve root anomaly might easily be mistaken for a disc fragment with grave consequences. The most common anomaly is the conjoint nerve root with two nerve roots derived from a common dural sheath followed by two nerve roots in one foramina.42-44
A facet joint cyst can compress the nerve root with leg pain
8. Lumbar extraforaminal area
A far lateral disc herniation may compress the nerve root after it has left the nerve root canal (Figure 15).

The corporotransverse ligament extends from the vertebral body to the transverse process of the same vertebra.45 It may entrap the nerve root in rotatory subluxation in combination with complete disc space narrowing.
Psoas pathology, for example abscess, haematoma, tumour or any other space-occupying lesion, may compress the nerve root in its course through the psoas.46
In spondylolisthesis with severe disc space narrowing, the L5 nerve root may be impinged between the L5 transverse process and the ala of the sacrum (Figure 14). This may also occur in degenerative scoliosis with tilting of L5 and depression of the concave L5 transverse process (Figure 16).

9. Pelvis
At the entrance to the pelvis, a large osteophyte from the L5 transitional vertebra pseudoarthrosis might compress the L5 nerve root (Figure 17).

The lumbosacral ligament47 extends from the L5 vertebra to the ala of the sacrum and forms the roof of the lumbosacral tunnel through which the L5 nerve root passes. Ossification of the lumbosacral ligament in combination with osteophytes from the inferior L5 vertebral body endplate may impinge the L5 nerve root.47
A stress fracture of the sacrum can give back, buttock, groin and thigh pain.48 It may also give direct compression or a neuropathy of the nerve root.49 The most common associated stress fracture is that of the ipsilateral, contralateral or both pubic rami.50 It may remain undetected on plain radiographs for months but readily demonstrated on the STIR and T2 weighted MRI images (Figure 18).51 MRI alone identifies 99, 2% of stress fractures compared to 69% with the CT scan alone.51 The MRI remains the investigation of choice in these cases.

Degenerative sacroiliitis can give buttock, groin and thigh pain.52 An osteophyte from the sacroiliac joint extending anteriorly can compress a nerve root. Any type of infectious sacroiliitis (tuberculosis, brucellosis and piogenic)53 and tumours of bone and the soft tissue in and around the sacroiliac joint can give pain in a similar distribution.54 The sero-negative spondyloarthropathies (ankylosing spondylitis, psoriatic arthritis, reactive arthritis and Behcet's disease) should also be considered.55
A lumbosacral radiculoplexus neuropathy presents with asymmetrical lower limb pain, weakness, atrophy and paraesthesia.56 It can be caused by diabetic lumbosacral radiculoplexus neuropathy, non-diabetic lumbosacral radiculoplexus neuropathy, chronic inflammatory demyelinating polyneuropathy, connective tissue disease, Lyme disease, sarcoidosis, HIV and cytomegalovirus-related polyradiculopathy.56,57 The typical MRI neurography findings (Figure 19) are increased signal intensity and enlargement of the nerve and blurring of the perifas-cicular fat on the T2 weighted neurography sequences58-55 (long echo times, radiofrequency saturation pulses tosuppress signals from adjacent vessels and frequency-selective fat suppression).60,61 EMG, fasting blood glucose levels, sedimentation rate, rheumatoid factor, antinuclear antibodies, antineutrophil cytoplasmic antibodies, extractable nuclear antigen, HIV,56 serum antibodies associated with neurological disorders (antineuronal, antiganglioside and anti-astrocyte antibodies)62 and CSF evaluation are some of the special investigations of diagnostic value.

In the piriformis muscle syndrome hypertrophy, spasm, contracture or inflammation of the piriformis muscle may compress the ischiadic nerve with pain in any part of the ischiadic nerve distribution (Figure 20).

The diagnostic accuracy of a wide variety of clinical tests (tenderness on deep palpation over the piriformis,63 Pace's sign,64 Freiberg's sign,65 FAIR test [flexion- adduction-internal rotation],66 persistent piriformis position in external rotation) remain controversial.67 The imaging diagnosis with an MRI of the pelvis is based on enlargement of the piriformis and MRI neurography.55 Enlargement of the piriformis alone has a specificity of 66% and a sensitivity of 64%. If in addition the MRI neurography demonstrates an increased signal intensity and enlargement of the ischiadic nerve (Figure 21), the specificity increases to 53% and the sensitivity to 64%.68

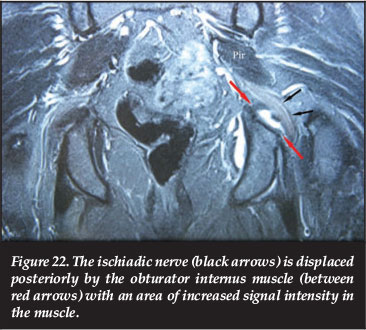
The ischiadic nerve passes underneath the piriformis and then over the obturator internus (Figure 20). Swelling and inflammation of the obturator internus might displace and irritate the ischiadic nerve with resultant leg pain (Figure 22).69
Superior gluteal nerve entrapment syndrome is caused by any condition which narrows the suprapiriformis foramen (Figure 20), for example enlargement or spasm of the piriformis muscle,70 superior gluteal nerve perforating the piriformis,71 aneurysm or pseudoaneurysm of the superior gluteal artery,72 direct injury to the superior gluteal nerve during hip surgery,73 percutaneous placement of iliosacral screws,74 abscess and pelvic fracture. The clinical diagnosis is based on the triad of buttock pain, weakness of hip abduction and marked tenderness on deep palpation in the region just lateral to the greater schiadic notch.75
Case 6
This 55-year-old male patient presented with the clinical triad of superior gluteal nerve entrapment. The MRI pelvis demonstrated sacroiliitis on the right side with an effusion and a neuropathy of the superior gluteal nerve with enlargement and an increased signal intensity (Figure 23).

The inferior gluteal nerve entrapment syndrome is caused by any condition which narrows the infrapiriformis foramen, for example an aneurysm of the inferior gluteal artery,63 pelvis fracture and abscess.76
In the pudendal nerve entrapment syndrome the pudendal nerve is compressed in the pudendal or Alcock's canal between the sacrospinous and sacrotuberous ligaments. The nerve might become ensheathed by ligamentous expansions that form a perineural compartment, by the falciform process of the sacrotuberous ligament or by duplication of the obturator fascia.77-79 It causes chronic perineal pain typically presenting as pain in the penis, scrotum, labia, perineum, anorectal area and medial proximal thighs.77
There are special considerations in female patients regarding leg pain which deserve mentioning. Sciatica in pregnancy can be caused by direct compression of the gravid uterus on the lumbosacral plexus and blood vessels.80,81 Incorrectly positioned leg stirrups on the obstetrical table can cause common peroneal nerve pain and weakness.80,82 Obstetric neuropathy of the sciatic nerve during vaginal labour can be caused by continuous pressure of the foetus on the lumbosacral plexus as it crosses the pelvic brim during prolonged labour.83 Piriformis syndrome can be caused during labour following a piriformis haematoma after prolonged labour, and a piriformis abscess after epidural anaesthesia and forceps delivery.84 Sciatica secondary to pathological conditions80 include leiomyomas with lumbosacral plexus compression,85 endometriosis with infiltration or compression of the sciatic nerve86 and a large retroverted uterus with compression on the lumbosacral plexus.80 Sciatica secondary to iatrogenic trauma includes vaginal procedures which might be complicated by ischiadic neuropathy,87 superior gluteal artery aneurysm with superior gluteal nerve compression and the superior gluteal nerve neuropathy due to stretching of the superior gluteal nerve.
Case 7
This 29-year-old female patient presented after two years of diligent conservative treatment for a small L5/S1 disc bulging with progressive deterioration of buttock and leg pain. The MRI pelvis demonstrated a large ovarian cyst which displaced a large uterus lateral and posterior with an underlying large piriformis muscle, and an increased signal intensity and enlargement of the ischiadic nerve (Figure 24).
Piriformis syndrome can be caused during prolonged labour, and a piriformis abscess after epidural anaesthesia and forceps delivery
10. Lower extremity
Greater trochanter bursitis and gluteus medius tendinitis remain some of the most common causes of leg pain in patients above 60 years of age.
A stress fracture of the femur neck, intertrochanteric area and the proximal femur presents with pain in the groin and anterior aspect of the proximal thigh. Plain radiographs may remain normal for a considerable time. An MRI and bone scan are useful in the early stages to confirm the diagnosis.
Iliopsoas88 and iliopectineal89 bursitis and synovitis present with pain in the groin and anterior aspect of the proximal thigh. They might also give femoral nerve palsy and pain.
Non-traumatic focal mononeuropathies may present with symptoms indistinguishable from a lumbar disc herniation and include the following conditions:
1. Compression neuropathies: Acute, chronic and hered-itary.90
2. Infectious neuropathies: Herpes zoster, Lyme disease, acquired immunodeficiency syndrome (AIDS), leprosy mononeuropathy, hepatitis B and C, cytomegalovirus, Epstein-Barr virus, various bacterial infections.11-15
3. Inflammatory/vasculitic neuropathies: Churg-Strauss syndrome, microscopic polyangiitis, classic polyarteritis nodosa, Wegener granulomatosis, systemic lupus erythematosus, rheumatoid arthritis, Sjogren syndrome.11-15
4. Drug-related neuropathies: Sulphonamides, other antibiotics, anti-viral agents.11-15
5. Metabolic neuropathies: Diabetic amyotrophy, alcoholic and uraemic.6-8
6. Paraneoplastic neuropathies: Paraneoplastic vasculitis, paraneoplastic sensorimotor neuropathy.16,17
7. Radiation neuropathy.90
8. Nerve and nerve sheath tumours.90
9. Hereditary: Familial amyloid polyneuropathy,91 hereditary compression neuropathy.90
The nerve entrapment syndromes of the lower extremity include the following conditions:
The iliohypogastric (T12, L1), ilioinguinal (T12, L1) and genitofemoral (L1, 2) entrapment syndromes usually occur after lower abdominal surgery.92
Entrapment of the lateral femoral cutaneous nerve (L2-4) is also known as meralgia paraesthetica. Symptoms include anterior and lateral thigh burning, tingling and numbness.92
The femoral nerve (L2-4) is usually entrapped below the inguinal ligament where it lies in close proximity to the femoral head, the tendon insertion of the vastus inter-medius, the psoas tendon, and the hip joint and hip joint capsule, all of which can cause entrapment.92 Other causes of entrapment include methylmethacrylate heat during total hip arthroplasty, pelvic procedures requiring acutely flexed, abducted and externally rotated positioning, compression by a foetus in difficult childbirth, pelvic fractures and radiation.92
The saphenous nerve (L3,4) is the terminal and longest branch of the femoral nerve. It can become entrapped as it pierces the roof of the adductor canal, by femoral vessels, pes anserine bursitis, varicose vein operations and medial knee surgery.92,93
Common peroneal neuropathy is the most common mononeuropathy of the lower extremity. It is most commonly entrapped as it courses along the lateral aspect of the fibula neck through a fibro-osseous tunnel by tight plaster casts, knee surgery, osteophytes, synovial cysts or ganglions and sitting in a prolonged crossed leg position.93-96 Repetitive exercises involving inversion and pronation (for example runners and cyclists) stretch the common peroneal nerve against the fibula neck and fibrous arch of the peroneal tunnel.93 Many cases are idiopathic in origin.97
The deep peroneal nerve travels in the anterior compartment of the leg between the extensor digitorum longus and the extensor hallucis longus tendon until it reaches the ankle where it courses under the extensor retinaculum to enter the anterior tarsal tunnel where most of the entrapment occurs, referred to as the anterior tarsal tunnel syndrome with symptoms referring to the dorsum of the foot.98,99 The entrapment is usually sport related.93
The superficial peroneal nerve courses through the antero-lateral compartment of the leg until it pierces the deep fascia of the lateral compartment approximately 10 to 15 cm above the ankle where it may become entrapped. This is commonly seen in dancers in whom the nerve may become stretched during inversion or plantar flexion injuries.99,100
Proximal tibial entrapment neuropathy is uncommon, owing to the deep location of the nerve and abundant surrounding muscle tissue. Entrapment is usually related to space-occupying lesions in the popliteal fossa such as tumours, popliteal cyst, popliteal artery aneurysm and ganglia.99
Distal tibial entrapment usually occurs in the tarsal tunnel causing tarsal tunnel syndrome. Entrapment follows trauma (fracture, surgery and scarring), space-occupying lesion (tumour, ganglia, varicosities and an anomalous muscle) and foot deformities.99,101
The sural nerve runs distally between the two heads of the gastrocnemius beneath the crural fascia until it pierces the fascia between the middle and distal thirds of the calf. Compression of the nerve by mass lesion, scar tissue, ganglion, surgical trauma and thrombophlebitis has been reported. Extrinsic compression can occur from tight ski boots or casts. The crural fascia may act as a compression or fixation point in athletes with stretching of the nerve.92,102-104
At the distal branches of the tibial nerve, medial plantar nerve neuropathy (Jogger's foot) can develop owing to repetitive trauma to the nerve while running with increased heel valgus and foot pronation.99,105
Entrapment of the inferior calcaneal nerve, the first branch of the lateral plantar nerve, often manifests as heel pain (Baxter neuropathy).95
Interdigital neuropathy (Morton neuroma) is caused by a fibrotic nodule of the interdigital nerve most commonly at the second and third intermetatarsal spaces. The patients present with web space pain, tingling, numbness and paraesthesias99,106
Medial plantar proper digital neuropathy (Joplin's neuroma) is an entrapment neuropathy of the plantar proper digital nerve which is particularly susceptible to repetitive trauma.99,107,108
The diagnosis of an entrapment neuropathy is based on a good clinical examination, typical EMG findings, MRI of the nerves and muscle, and blood investigations as mentioned under lumbosacral radiculoplexus neuropathy. The MRI includes signs of nerve entrapment, increased signal intensity, enlargement of the nerve, loss of the normal fascicular appearance or blurring of perifas-cicular fat.57 The indirect signs of nerve injury are seen in the muscles supplied by the nerve with an increased signal intensity followed by atrophy and fat placement of the muscle.59,99
Leg pain may also be caused by myositis or myopathy. Palpation of the muscle may reveal local tenderness which is more pronounced than what would be expected of neurological compression, swelling and oedema, atrophy or hypertrophy and overlying skin changes.
One of four basic patterns of abnormality may be present on the MRI, namely muscle oedema with an increased signal intensity, muscle atrophy with an increased fat content, mass within a muscle or an accessory muscle.105 The aetiology of muscle oedema is inflammatory myopathy (dermatomyositis, polymyositis and inclusion body myositis), polymyositis, myositis in collagen vascular disease (rheumatoid arthritis, systemic lupus erythematosus, mixed connective tissue disease and Sjogren syndrome), radiation myositis, Graves' disease, drug-induced myositis (lipid-lowering statins, antiretro-viral medication), HIV myositis, myositis due to infection (high risk patients include diabetes, immuno-compro-mised patients, penetrating wounds), polymyositis, necro-tising fasciitis and sarcoidosis. The denervation phases include a normal MRI in the acute phase, mixed oedema and paradoxical hypertrophy in the early subacute phase, mixed oedema and atrophy in the late subacute phase and atrophy in the chronic phase.109
Sport-related leg pain111 includes tibial and fibular stress fractures,111 medial tibial stress syndrome,112 chronic exertional compartment syndrome,113 tendinopathies,114,115 nerve entrapment syndromes,116 vascular syndromes117,118 and myopathies.115
Finally, leg pain may be associated with three poorly understood neurological conditions, namely complex regional pain syndrome,120,121 restless leg syndrome122 and painful legs and moving toes syndrome.123
Discussion
At least 300 conditions were identified that might cause neurogenic and referred leg pain. Table I is presented as an aetiological classification for neurogenic and referred leg pain and can serve as a checklist to ascertain that all appropriate conditions are evaluated. Each one of these conditions deserves careful consideration and if overlooked might result in a missed diagnosis or even the disaster of the creation of yet another failed back. Our treatment can only be as good as our diagnosis. If the diagnosis is missed, the treatment will obviously fail.

Each one of these conditions deserves careful consideration and if overlooked might result in a missed diagnosis
References
1. Quiros-Moreno R, Lezama-Suárez G, Gomes-Jimenez C. Disc alterarions of the lumbar spine on magnetic resonance images in asymptomatic workers. Rev Med Inst Mex Sequro Soc. 2008;46:185-90. [ Links ]
2. Jensen MC, Brant-Zawadski MN, Obuchowski N, et al. Magnetic resonance imaging of the lumbar spine in people without back pain. N Engl J Med. 1994;331:69-73. [ Links ]
3. Pasnoor M, Dimachkie MM, Kluding P, et al. Diabetic neuropathy part 1: overview and symmetric phenotypes. Neurol Clin. 2013;31(2):425-45. [ Links ]
4. Pasnoor M, Dimachkie MM, Barohn RJ. Diabetic neuropathy part 2: proximal and asymmetric phenotypes. Neurol Clin. 2013:31(2):447-62. [ Links ]
5. Kamenov ZA, Traykov LD. Diabetic autonomic neuropathy. Adv Exp Med Biol. 2012;771:176-93. [ Links ]
6. Indiculla J, Shirazi N, Opacka-Juffry J, et al. Natl Med J India. 2004;17:200-202. [ Links ]
7. Arnold R, Kwai NC, Krishnan AV. Mechanisms of axonal dysfunction in diabetic and uraemic neuropathies. Clin Neurophysiol. 2013 May 14; In print. [ Links ]
8. Lozeron P, Adams D. Metabolic neuropathies. Rev Prat. 2008;58:1903-909. [ Links ]
9. Collins MP, Periquet-Collins I. Nonsystemic vasculitic neuropathy: update on diagnosis, classification, patho-genesis, and treatment. Front Neurol Neurosci. 2009;26:26-66. [ Links ]
10. Gorson KC. Vasculitic neuropathies. An update. Neurol. 2007;13:12-9. [ Links ]
11. Collins MP, Periquet MI. Nonsystemic vasculitic neuropathy. Curr Opin Neurol. 2004;17:587-98. [ Links ]
12. Said G, Lacroix C. Primary and secondary vasculitic neuropathy. J Neurol. 2005;252:633-41. [ Links ]
13. Somer T, Finegold SM. Vasculitides associated with infections, immunization, and antimicrobial drugs. Clin Infection Dis. 2004;36:392-93. [ Links ]
14. Ferrari S, Lanzafame M, Faggian F, et al. Painfull neuropathy vasculitis in 2 patients with longstanding human immunodeficiency virus-1 infection. Scand J Infection Dis. 2004;36:392-93. [ Links ]
15. Oh SJ. Paraneoplastic vasculitis of the peripheral nervous system. Neurol Clin. 1997;15:849-63. [ Links ]
16. Dalmau J. Carcinoma associated paraneoplastic peripheral neuropathy. J Neurol Neurosurg Psychiatry. 1999;67:4. [ Links ]
17. Campbell MJ, Paty DW. Carcinomatous neuromyopathy: Electrophysiological studies. J Neurol Neurosurg Psychiatry. 1974;37:131-41. [ Links ]
18. Tsang BK, Macdnell R. Multiple sclerosis- diagnosis, management and prognosis. Aust Fam Physician. 2011;40:948-55. [ Links ]
19. Piwko C, Desjardins OB, Bereza BG, et al. Pain due to multiple sclerosis: analysis of the prevalence and economic burden in Canada. Pain Res Manag. 2007;12:259-65. [ Links ]
20. Sage JI. Pain in Parkison's disease. Curr Treatm Opti Neurol. 2004;6:191-200. [ Links ]
21. Brettschneider J, Kurent J, Ludolph A. Drug therapy for pain in amyotrophic lateral sclerosis. Cochrane Database of Systematic Reveiws. 2013; Issue 6. [ Links ]
22. Kim JS. Central post-stroke pain or parasthesias in lenticulo-capsular hemorrhages. Neurol. 2003;61:679-82. [ Links ]
23. Berman M, Feldman S, Alter M, et al. Acute transverse myelitis: incidence and etiological considerations. Neurol. 1981;31:966. [ Links ]
24. Campos WK, Almeida de Oliviera YS, Campi de Andrade D, et al. Spinal cord stimulation for the treatment of neuropathic pain related to syringomyelia. Pain Med. 2013;14:767-68. [ Links ]
25. Waseem M, Raja A, Jeun E, et al. Paroxysmal neuropathic pain in an adolescent female with syringomyelia: a review of the literature. Pediatr Emerg Care. 2012;28:472-74. [ Links ]
26. Last, RJ. Anatomy: Regional and Applied, 5th ed. Churchill Livingstone, Edinburgh and London 1973. [ Links ]
27. Yeung JT, J----ohnson JI, Karim AS. Cervical disc herniation with neck pain and contralateral symptoms: a case report. J Med Case Reports. 2012;6:166. [ Links ]
28. Sasaoka R, Nakamura H, Yamano Y. Idiopathic spinal cord herniation in the thoracic spine as a cause of intractable leg pain: Case report and review of the literature. J Spinal Disord Techn. 2003;16:288-94. [ Links ]
29. Langfitt TW, Elliot FA. Pain in the back and legs caused by cervical spinal cord compression. JAMA. 1967;200:112-15. [ Links ]
30. Baastrup C. On the spinous processes of the lumbar vertebrae and the soft tissue between them, and on pathological changes in that region. Acta Radiol. 1933;14:52-54. [ Links ]
32. Sartoris DJ, Resnick D, Haghighi P. Age-related alterations in the vertebral spinous processes and intervening soft tissue: radiologic-pathologic correlation. Am J Roentgenol. 1985;145:1025-30. [ Links ]
33. Chen CKH, Yeh L, Resnick D, et al. Interspinous posterior epidural cyst associated with Baastrup's disease: Report of 10 patients. Am J Roentgenol. 2004;182:191-94. [ Links ]
34. Lilius G, Laasonen EM, Myllynen P, et al. Lumbar facet joint syndrome. A randomised clinical trial. J Bone Joint Surg Br. 1989;71B:681-84. [ Links ]
35. McCall IW, Park WM, O'Brein JP. Induced pain referral from posterior lumbar elements in normal subjects. Spine. 1979;4:441-46. [ Links ]
36. Coulon A, Milin S, Laban E, et al. Pathological characteristics of the most frequent peripheral nerve tumours. Neurosurg. 2009;55:454-58. [ Links ]
37. Lonser RR, Wait SD, Butman JA, et al. Surgical management of lumbosacral nerve root haemangiomablastomas in von Hippel-Lindau syndrome. J Neurosurg. 2003;99:64-69. [ Links ]
38. Glasker S, Bertis A, Pagenstecher A, et al. Characterization of heamangioblastomas of the spinal nerves. Neurosurg. 2005;56:503-509. [ Links ]
39. Tsai MC. Non-Hodgkin's B-cell lymphoma of a lumbar nerve root: A rare cause of lumbar radiculopathy. J Clin Neurosci. 2013;20:1029-31. [ Links ]
40. Jinnai T, Koyama T. Clinical characteristics of nerve sheath tumours: analysis of 149 cases. Neurosurg. 2005;56:510-15. [ Links ]
41. Gupta G, Mammis A, Maniker A. Malignant peripheral nerve sheath tumours. Neurosurg Clin N Am. 2008;19:533-43. [ Links ]
42. Neidre A, Macnab I. Anomalies of the lumbosacral nerve roots: review of 16 cases and classification. Spine. 1983;8:294-99. [ Links ]
43. Kadish LJ, Simmons EH. Anomalies of the lumbosacral nerve roots: An anatomical investigation and myelographic study. J Bone Surg Br. 1984;66-B:411-16. [ Links ]
44. Taghipour M, Razmkon A, Hosseini K. Conjoined lumbosacral nerve roots. Analysis of cases diagnosed intra-operatively. J Spinal Disord Tech. 2009;22:413-16. [ Links ]
45. Transfeldt EE, Robertson D, Bradford DS. Ligaments of the lumbosacral spine and their role in possible extraforaminal spinal nerve entrapment and tethering. J Spinal Disord. 1993;6:507-12. [ Links ]
46. Demetriou GA, Nair MS, Navaratnam R. Right-sided colonic tuberculosis: a rare cause of ilio-psoas abscess. BMJ. 2013; in press. [ Links ]
47. Nathan H, Weizenbluth M, Halperin N. The lumbosacral ligament (LSL), with special emphasis on the lumbosacral tunnel and entrapment of the 5th lumbar nerve. Intern Orthop. 1982;6;197-202. [ Links ]
48. Tsirides E, Upadhyay N, Giannoudes PV. Sacral insufficiency fractures : current concepts of management. Osteoporos Int. 2006;17:1716-25. [ Links ]
49. Jones JW. Insufficiency fracture of the sacrum with displacement and neurological damage: a case report and review of the literature. J Am Geriatr Soc. 1991;39:280-3. [ Links ]
50. Aretxabala I, Fraiz E, Perez-Ruiz F, et al. Sacral insufficiency fractures. High association with pubic rami fractures. Clin Rheumatol. 2000;19:399-401. [ Links ]
51. Cabarrus MC, Ambekar A, Lu Y, et al. MRI and CT of insufficiency fractures of the pelvis and the proximal femur. Am J Roentgenol. 2008;191:995-1001. [ Links ]
52. Strak JG, Fuentes J, Fuentes TI, et al. The history of sacroiliac joint arthrodesis: a critical review and introduction of a new technique. Cur Orthop Pract. 2011;22:545-57. [ Links ]
53. Hermet M, Minichiello E, Flipo RM, et al. Infectious sacroiliitis: a retrospective, multicentre study of 39 adults. BMC Infect Dis. 2012;12:305. [ Links ]
54. Wang J, Tang Q, Xie X, et al. Iliosacral resection of pelvic malignant tumors and with reconstruction non-vascular bilateral fibular autografts. Ann Surg Oncol. 2012;19:4043-51. [ Links ]
55. Deesomchok U, Tumrasvin T. Clinical comparison of patients with ankylosing spondylitis, Reiter's syndrome and psoriatic arthritis. J Med Assoc Thai. 1993;76:61-70. [ Links ]
56. Dyck PJB, Norell JE, Dyck PJ. Non-diabetic lumbosacral radiculoplexus neuropathy. Natural history, outcome and comparison with the diabetic variety. Brain. 2001;124:1197-207. [ Links ]
57. Chanin N, Temesgen Z, Kurtin PJ, et al. HIV lumbosacral radiculoplexus neuropathy mimicking lymphoma: Diffuse infiltrative lymphocytosis syndrome (DILS) restricted to nerve? Muscle Nerve. 2010;41:276-82. [ Links ]
58. Filosto M, Pari E, Cotelli M, et al. MR neurography in diagnosing nondiabetic lumbosacral radiculoplexus neuropathy. J Neuroimaging. 2013; June 10: In press. [ Links ]
59. Petchprapa C, Rosenburg ZS, Sconfienza LM, et al. MR Imaging of entrapment neuropathies of the lower extremity Part 1. The pelvis and hip. Radiographics. 2010;30:983-1000. [ Links ]
60. Grant GA, Goodkin R, Maravilla KR, et al. MR Neurography: diagnostic utility in the surgical treatment of peripheral nerve disorders. Neuroimaging Clin N Am. 2004;14:115-33. [ Links ]
61. Maravilla KR, Brown BC. Imaging of the peripheral nervous system: evaluation of peripheral neuropathy and plexopathy. Am J Neuroradiol. 1998;19:1011-23. [ Links ]
62. Vincent A. Antiganglioside antibodies in the peripheral neuropathies. J Clin Pathol. 1998;51:641-42. [ Links ]
63. Papadopoulos SM, McGillicuddy JE, Albers JW. Unusual case of piriformis muscle syndrome. Arch Neurol. 1990;47:1144-46. [ Links ]
64. Pace JB, Nagle D. Piriformis syndrome. West J Med. 1976;124:435-39. [ Links ]
65. Freiberg AH. Sciatic pain and its relief by operations on the muscle and fascia. Arch Surg. 1937;34:337-50. [ Links ]
66. Kean Chen C, Nizar AJ. Prevalence of piriformis syndrome in chronic low back pain patients. A clinical diagnosis with modified FAIR test. Pain Pract. 2013;13:276-78. [ Links ]
67. Miller TA, White KP, Ross DC. The diagnosis and management of piriformis syndrome: myths and facts. Can J Neurol Sci. 2012;39:577-83. [ Links ]
68. Filler AG, Haynes J, Jordan SE, et al. Sciatica of non-disc origin and piriformis syndrome: diagnosis by magnetic resonance neurography and interventional magnetic resonance imaging with outcome study of resulting treatment. J Neurosurg Spine. 2005;2:99-115. [ Links ]
69. Meknas K, Christensen A, Johansen O. The internal obturator muscle my cause sciatic pain. Pain. 2003;104:275-80. [ Links ]
70. Diop M, Parratte B, Tatu L, et al. Anatomical basis of superior gluteal nerve entrapment syndrome in the suprapiriformis foramen. Surg Radiol Anat. 2002;24:155-59. [ Links ]
71. Yadav Y, Mehta V, Roy S et al. Superior gluteal nerve entrapment between two bellies of piriformis muscle. Int J Anat Variations. 2010;3:203-204. [ Links ]
72. Lowenthal RM, Taylor BV, Jones R et al. Severe persistent sciatic pain and weakness due to gluteal artery pseudoa-neurysm as a complication of bone marrow biopsy. J Clin Neurosci. 2006;13:384-85. [ Links ]
73. Ramesh M, O'Byrne JM, McCarthy N, et al. Damage to the superior gluteal nerve after the Hardinge approach to the hip. J Bone Joint Surg Br. 1996;78:903-906. [ Links ]
74. Collinge C, Coons D, Aschenbrenner J. Risks to the superior gluteal neurovascular bundle during percutaneous iliosacral screw insertion: an anatomical cadaver study. J Orthop Trauma. 2005;19:96-101. [ Links ]
75. Rask MR. Superior gluteal nerve entrapment syndrome. Muscle Nerve. 1980;3:304-307. [ Links ]
76. Fielder J, Miriti K, Bird P. Mycotic aneurysm of the inferior gluteal artery caused by non-typhi salmonella in a man infected with HIV. A case report. J Med Case Rep. 2010;18:273 [ Links ]
77. Hough DM, Wittenberg KH, Pawlina W, et al. Chronic perineal pain caused by pudendal nerve entrapment: anatomy and CT-guided perineum injection technique. Am J Roentgen. 2003;181:561-67. [ Links ]
78. Robert R, Prat-Pradal D, Labat JJ, et al. Anatomical basis of chronic perineal pain: role of the pudendal nerve. Surg Radiol anat. 1998;20:93-98. [ Links ]
79. Shafik A, el-Sherif M, Youssef A, et al. Surgical anatomy of the pudendal nerve and its clinical implications. Clin Anat. 1995;8:110-15. [ Links ]
80. Al-Khodairy A-WT, Bovay P, Gobelet C. Sciatica in the female patient: anatomical considerations, aetiology and review of the literature. Eur Spine J. 2007;16:721-31. [ Links ]
81. Ashkan K, Casey ATH, Powel M, et al. Back pain during pregnancy and after childbirth: an unusual case not to miss. JR Soc Med. 1998;19:88-90. [ Links ]
82. Tubridy N, Redmond JM. Neurological symptoms attributed to epidural analgesia in labour: an observational study of seven cases. Br J Obstet Gynecol. 1996;103:832-33. [ Links ]
83. Gonik B, Stringer CA, Cotton, DB. Intrapartum maternal lumbosacral plexopathy. Obstet Gynecol. 1984;63:45S-6S. [ Links ]
84. Ku A, Kern H, Lachman E, et al. Sciatic nerve impingement from piriformis haematoma due to prolonged labour. Muscle Nerve. 1995;18:789-90. [ Links ]
85. Heffernan LPM, Fraser RC, Purdy RA. L-5 Radiculopathy secondary to uterine leiomyoma in a primigravid patient. Am J Obstet Gynecol. 1980;138:460-61. [ Links ]
86. Vilos GA, Vilos AW, Haebe JJ. Laparoscopic findings, management, histopathology, and outcome of 25 woman with cyclic leg pain. J Am Assoc Gynecol Laparosc. 2002;9:145-51. [ Links ]
87. Ras S. Complications of vaginal surgery. In: Raz S, editor. Atlas of transvaginal surgery. Philadelphia: Saunders;1992, pp 37-47. [ Links ]
88. Tokita A, Ikari K. Tsukahara S, et al. Iliopsoas bursitis-associated femoral nerve neuropathy exacerbated after internal fixation of an intertrochanteric hip fracture in rheumatoid arthritis: a case report. Mod Rheumatol.2008;18:394-8. [ Links ]
89. Tatsumura M, Mishima H, Shiina I, et al. Femoral nerve palsy caused by a huge iliopectineal synovitis extending to the iliac fossa in a rheumatoid arthritis case. Mod Rheumatol. 2008;18:81-85. [ Links ]
90. Bodur H. Nontraumatic focal neuropathies. Turk J Phys Med Rehab. 2012;58:114-20. [ Links ]
91. Adams D. Recent advances in the treatment of familial amyloid polyneuropathy. Ther Adv Neurol Disord. 2013;6:129-39. [ Links ]
92. Hollis MH, Lemay DE. Nerve entrapment syndromes of the lower extremity. Medscape Reference. 2010;Nov 10. [ Links ]
93. McCoy P, Bell S, Bradshaw C. Nerve entrapments of the lower leg, ankle and foot in sport. Sports Med. 2002;32:371-91. [ Links ]
94. Flu HC, Breslau PJ, Hamming JF, et al. A prospective study of saphenous nerve injury after total great saphenous vein stripping. Dermtol Surg. 2008;34:1333-39. [ Links ]
95. Kaminsky F. Peroneus palsy by crossing the legs. JAMA. 1947;134:206. [ Links ]
96. Anselmi SJ. Common peroneal nerve compression. J Am Podiatr Med Assoc. 2006;96:413-17. [ Links ]
97. Kopell HP, Thompson W. Peripheral nerve entrapments of the lower extremity. New Engl J Med. 1962;266:16-19. [ Links ]
98. DiDomenico LA, Masternick EB. Anterior tarsal syndrome. Electromyography. 1968;8:123-34. [ Links ]
99. Donovan A, Rosenburg ZS, Cavalcanti CF. MR Imaging of entrapment neuropathies of the lower extremity. Radiographics. 2012;30:1001-19. [ Links ]
100. Kennedy JG, Baxter DE. Nerve disorders in dancers. Clin Sports Med. 2008;27:329-34. [ Links ]
101. Sammarco GF, Conti SF. Tarsal tunnel syndrome caused by an anomalous muscle. J Bone Joint Surg Am. 1994;76:1308-14. [ Links ]
102. Coert J, Dellon A. Clinical implications of the surgical anatomy of the sural nerve. Plast Reconstr Surg. 1994;94:850-55. [ Links ]
103. Pringle R, Protheroe K, Mukherjee S. Entrapment neuropathy of the sural nerve. J Bone Joint Surg Br. 1974;56:465-68. [ Links ]
104. Schon L. Nerve entrapment, neuropathy and nerve dysfunction in athletes. Orth Clin North Am. 1994;25:47-59. [ Links ]
105. Schon L. Chronic pain. In: Meyerson's foot and ankle disorder. Vol 2. Philadelphia. Saunders, 2000;851-81. [ Links ]
106. Morscher E, Ulrich J, Dick W. Morton's intermetatarsal neuroma: morphology and histological substrate. Foot Ankle Int. 2000;21:558-62. [ Links ]
107. Merritt GN, Subotnick SI. Medial plantar digital proper nerve syndrome (Joplin's neuroma): typical presentation. J Foot Surg. 1982;21:166-69. [ Links ]
108. Still GP, Fowler MB. Joplin's neuroma or compression neuropathy of the plantar proper digital nerve to the hallux: clinicopathologic study of three cases. J Foot Ankle Surg.1998;37:524-30. [ Links ]
109. Pathria M, Bradshaw J. Muscle MR imaging- Part 2. Non-traumatic changes. In: The Radiology Assistant; Jan 2010. [ Links ]
110. Reinking MF. Exercises related leg pain (ERLP): A review of the literature. North Am J Sports Phys Ther. 2007;2:170-80. [ Links ]
111. Verma RB, Sherman O. Athletic stress fracture: Part1. History, epidemiology, physiology, risk factors, radiology, diagnosis, and treatment. Am J Orthop. 2001;30:798-806. [ Links ]
112. Mubarak SJ, Gould RN, Lee YF, et al. The medial tibial stress syndrome. A cause of shin splints. Am J Sports Med. 1982;10:201-205. [ Links ]
113. Mavor GE. The anterior tibial syndrome. J Bone Joint Surg Br. 1956;38B:513-7. [ Links ]
114. Khan KM, Cook JL, Taunton JE, et al. Overuse tendinosis, not tendinitis. Phys Sportsmed. 2000;28:38-47. [ Links ]
115. Kaufman KR, Brodine SK, Shaffer RA, et al. The effect of foot structure and range of motion on musculoskeletal overuse injuries. Am J Sports Med. 1999;27:585-93. [ Links ]
116. Hirose CB, McGarvey WC. Peripheral nerve entrapments. Foot Ankle Clin. 2004;9:255-69. [ Links ]
117. Korkola M, Amendola A. Exercise-induced pain. Phys Sportsmed. 2001;29:35-38. [ Links ]
118. Bradshaw C. Exercise-related lower leg pain: Vascular. Med Sci Sports Exerc. 2000;32(3 Suppl):S34-6. [ Links ]
119. McCrory P. Exercise related leg pain: Neurological perspective. Med Sci Sports Exerc. 2000;32(3 Suppl):S11-4. [ Links ]
120. Grande LA, Loeser JD, Ozuna J, et al. Complex regional pain syndrome as a stress response. Pain. 2004;110:495-98. [ Links ]
121. Wasner G, Schattschneider J, Binder A, et al. Complex regional pain syndrome- diagnostic, mechanisms, CNS involvement and therapy. Spinal Cord. 2003;41:61-75. [ Links ]
122. Bogan RK, Cheray JA. Restless legs syndrome: A review of diagnosis and management in primary care. Postgrad Med. 2003;125: 99-111. [ Links ]
123. Reich SG. Painfull legs and moving toes. Handb Clin Neurol. 2011;100:375-83. [ Links ]
 Correspondence:
Correspondence:
Dr JA Louw
PO Box 24
Moreleta Plaza 0167
The content of this article is the sole work of the authors, and no benefit of any form has been received or will be received from any commercial party.
This article is also available online on the SAOA website (www.saoa.org.za) and the SciELO website (www.scielo.org.za). Follow the directions on the Contents page of this journal to access it.














